Science 56
1/8
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
What are quarks?
Elementary particles that are indivisible
U: up
C: charm
T: top
D : down
S: strange
B: bottom
What are leptons?
Leptons are a certain type of particle in an electron
What are the differences between subatomic and elementary particles?
Subatomic particles can be separated into smaller particles, while elementary particles are indivisible.
What are protons and neutrons made out of?
Pro: 2 up 1 down
Neu: 2 down 1 up
How does the position on a periodic table help you predict an elements ion charge?
The family/group tells valence electrons and this determines their charge
What is the correct order?
Valence shell
Neutrons
Electrons
Protons
Atomic number
Element name
Atomic mass
Symbol
What charge? ( Ion )
Name
Symbol
Number
Mass
Pro
Neu
Elect
Valence
What charge
Protons:
Electrons:
Neutrons:
Ion is:
Atomic number
#Protons
Atomic mass - Atomic number
The number of electrons that has changed

What is non-metal/Metal/Metalloid?
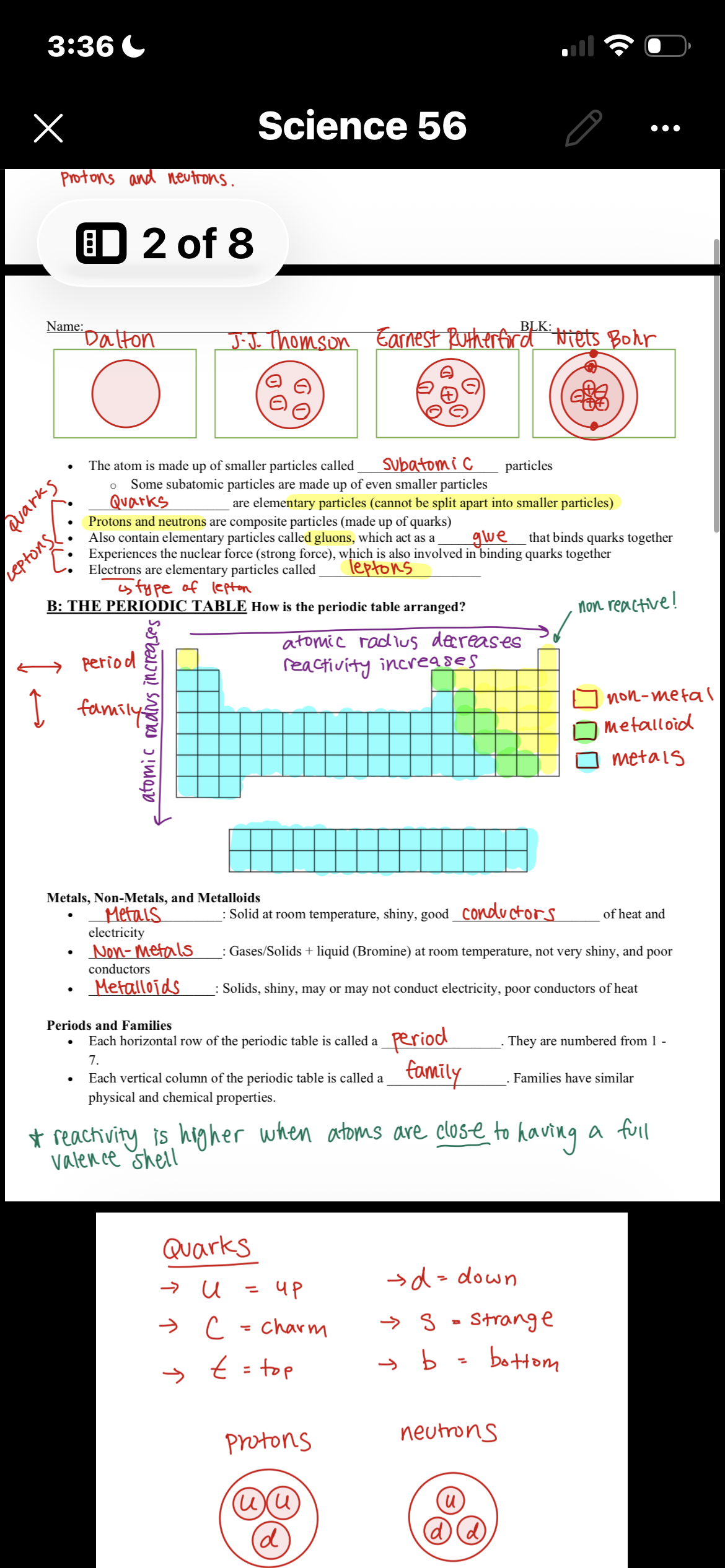
How will the position of the periodic table help you predict its reactivity?
The reactivity depends on how close to full valence shell because atoms will gain or lose electrons to become stable.