Immuno Test 3
1/94
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
95 Terms
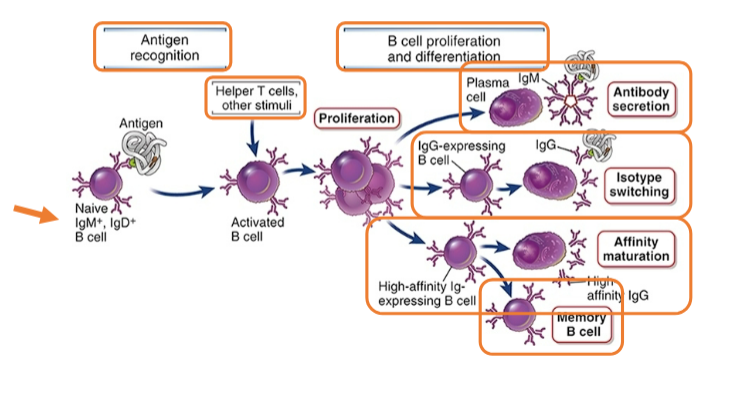
Phases of the humoral immune response
In one week, a single B cell may give rise to ~5000 antibody secreting cells, which collectively produce > 1 trillion antibody molecules per day
Antibody responses to protein antigens
Require T cell assistance
Involve follicular B cells (lymph nodes)
We’ll focus on T-dependent responses
Antibody response to Multivalent antigens
Repeating determinants: polysaccharides and nucleic acids
No T cell help required
Rapid and simple
Involve marginal zone B cells (spleen) and B1 cells (mucosa)
“Helper T cell” term comes from discovery that
T cells stimulate B cells to produce antibodies
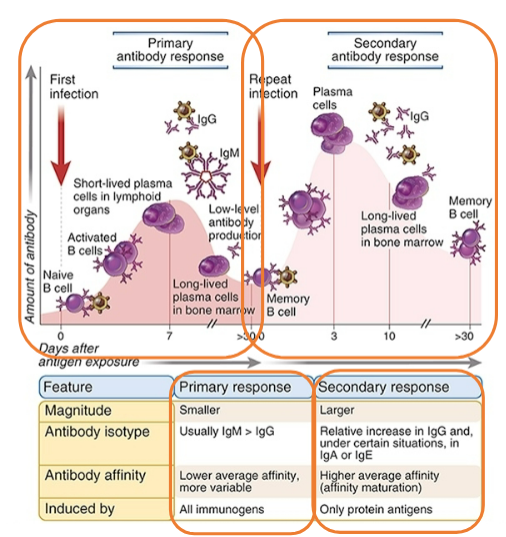
Summary of primary and secondary antibody responses
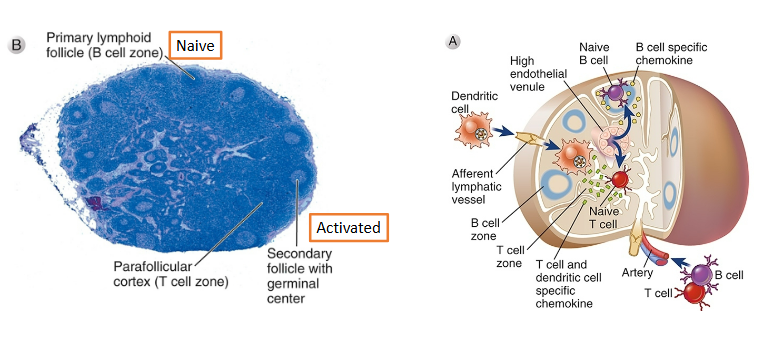
Lymphocytes are organized with lymph nodes
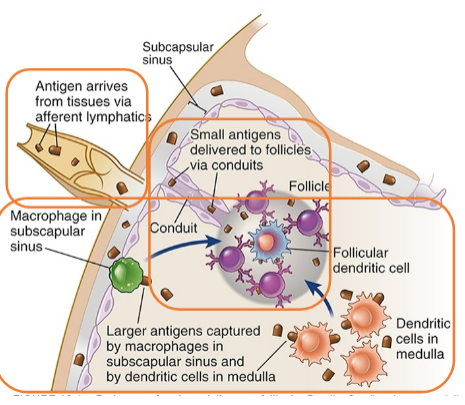
Antigen delivery to naive follicular B cells
Small antigens delivered to follicular B cells via conduits
Larger antigens captured by macrophages in subcapsular sinus or resident DCs in medulla
Antigen transferred to FDCs for display to B cells
Antigen generally in its native, folded three-dimensional conformation
Recognition of the antigen by appropriate B cell receptor initiates B cell activation
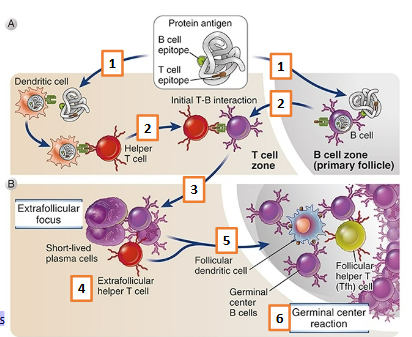
Sequence of events in humoral immune responses to T cell-dependent protein antigens
1. Independent recognition of antigen by Th cells and B cells
2. Activated T cells and B cells migrate toward each other and
interact at interface of T-cell zone and B-cell zone
3. T cell-dependent B cell proliferation and differentiation creates
extrafollicular focus (where B cells proliferate, undergo isotype
switching and differentiate into short-lived plasma cells)
5. Tfh cells and activated B cells migrate back to the follicle to
form a cell cluster termed the germinal center.
4. Some T cells develop into follicular helper T cells
6. The late events in B cell response occur in germinal centers,
including affinity maturation, additional isotype switching,
memory B cell generation, & generation of long-lived plasma cells
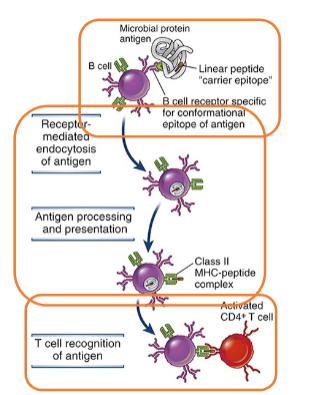
B cells present protein antigen to Th cells
Antigen recognition is necessary but NOT always sufficient to stimulate significant B cell proliferation and differentiation
=> full response to protein antigen also requires Th cells
B cells present peptide antigens to Th cells via class II MHC
=> B cells are type of APC!
