Functional Groups for CHM 115 S26
1/66
Earn XP
Description and Tags
For CHM 115 Exam 3
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
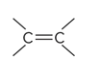
compound type
alkene
prefix/suffix
-ene
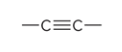
compound type
alkyne
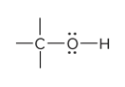
compound type
alcohol
alcohol is recognizable for what traits?
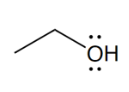
Alcohols are recognizable for having a hydroxyl group (-OH) attached via single bond to a carbon atom
What makes alcohol different from carboxylic acid?
the -OH group in an alcohol is singularly bonded to a C atom; carboxylic acid is an -OH group bonded to a C atom that is doubled bonded to an Oxygen atom
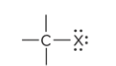
compound type
haloalkane
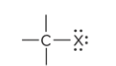
prefix/suffix
halo-
what is the defining trait for a haloalkane
a haloalkane is a halogen atom bonded to a Carbon
What are the halogen atoms?
F, Cl, Br, I
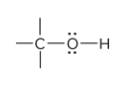
prefix/suffix
-ol
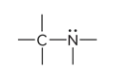
compound type
amine
What is the defining trait for amines?
A nitrogen atom bonded to one or more carbon atoms
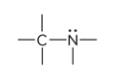
prefix/suffix
-amine
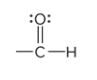
compound type
aldehyde
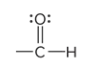
prefix/suffix
-al
What is the defining trait of aldehydes?
a carbonyl group at the end of a carbon chain; the C atom is also bonded to at least one H atom
describe a carbonyl group
a C atom double bonded to an O atom
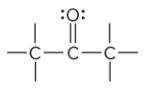
compound type
ketone
prefix/suffix
-one
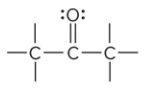
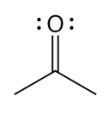
what is the defining trait of a ketone?
a carbonyl group that is in a carbon chain; is bound to two other C atoms
What is the key difference between a ketone and aldehyde?
a ketones have a carbonyl group within a carbon chain; aldehydes are a carbonyl group bound to a hydrogen at the end of a carbon chain
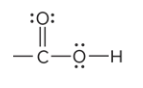
compound type
carboxylic acid
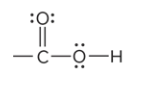
prefix/suffix
-oic acid
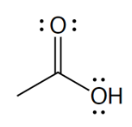
what is the defining feature of a carboxylic acid funtional group
the C atom of carbonyl group bound to an O of a hydroxyl group; located at the end of a carbon chain
What is the structure of a carboxylic acid?
COOH
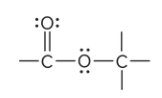
compound type
ester
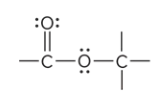
prefix/suffix
-oate
What is the defining feature of an ester functional group?
the C of a carbonyl group bound to an O atom; O atom is bound to another atom other than H (R’)
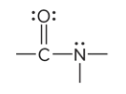
compound type
amide
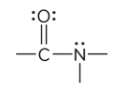
prefix/suffix
-amide
What is the defining feature of an amide functional group?
a carbonyl group bound to a nitrogen atom
What makes an amine and amide different?
an amine: is a carbon bound to a nitrogen atom while an amide is a carbonyl group attached to a nitrogen atom
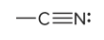
compound type
nitrile
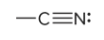
prefix/suffix
-nitrile
what is the defining feature of a nitrile functional group?
a carbon atom triple bonded with a nitrogen atom; typically appear at the end of structures
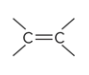
what is the defining feature of alkenes?
a carbon-carbon double bond
what is the defining feature of a alkyne
a carbon-carbon triple bond
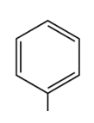
compound type
phenyl
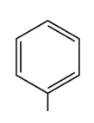
prefix/suffix
phenyl-
What is the defining feature of a phenyl functional group?
a benzene ring attached to a carbon chain with one hydrogen atom removed; attached to another atom (R).
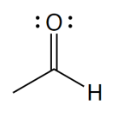
functional group
aldehyde
functional group
alcohol
functional group
ketone
functional group
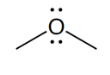
ether
functional group
carboxylic acid
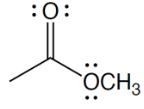
functional group
ester
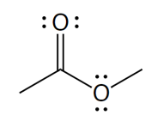
functional group
ester
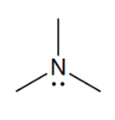
functional group
amine
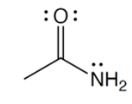
functional group
amide
alkanes have what electron geometry?
tetrahedral electron geometry due to the sp^3 hybridization of the carbon atoms.
alkenes have what electron geometry?
trigonal planar electron geometry due to the sp^2 hybridization of the carbon atoms.
alkynes have what electron geometry?
linear electron geometry due to the sp hybridization of the carbon atoms.
Describe Aromatic carbons
compound of C and H, characterized by a ring structure and resonance; exhibit delocalized π electrons.
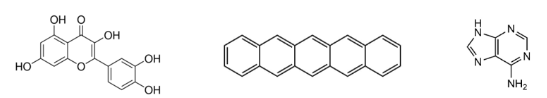
these are an example of
aromatic hydrocarbons/compounds
Heteroatoms
atoms in organic compounds that are not carbon or hydrogen, such as nitrogen, oxygen, sulfur, or halogens. They can influence the chemical properties and reactivity of the compounds.
Describe Cycloalkanes
carbon atoms arranged in a ring, with single bonds connecting the carbons (no ends); no double or triple bonds.
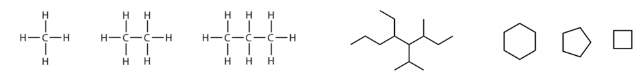
an example of
alkanes
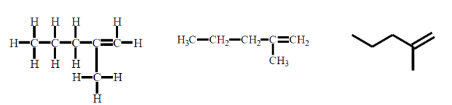
an example of
alkenes
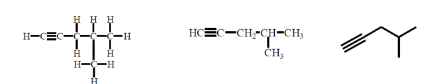
an example of
alkynes
An expanded formula shows
all atoms and bonds
a condensed formula shows
bonds between C and not H
a skeleton formula shows
bonds between C; does not show H bonds
descibe constitutional isomers
compounds with the same molecular formula but different arrangements of bonded atoms and properties.
descibe geometric isomers
same molecular formula and connectivity, but different spatial arrangements due to restricted rotation
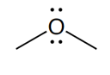
prefix/suffix
alkoxy-
What is the defining feature of an ether functional group?
a single oxygen atom between 2 C atoms (R-O-R’); does not include a carbonyl group