mbb 201 lec 15-17
1/134
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
135 Terms
what is the purpose of membrane enclosed organelles
they are important for creating distinct environments with different metabolic functions
functions of cytosol
they contain many metabolic pathways; protein synthesis, cytoskeleton
nucleus main function
contains main genome; DNA/RNA synthesis
endoplasmic reticulum (ER) main function
synthesis of most lipids, synthesis of proteins for distribution to many organelles and plasma membrane
golgi apparatus main function
modificaiton, sorting, packaging of proteins + lipids for secretion or delivery to other organelles
lysosome main function
intracellular degradation
endosomes main function
sorting of endocytosed material
mitochondria main function
atp synthesis by oxidative phosphorylation
chloroplasts in plants main function
atp synthesis and carbon fixation by photosynthesis
main function of peroxisome
oxidation of toxic molecules
evolution of membrane enclosed organelles
1) nuclear membranes and membranes of endomembrane system (er, golgi, peroxisomes, endosome, lysosomes) my have arisen through invagination of plasma membrane
2) interiors of endomembrane system communicate with each other exclusively
protein sorting
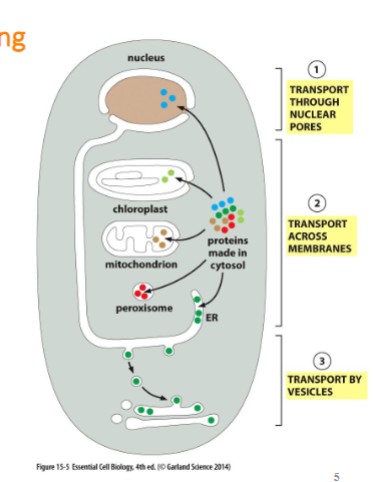
almost all proteins begin synthesis in cytosol and are transported through 3 mechanisms:
transport through nuclear pore (to nucleus)
transport across organelle membranes (proteins translocator)
transport by vesicles (endomembrane system)
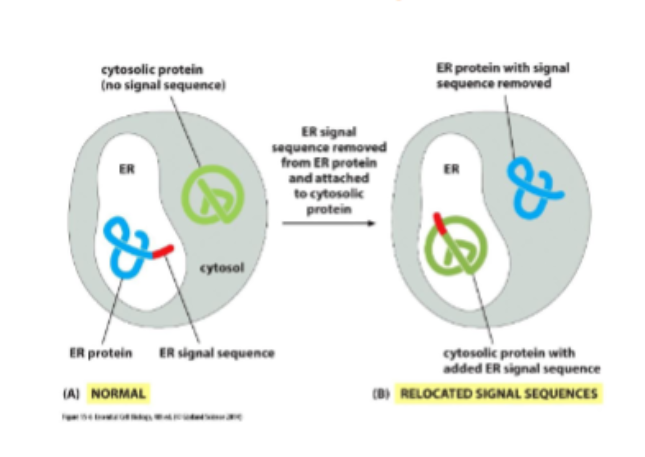
they are directed by the signal sequence created from the amino acid
signal sequencing
usually 15-60AA long and are necessary to direct a protein to a particular destination. they are removed often after the finished protein has been sorted
transport through nuclear pore
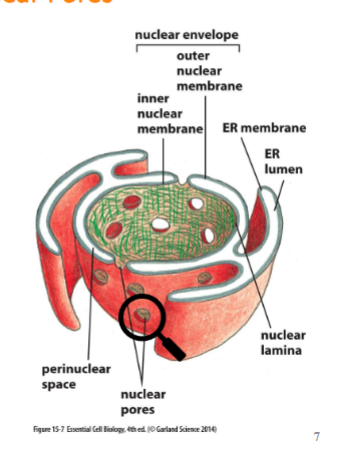
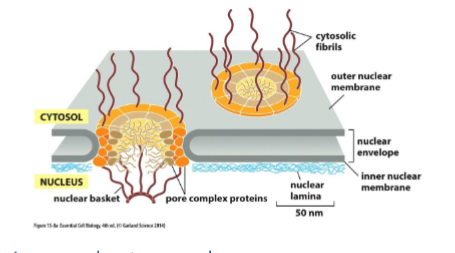
nucleus has nuclear envelope with two membranes (inner and outer, outer is connected w/ ER) and it has pores to allow the passes of molecules in and out
nuclear pore complex
present in both inner and outer membranes. composed of 30 or so proteins which are disordered and unstructured. they create a fibril mesh that fills the center of the channel and prevents the passage of molecules through it. only small, water soluble molecules can pass non-selectively.
protein transport through nuclear pore #2
cytosolic proteins that are for the nucleus must contain a nuclear localization signal NLS in order to pass. it is recognized by nuclear import receptors which direct the protein to the pore by interacting with cytosolic fibrils (it binds and unbinds, walking it through) and through the pore by disrupting the interactions between nuclear fibrils (usually they stick to each other to prevent large molecules but by binding na unbinding, it disrupts the local connection). proteins are fully folded when being transported.