Peptide presented to Th cell is the “same” peptide that activated the naïve CD4+ T cell
Activated Th cells in turn promote B cell response via CD40L and cytokines
Germinal centers in secondary lymphoid organs Three identifiable regions
Mantle zone
Germinal center
Light zone
Dark zone
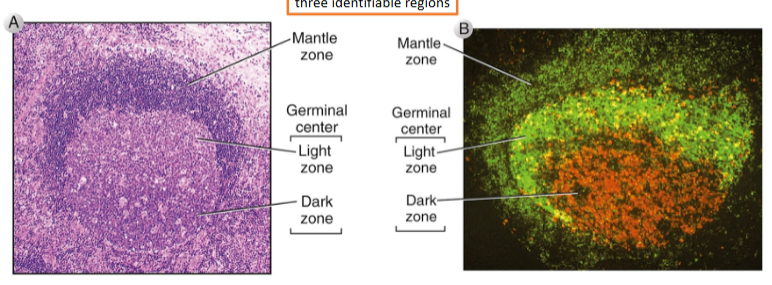
Germinal center light zone
abundant follicular dendritic cells
Germinal center dark zone
mainly proliferating B cells
Germinal center mantle zone
mainly naive B cells
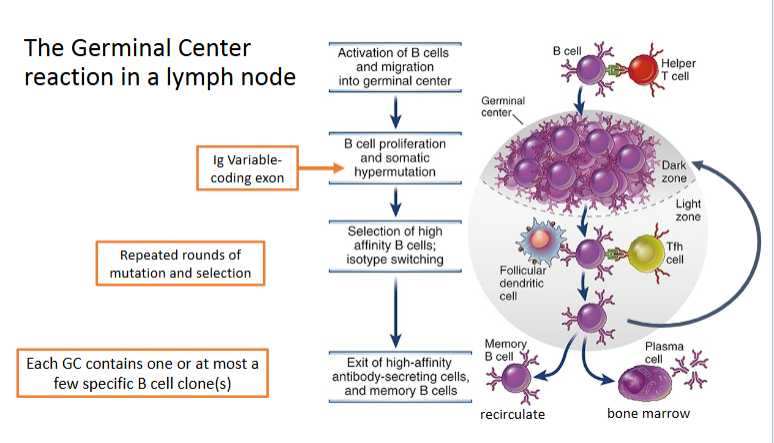
Germinal center reaction in a lymph node
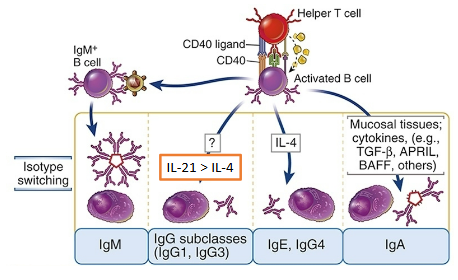
Ig heavy chain isotype switching
Driven by Tfh cells in light zone
Does not affect antibody specificity
Regulated by Th-produced cytokines activated by microbe type
Occurs via “class switch recombination” (CSR) of Ig heavy chain
Previously formed VDJ exon is placed adjacent to different downstream constant region (Intervening sequence is deleted
Overview of affinity maturation
Depends on “somatic hypermutation”
Early in the immune response (after gene rearrangement)
As T-dependent humoral responses progress
Somatic mutations in Ig V genes => selection of high affinity B cells
Higher affinity antibodies are more effective at neutralizing and eliminating microbes and toxins
Both CSR and SHM depend on activation-induced cytidine deaminase (AID)
Induces C to U mutations subject to error-prone repair
B cell selection in germinal centers
Many somatic mutations result in reduction or loss of
antigen-binding ability
B cells experience alternating rounds of selection in the
light zone and proliferation/mutation in the dark zone
In light zone, B cells die by apoptosis unless “rescued” by
binding to antigen
After mutation, B cells undergo form of Darwinian selection
As more antibody molecules are produced, more of the
antigen is eliminated
Thus, B cells must express antigen receptors with
increasingly higher affinity to avoid apoptosis
GCs are sites of significant B cell turnover
Re-enters dark zone or becomes plasma cell or memory cell
Plasma cells =
terminally-differentiated B cells committed to Ab production
short lived plasma cells
Generated in early T-dependent responses in extrafollicular foci & in T-independent responses
• Generally found in secondary lymphoid organs and peripheral non-lymphoid tissues
long lived plasma cells
• Generated in T-dependent germinal centers
• Precursors called plasmablasts enter the circulatory system, then move to the bone marrow
where they complete differentiation
• Survive long periods without antigen stimulation due to expression of anti-apoptotic proteins
Antibody production in long-lived plasma cells
• Bone marrow becomes major site of antibody production 2-3 weeks after exposure
