Organic Chem 31 Functional Groups
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
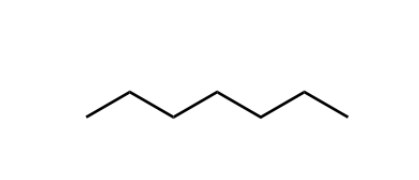
Alkane (sp3)
comprising solely sp3 carbons and hydrogens
Amine (sp3)
Nitrogen atoms singly-bonded to a combination of hydrogens and
sp3 carbons. The attached carbons may not be attached to any atom types other than carbon and hydrogen.
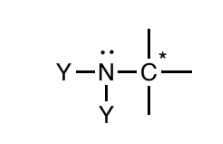
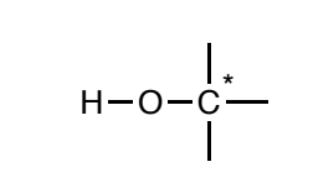
Alcohol (sp3)
Oxygen atoms singly-bonded to one hydrogen and one sp3
carbon.The carbon may not be attached to any atom types other than carbon and hydrogen.
Thiol (sp3)
Sulfur atoms singly-bonded to one hydrogen and one sp3 carbon. Further, the carbon may not be attached to any atom types other than carbon and hydrogen.
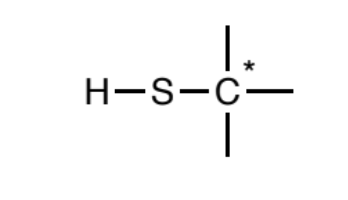
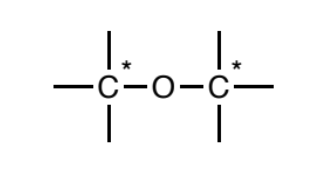
Ether (sp3)
Oxygen atoms singly-bonded to two sp3 carbons. Further, the carbons
may not be attached to any atom types other than carbon and hydrogen.
Thioether (sp3)
Sulfur atoms singly-bonded to two sp3 carbons. Further, the carbons may not be attached to any atom types other than carbon and hydrogen.
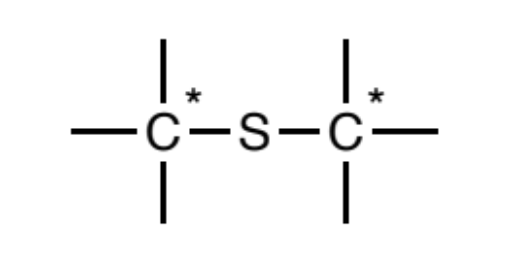
Alkyl halides (sp3)
a halogen (F, Cl, Br, I) bonded to a sp3 carbon. Further,
that carbon may not be attached to any atom types other than carbon and hydrogen.
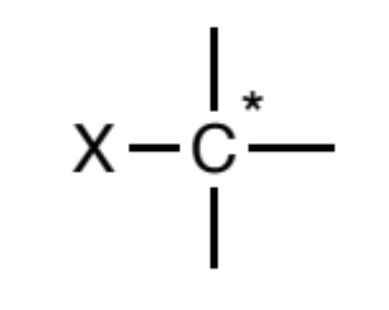
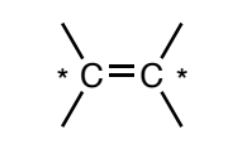
Alkene (sp2)
Two sp2 carbons forming a double bond. In general, only carbons and hydrogens can be attached to the alkene carbons without changing the functional group. However, if halogens are attached it will typically still be considered an alkene.
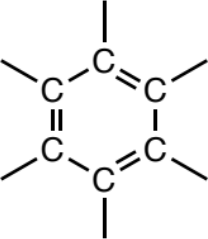
Aromatic (benzene) ring
Six-membered rings comprising solely sp2 carbons and having the appearance of alternating single and double bonds in the ring. A wide variety of atom types may be attached to the ring without changing the functional group.
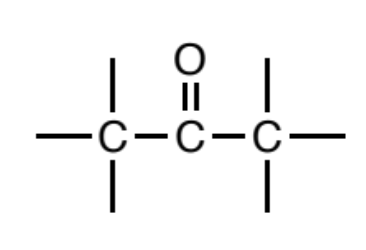
Ketone
A carbonyl (C=O) where carbons are attached to both sides of the
carbonyl carbon.
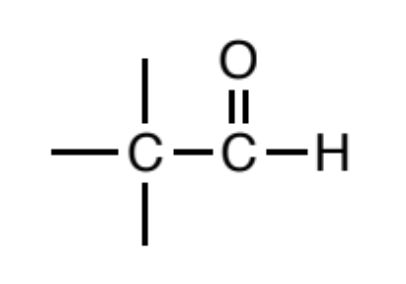
Aldehyde
a carbonyl (C=O) where a hydrogen is attached to one side of the carbonyl carbon and the other side is hydrogen (for the special case of formaldehyde) or carbon.
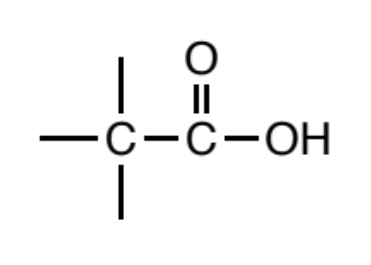
Carboxylic acid
as a carbonyl (C=O) where a carbon or hydrogen is attached to one side of the carbonyl carbon and the other side is oxygen. In turn, this oxygen MUST be bonded to a hydrogen on its other side.
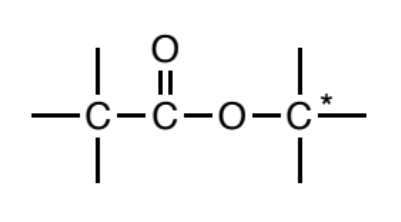
Ester
a carbonyl (C=O) where a carbon or hydrogen is attached to one side of the carbonyl carbon and the other side is oxygen. In turn, this oxygen MUST be bonded to another carbon on its other side, and this carbon should not be bonded to any other non C/H atoms.
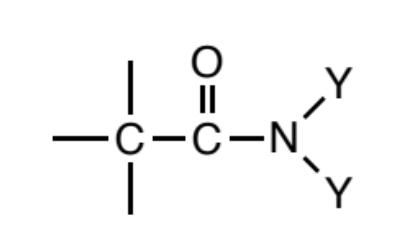
Amide
a carbonyl (C=O) where a carbon or hydrogen is attached to one side of the carbonyl carbon and the other side is nitrogen. In turn, this nitrogen can be attached to any combination of C/H atoms, but those attached carbons (if any) should not be bonded to any other non C/H atoms.
Acyl Halide
a carbonyl (C=O) where a where a carbon or hydrogen is attached to one side of the carbonyl carbon and the other side is a halogen.
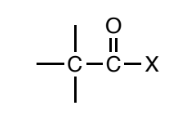
Anhydride
two carbonyls (C=O) connected through an oxygen. The non-oxygen sides of the carbonyl carbons may be carbons or hydrogens.
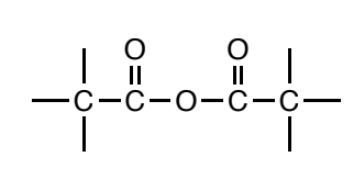
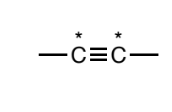
Alkyne
two sp carbons triply bound to each other. These two carbons must not have any non C/H atoms attached to them.
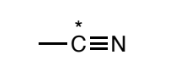
Nitrile
by a carbon atom triply bound to a nitrogen atom. The carbon must not have any other non C/H atoms attached to it.