what are cytosolic proteins
Cytosolic proteins are proteins synthesized on free ribosomes and located in the cytosol, the fluid component of the cytoplasm.
gtp hydrolysis drives nuclear transport
happens right after it passes the nuclear pore. once at the nucleus, Ran-GTP knocks the cargo off and binds to the nuclear import receptor instead. the receptor and ran goes back to the cytosol (easily) and once there, an enzyme triggers GTP hydrolysis causing Ran-GTP → Ran-GDP and as a result, the new receptor is ready to be used.
nuclear export
similar to process of exporting proteins and also depends on ran-gtp.
protein sorting in mitochondria and chloroplast
most processing happens in the cytosol. proteins contain a signal sequence at N-terminus to allow for their import. proteins are unfolded as it is transported by translocator →signal sequence is removed after arrival and chaperone proteins help proteins fold again
protein sorting/transport in endoplasmic reticulum
have ER signal sequence and once at ER it will either
soluble proteins: end up in the lumen of the ER and then travel inside the vesicle and get secreted out of cell (golgi → secreted) insulin and hormones
transmembrane protein: end up in the membranes of the ER and stuck there. they become apart of the wall and become the receptors and channels of the surface of the cells.
soluble proteins and ER
soluble proteins made on the ER are released into teh ER lumen. most protein that enter the ER are threaded across the ER membrane before the polypeptide chain is fully synthesized.
ribosomes synthesizing proteins
membrane bound ribosomes: attached to cytosolic side of ER
Free Ribosomes: not attached to any membrane, identical to Membrane bound
Polyribosomes: many ribosomes bound to one mRNA molecule
directing the ribosome to the ER
two protein components help guide ribosome to ER
works by guiding ER signal to Er membrane
signal recognition particle (SRP) presented in cytosol and binds to ER signal sequence and ribosome
SRP receptor: embedded in ER membrane. bind to SRP and passes ribosome to a protein translocator. SRP is released.
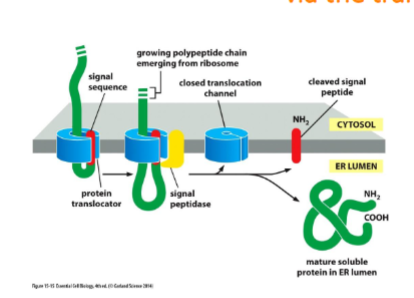
protein synthesis occurs passing the protein through the channel in the protein translocator
Soluble proteins cross the ER membrane and enter the lumen via the translocator channel
ER signal sequence causes the opening of the channel. signal sequence remains bound to the channel as the rest of the protein is threaded through. Once the C-terminus has passed through, the signal sequence is removed by a signal peptidase on luminal side of ER and protein is released into lumen. cleaved signal sequences degrade
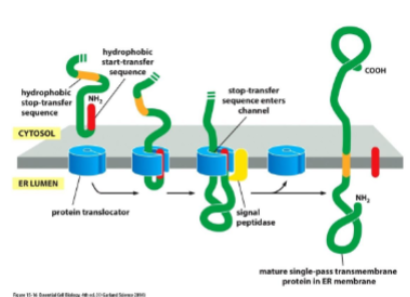
transmembrane proteins in the ER
Some of the polypeptide chain must be translocated completely, while other parts must be fixed in the membrane. for a single-pass transmembrane protein, translocation is initiated by start-transfer sequence. translocation continues until a stop-transfer sequence is reached. stop-transfer forms alpha helix and remains embedded din membrane. orientation of N and C will not change. sometimes the start-transfer seq is internal and does not get removed like n-terminus.
enter into ER lumen or membrane is usually only the first step on a pathway to another destination
the end goal is usually the golgi apparatus where they are modified and sorted for shipment to other places.
vesicular transport functio
the continual budding and fusion of transport vesicles from ER → golgi → other compartments of endomembrane system
the movement of material between organelles in the eukaryotic cell via membrane-enclosed vesicles. allows for transport of both soluble and transmembrane proteins to various parts of the cell including the endomembrane system and plasma membrane. each organelle must maintain its own distinct identity
vesicle budding and coated vesicles
driven by assembly of protein coat.
coated: membeanr enclosed sacs that wear a distinctive layer of proteins on its cytosolic surface to help shape the membrane into a bud and captures molecules for onward transport.