• Plasma cells in marrow may continue to secrete antibodies for decades
• Antibodies enter circulation and mucosal secretions (plasma cells do not recirculate)
• Plasma cells are morphologically distinct from the typical B cell
=> Major structural alterations in the ER and other components of secretory pathway
Memory B cells
• Derived from some activated B cells in the germinal center
• May remain in the lymphoid organ of origin or enter circulation
• Responsible for much of enhanced secondary antibody response to protein antigens
• Effective vaccines against microbes induce both efficient affinity maturation and
memory B cells => these only occur if vaccine also activates helper T cells
Effector mechanisms of humoral immunity
• Secreted antibodies eliminate antigens and associated microbes
• Involves collaboration with components of the innate immune system
• Antibodies perform effector functions at locations distant from site of production
• Primary targets include:
• Extracellular bacteria and fungi
• Viruses (pre-infection and post-release)
• Primary basis of vaccine protection
• May contribute to tissue damage in certain immune diseases and transplant rejection
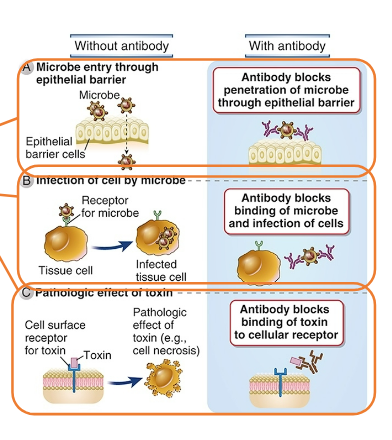
Neutralization of microbes and toxins by antibodies
Antibody binding:
• increases size of antigen or microbe
• blocks ability of microbe to infect host cell
• blocks ability of microbial toxin to harm host cell
Mainly IgA in gut
Mainly IgG in blood
Requires only antigen-binding region (Fab)
=> Any isotype could “work”
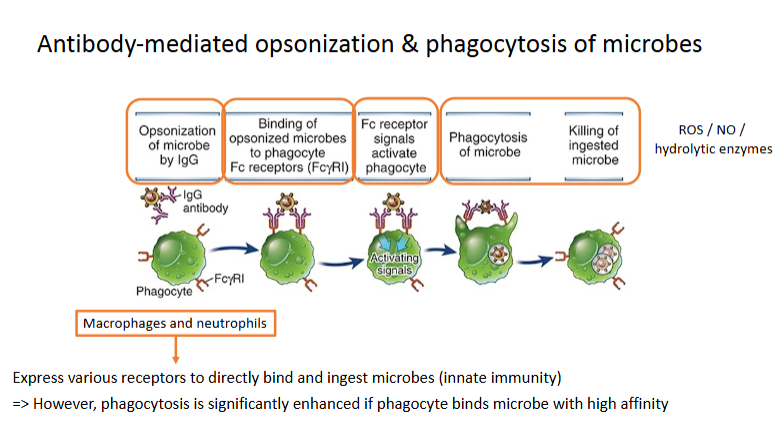
Antibody-mediated opsonization
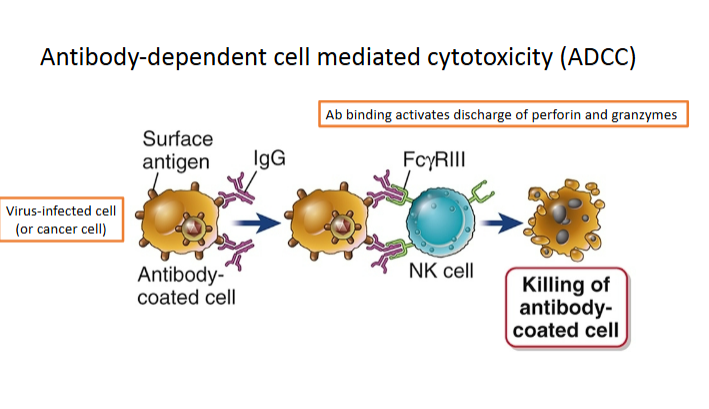
Antibody dependent cell mediated cytotoxicity
Complement activation depends on a proteolytic cascade
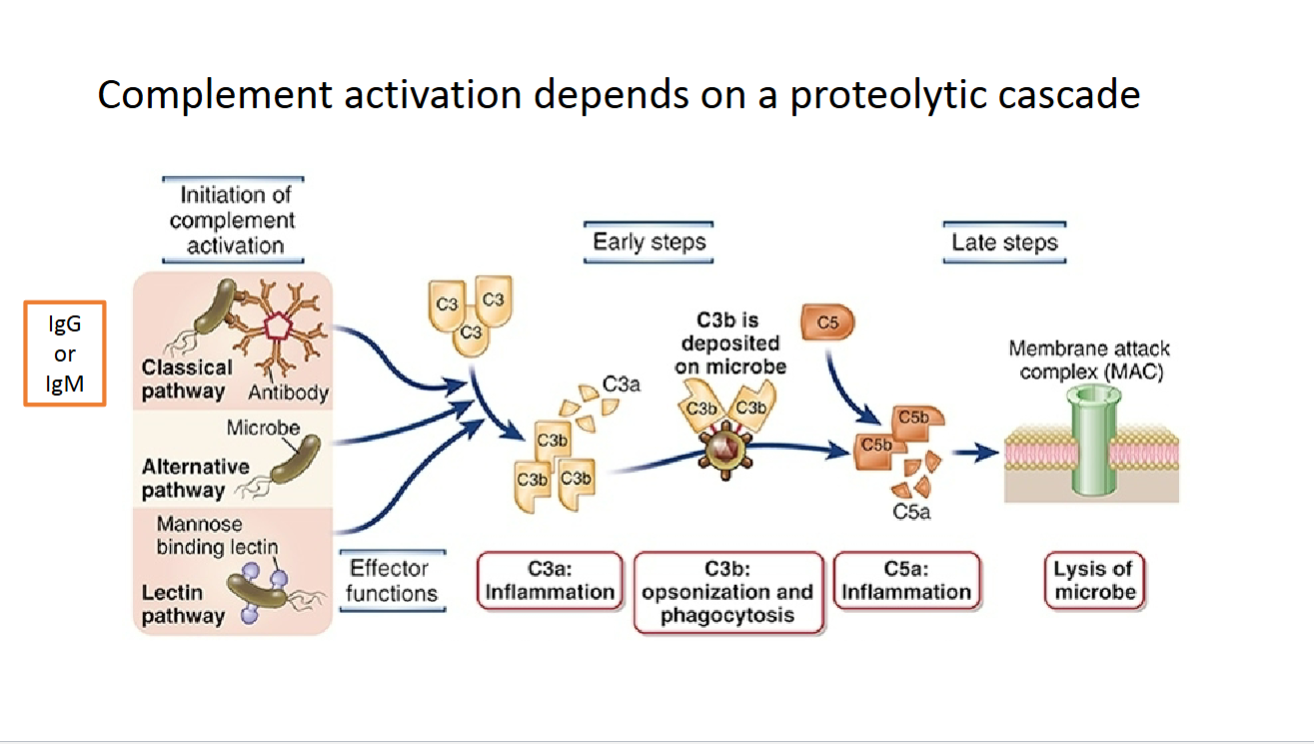
Functions of complements (opsonization and phagocytosis)
Neutrophils and macrophages
Last step caused by activating signals
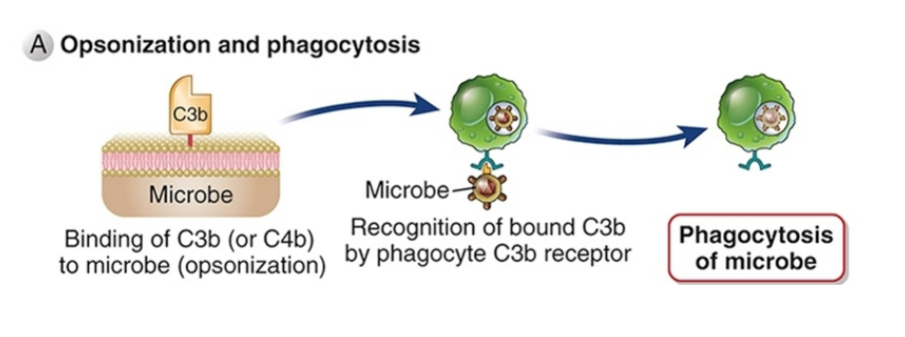
Function of complements (stimulation of inflammatory reactions)
And activation of endothelial cells
Degranulation of mast cells releases multiple “vasoactive mediators” (e.g., histamine)
C3a and C3b = “anaphylatoxins” = can produce anaphylactic shock
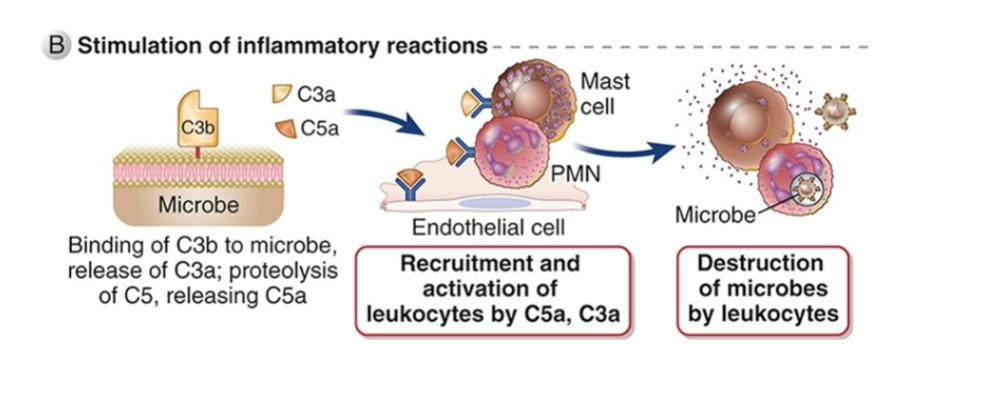
Functions of complement (complement-mediated cytolysis)
MAC related to perforin
Creates pores in microbial cell membrane
Pores allow movement of water into cell
Specialized immunity
Regional immune systems
Privileged tissues
Regional immune systems
Provide protection against microbial challenges encountered at certain locations
Allow for appropriate balance with nonpathogenic commensal organisms
Examples:
Mucosal epithelial barriers: gastrointestinal, respiratory, urogenital
Cutaneous (skin)
Regional immune systems share certain basic features
But each contains own specialized anatomic features, cell types, and molecules
Challenges for GI immunity
Large surface area to defend
Abundance and diversity of nonpathogenic, commensal microbes
Consistent exposure to diverse food antigens
Inflammation has negative impact on necessary gi functions
Must limit response to commensal and food antigens
But must still be able to detect small pathogen antigen “signals” amongst background
The gastrointestinal immune system
Peyer’s patch
Organized secondary lympoid tissue
Contains B cells, T cells, DCs, Macrophages
“Mucosal-associated lymphoid tissue” = MALT
“Gut-associated lymphoid tissue” = GAL
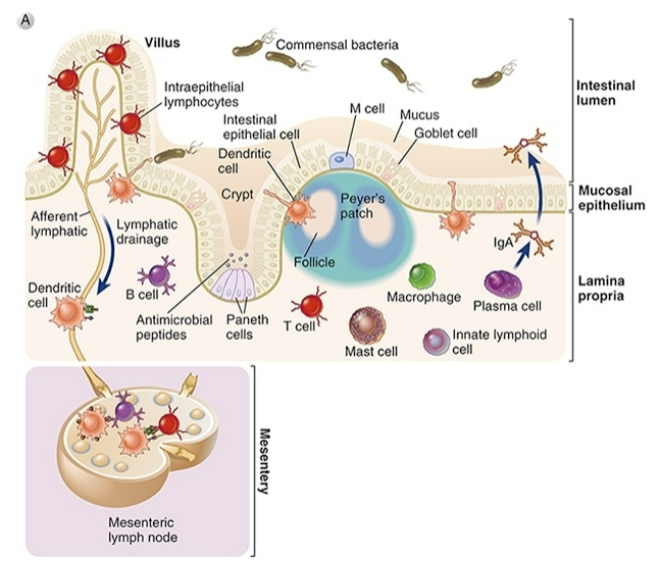
The gastrointestinal immune system: Innate Immunity
Barrier aspects
• Mucus secreted by Goblet cells
• Defensins secreted by Paneth cells
• Epithelial sheet sealed by tight junctions
• Basement membrane (ECM)
TLR’s and other PAMP / DAMP receptors on various cells
trigger innate immune response to pathogens
BUT inflammatory response to commensals is limited by
multiple mechanisms:
• TLRs on basolateral surface of epithelial cells
• TLRs in GI tract require high threshold for activation
• Phagocytes secrete IL-10 (inhibitory cytokine)
The gastrointestinal immune system: Adaptive Immunity
Mainly humoral immunity (via IgA)
directed at microbes in the lumen
Dominance of IgA because:
1. B cells in gut region class switch to IgA
2. IgA-expressing B cells home to the gut
Abundant Treg cells act to
control inflammatory reactions
Gut-antigen sampling: the role of M cells
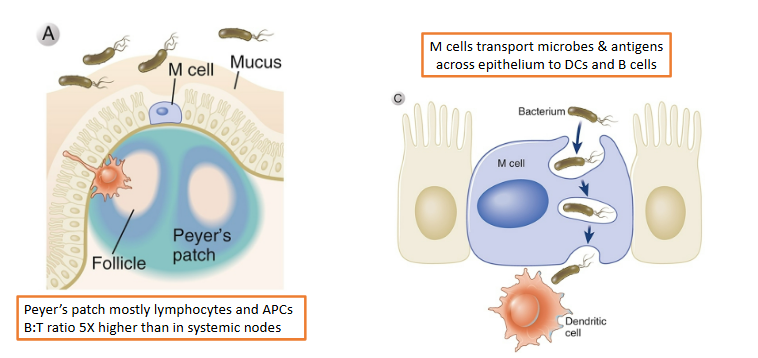
Gut-antigen sampling: intestinal dendritic cells
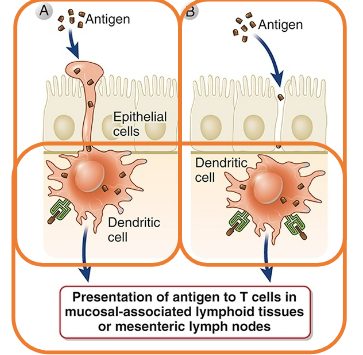
The gastrointestinal immune system: Humoral Immunity
Major function
=> neutralize microbes in gut lumen via IgA (termed “secretory immunity”)
Transcytosis of IgA
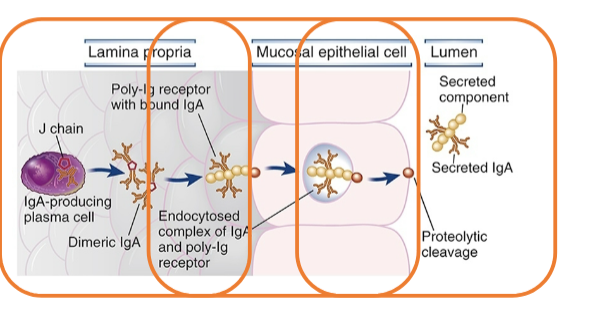
The gastrointestinal immune system: T cell-mediated Immunity
T cells found:
• within epithelia
• throughout lamina propria
• around and within Peyer’s patches
• in draining lymph nodes
Most intraepithelial T cells are CD8+
Most in / around Peyer’s patches or GALTs are CD4+ Th or Tfh or Treg
Regulating immune responses in the gut
Understanding remains incomplete but key factors include:
• Abundant regulatory T cells prevent inflammatory actions against commensals
• Inhibitory cytokines (especially IL-10) from Treg and other cells
• The commensal microbiome influences gut and systemic immune responses
Several inflammatory diseases of the GI tract are related to unregulated responses
to commensal organisms or to food antigens in genetically susceptible individuals
Immune privilege - protection from immune response
Occurs in tissues where inflammation carries high risk of organ damage / failure
Examples: eye, brain, testes, fetus
Not well understood but some common contributing mechanisms
blood- tissue barrier: tight junctions seal endothelial cells of vasculature
Secretion of anti-inflammatory factors and/or signals that inactivate T cells
Reduced number of dendritic cells
Higher threshold for macrophage activation
Reduced vasculature and draining lymphatics
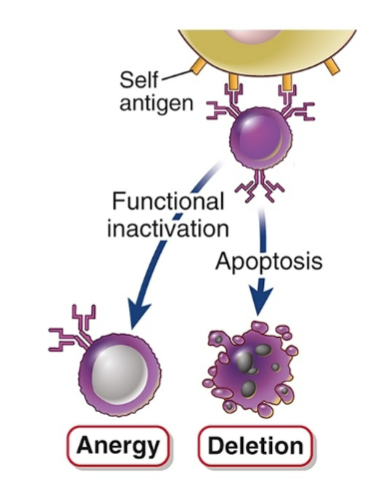
Immunologic tolerance
The unresponsiveness to an antigen that is induced by previous exposure to that antigen
Encountering an antigen can lead to either
Lymphocyte activation and an immune response (what we have focused on so far)
Lymphocyte inactivation or elimination (which leads to tolerance)
Which occurs depends on
Affinity of receptor-antigen interaction
Conditions of antigen exposure
Presence or absence of co-stimulators
Failure of self tolerance leads to immune reactions against self antigens (autoimmunity)
Central tolerance to self antigens
Immune lymphocytes
generative/primary/central lymphoid organs
Three possible mechanisms
Not perfect
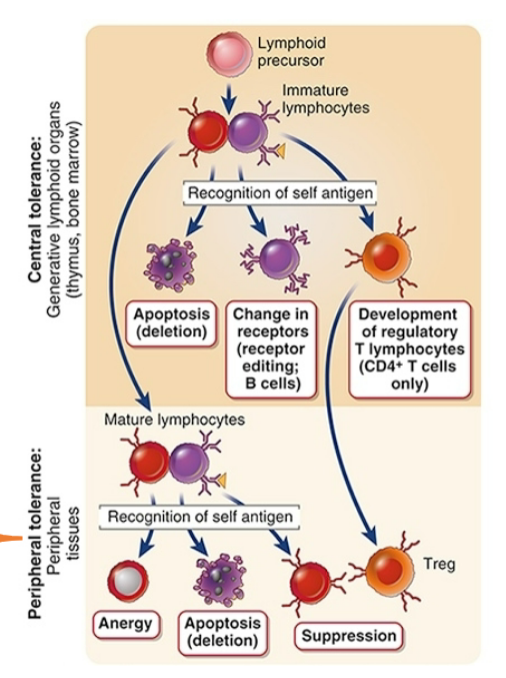
peripheral tolerance to self antigens
Mature lymphocytes
Peripheral tissues
Three possible mechanisms
Backup for central mechanisms
Maintain unresponsiveness to self antigens expressed
Only in peripheral tissues
Only in adults
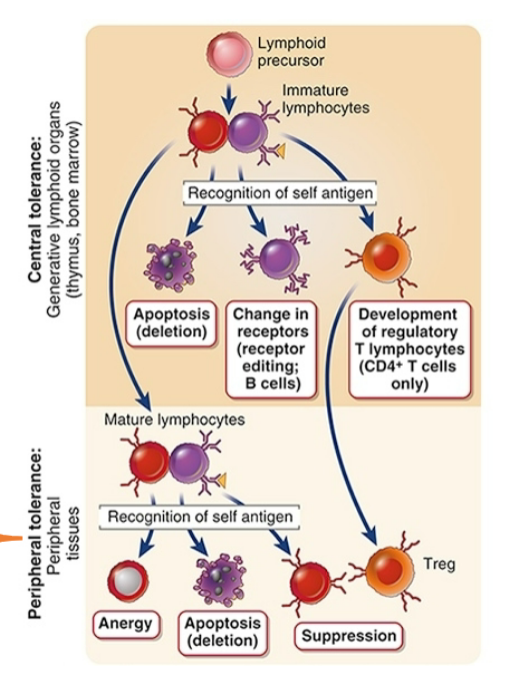
Control tolerance for immature T cells occurs in the thymus
Occurs for both CD8+ and CD4+ cells that express high-affinity receptor against self antigens
Most thymocytes that recognize self
Negative selection : deletion
Some CD4+ thymocytes that recognize self
Development of regulatory T cells
Unclear what factors determine outcome
Peripheral tolerance in T cells
Interaction with activated DC displaying both foreign peptide antigen and B7 costimulator
Three mechanisms provide T cell tolerance to tissue specific antigens not normally abundant in the thymus
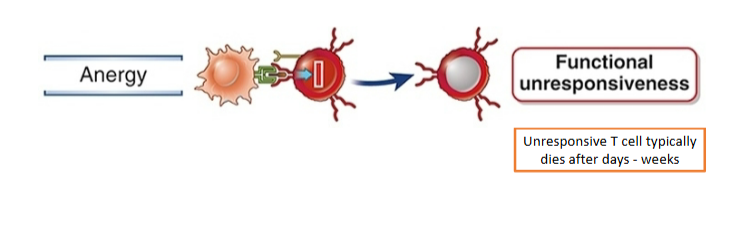
T cell anergy favored by prolonged exposure to self antigen presented by “resting DCs”
=> DCs are not activated by self antigen so don’t express B7 or other costimulators
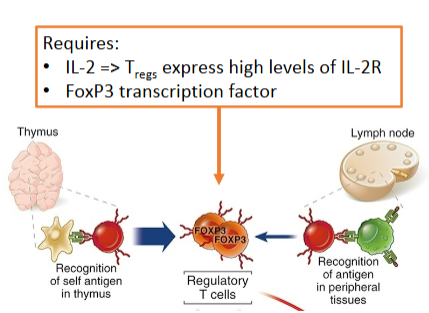
Regulatory T cells (Tregs) may develop centrally or peripherally
Regulatory T cells (Tregs) have multiple targets
T cell activation
Effector T cell activity
B cell activation
NK proliferation
IL-10 production inhibits DCs & macrophages
Defects in Treg-mediated suppression of immune response contribute to various autoimmune disease
Antibody avidity is important for B cell tolerance
Avidity = total strength of interaction between antibody and antigen
Depends on
Affinity of binding to antigen
Number of antigen-binding sites in antibody complex (valency)
For example, IgG has 2 binding sites, IgM has 10 binding sites
Arrangement and number of antibody binding sites on antigen
Repetitive polymer? Abundant cell surface protein?
As each of these factors increases, so does avidity of antibody or BCR
Central tolerance for immature B cells occurs in bone marrow
B cell tolerance important for preventing antibody responses to self antigen
If recombination is successful
Non-self reactive B cell
Low- avidity self antigens => will eventually die
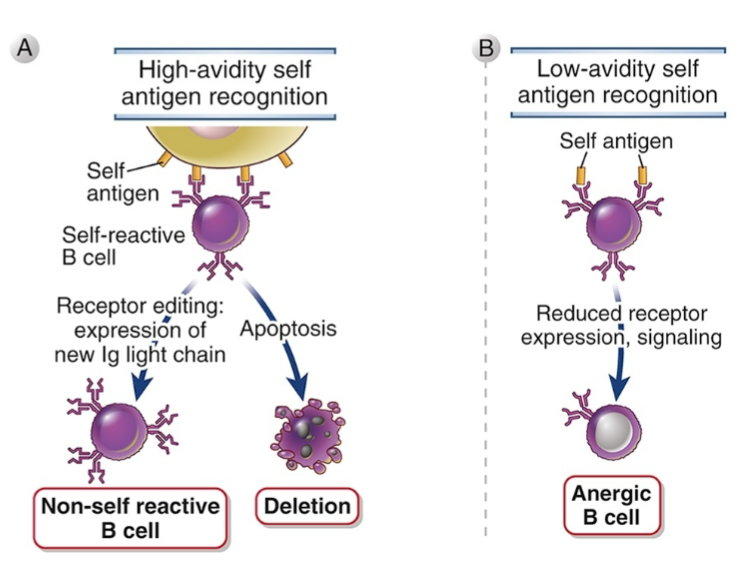
Peripheral tolerance for mature B cells
Similar possible outcomes as for T cells
Self antigens don't trigger innate immune responses
The immune system protects against five types of pathogens
Bacteria
Viruses
Protozoa
Fungi
Helminths (worms)
Immunity to microbes: viruses
Viruses
Are obligatory intracellular microorganisms
Use host nucleic acid & protein synthetic machinery to produce more viral particles
Debilitate and ultimately kill the infected cell (often through lysis)
May indirectly induce tissue damage by triggering host inflammatory response
Kinetics of innate and adaptive responses to virus infection
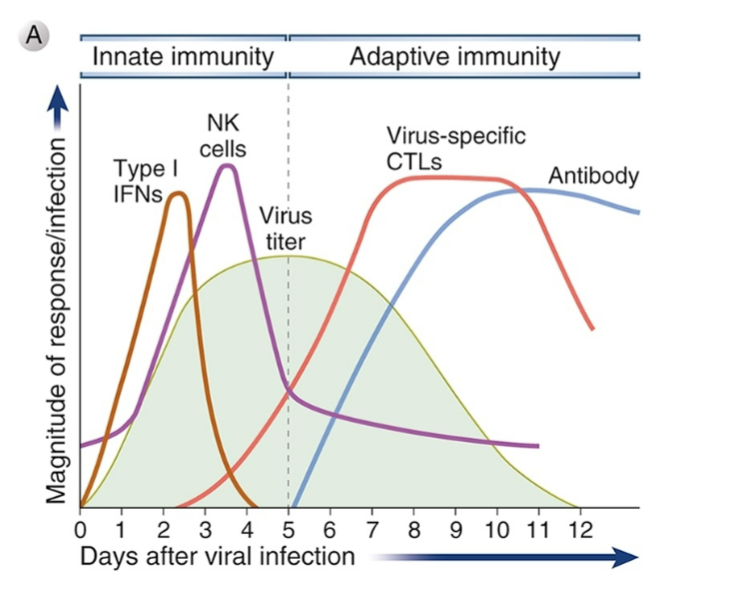
Innate and adaptive protection against viral infection
“Interferon” derived from ability to interfere with viral infection
Expression of type 1 IFNs is induced in many cell types through pattern recognition receptors
IFNs induce expression of enzymes that block viral replication
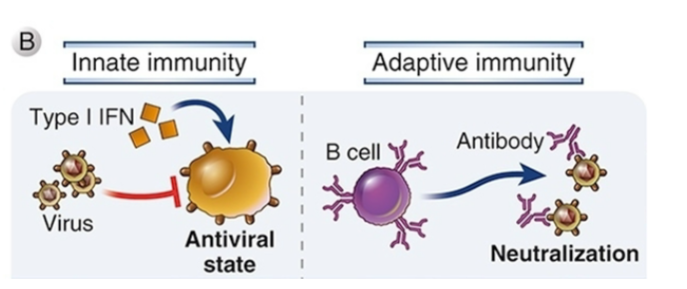
Antibodies:
Block binding of virus to its receptor
May also opsonize particles => phagocytosis
Effective only against extracellular particles
Prevent cell-to-cell spread and re-infection
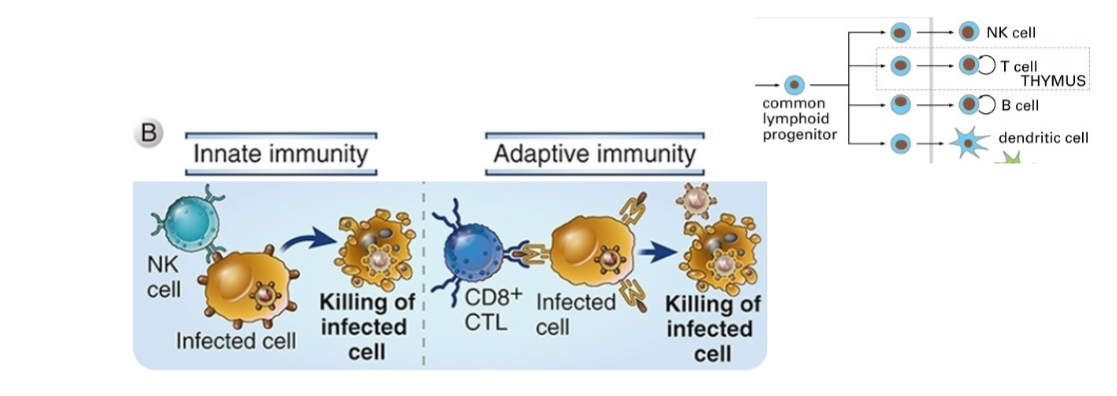
Innate and adaptive eradication of established infection
Via killing of virus-infected cells
NKs
Activated in inflammatory response
Use pattern recognition receptors
CD8+
Massive proliferation during infection
Most are specific for just a few viral peptides
NKs and CTLs use same killing mechanisms
Viral mechanisms for evading host immunity
Alter surface antigens recognized by antibodies or TCRs by one or both of:
Antigenic drift
Antigenic shift
Resulting variation creates strains no longer recognized by immune system
Antigenic drift
Results from point mutations that either
Accumulate throughout the antigen over time (minor changes add up)
Occur in key antigenic site (major change)
Influenza virus is reasonably well understood example
Also common in rhinoviruses and HIV
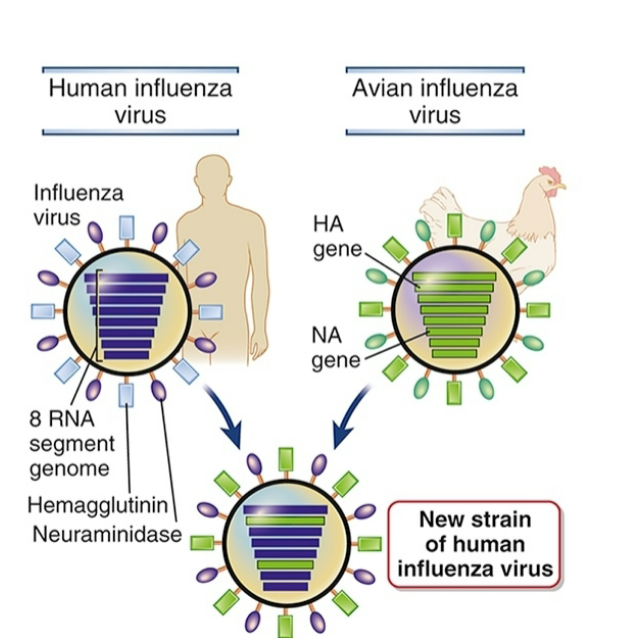
Antigenic shift
Genetic recombination of two viral strains
Less frequent than antigenic drift
But more sudden and significant change
Strains typically found in different hosts
Simultaneous infection by two strands
Allows for reassortment of RNA strands
Can create new, antigenically distinct virus
H1N1 influenza virus responsible for 2009 pandemic
Generated by reassortment of swine, avian and
Human viruses in pigs, then passed back to humans
2. Inhibition of antigen processing and MHC class I presentation
Different viruses inhibit different steps in the pathway
Infected cells not recognized or killed by CD8+ T cells
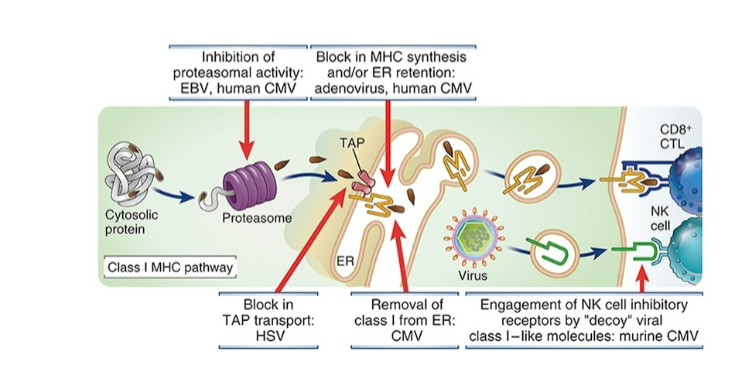
other mechanisms viruses use to evade the immune system
3. Coding for proteins that
Act as ligands for Nk cell inhibitory receptors
Function as decoy signal molecules that compete with cytokines
Resemble immunosuppressive cytokines such as IL-10
Bind to and inhibit pro-inflammatory cytokines
4. Exhaustion of CTLs cells
5. Killing or Inactivation of immune cells (e.g., HIV kills CD4+ T cells)
Immunity against bacteria, fungi, and parasites
immunity to extracellular bacteria
Can survive and reproduce in organ lumens, connective tissues and sometimes blood
Pathogenic extracellular bacteria promote inflammation and produce toxins
Innate immune response: complement, phagocytosis, inflammation
Adaptive immune response: antibody-dependent neutralization & opsonization
May induce cytokine-mediated, inflammatory damage to host
Use various immunoevasion and/or immunoresistance strategies
Immunity to intracellular bacteria
Survive and even reproduce within phagocytes => inaccessible to Abs
Innate immune response: phagocytes and NK cells
Adaptive immune response: CD4+ Th cells activate phagocytes to kill microbes
May result in macrophage-associated damage
Resistant bacteria may escape lysosomes or inactivate microbe-killing mechanisms
Immunity to fungi
Fungal infections may be endemic or opportunistic
Compromised immunity most important factor for significant fungal infection
Fungal infections may be extracellular or intracellular
Innate immune response: neutrophils and macrophages
Adaptive immune response: humoral (extracellular), T cell-mediated (intracellular)
Immunity to parasites
Protozoa and helminths
Infections often chronic
Innate immune response: phagocytosis (protozoa), inflammation (helminths)
Adaptive immune response: varied humoral and cell-mediated
Tissue damage varies with parasite and corresponding immune response
Multiple, effective immunoevasion and immunoresistance strategies
Features of immunity to microbes
The immune response is specialized to type of microbe
Microbial survival and pathogenicity are tied to immunoevasion / immunoresistance
Defects in immunity are important causes of susceptibility to infections
Analysis of immune responses can provide info on status of infection
Impact of vaccines
Vaccination programs have led to complete or nearly complete eradication of many infectious diseases in developed countries
Goal is to obtain “heard immunity”
The principle of vaccination
Administer a harmless form or component of a pathogen
Does not cause disease
Elicits humoral response to protect against live, pathogenic microbe
The best vaccines stimulate
Development of long-lived plasma cells that produce high affinity antibodies
Development of memory cells
A strong innate immune response
Minimal side effects
Vaccination success depends on properties of target microbe
Vaccination most effective if the target microbe does not:
Establish latency
Undergo antigenic variation (i.e., mutation, exhibit different life stages)
Interfere with host immune responses
Infect other animal hosts
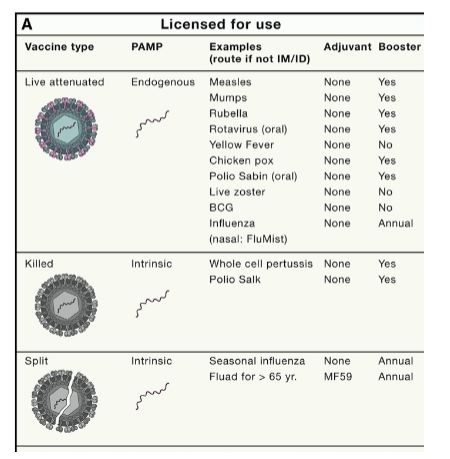
Live attenuated and killed vaccines
Live attenuated
Genetic or chemical modification
Very rarely, the microbe may be reactivated and caused the disease
Killed or Split
Heating, chemicals, or detergents
Subunit & synthetic antigen vaccines
Used purified, synthesized or conjugated antigens
Safer than live attenuated vaccines
Antigen alone typically does not induce a strong response
Requires adjuvant to trigger innate immune response
MRNA/DNA vaccines
RNA or DNA encodes microbial antigens => induces our cells to express protein fragments
Viral vector vaccines
Virus encodes microbial antigens => induces our cells to express protein fragments
Vaccine hesitancy
Loss of knowledge / concern over time
Anecdotal info perceived as significant
Misinformation
Mistrust in government / institutions
B cells use B cell receptors to recognize
solube antigens, microbial surface antigens, & host cell surface antigens
FDCs display protein antigen in the native, folded three-dimensional conformation (t/f)
True
T cell independent antigen responses are primary triggered in response to protein antigens (t/f)
false
All mutations in the variable coding region will imporve the affinity of antibody for its antigen (t/f)
False
the most effective somatic hypermutations are clustered in the complementarity determining regions (CDRs) of the ig heacy chain (t/f)
True
which cell also used perforins and granzymes to kill target cells
CTLs
the surface area of the human digestive tract is estimated to be equivalent to the surface area of
a Tenis court
within an epithelial sheet, adjacent cells are sealed to neighboring cells by
tight junctions
IgA is also a critical component of
Breast milk
self antigens generally activate the innate immune system (t/f)
False
IgA has ______ binding sites
4
Actions of NK cells and CTLs can _____ normal tissues
Damage
The “anti-viral state”______
Inhibits virus replication and assembly
immunoevasion or immunoresistance mechanisms are not exhibited by bacteria? (t/f)
false
which immune component would you expect to be least effective against listeria
antibodies