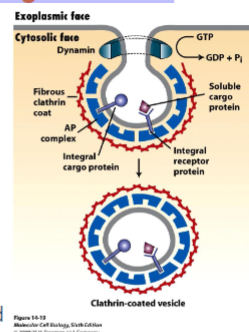
Clathrin-coated vesicles and COP-coated vesicles
Clathrin-coated vesicles: Found budding from the Golgi to endosomes as well as from the plasma membrane on the inward endocytic pathway
COP-coated vesicles: Found in vesicles between the ER and Golgi, as well as from one part of the Golgi to another part of the Golgi
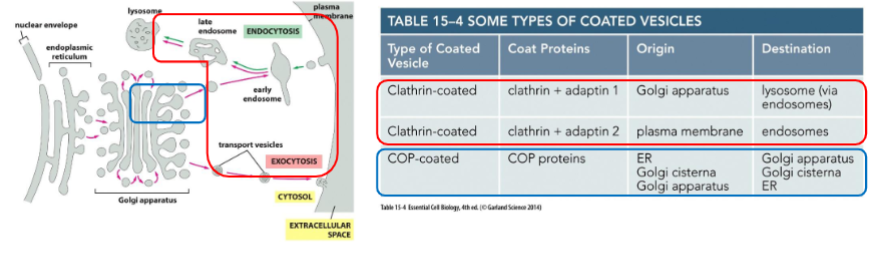
clathrin coated vesicle
vesicle begins as clathrin-coated pit
clathrin is a protein that creates a basker like network on the cytosolic surface of the membrane, helps shape membrane to a vesicle. small gtp-binding protein dynamin functions to pinch off vesicle and assembles a ring around the neck of each invaginated coat pit.
adaptins secure clathrins to vesicle and help select cargo molecules by binding to cargo receptors. appropriate cargo proteins will have transport signals that can be recognized by the cargo receptors.
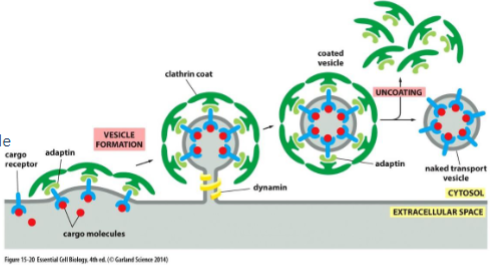
Clathrin-Coated Vesicles transport selected cargo molecules
different adaptin for different cargo ( they recognize different cargo receptors). once budding is complete the coat proteins are removed and the vesicle and fuse w/ target membrane
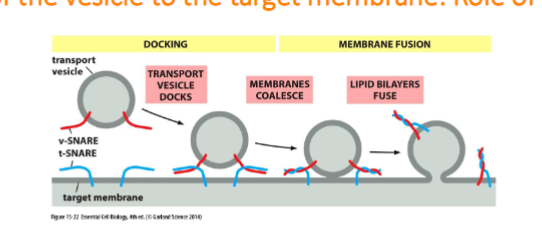
recognition, docking, and fusion of vesicles with its target organelle occurs.
markers must be recognized by complementary receptors on the appropriate target membrane. identification is based on diverse GTPase (rab protein) and transmembrane protein v-snare and t-snare
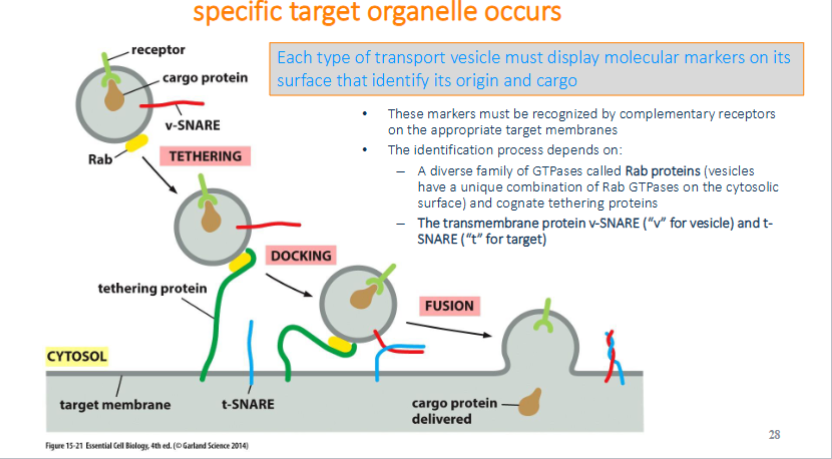
recognition of vesicle: tethering via Rab protein
tethering: rab protein are recognized and bounded by tethering proteins found on target membrane bringing two into close proximity
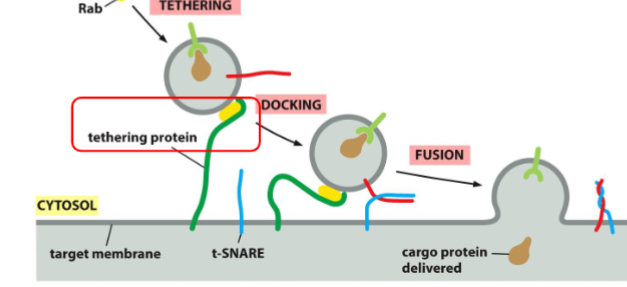
Recognition of Vesicles: Docking via the SNAREs
Docking: The v-SNARE on the vesicle interact with complementary t-SNAREs (“t” for target) which firmly docks the vesicle in place
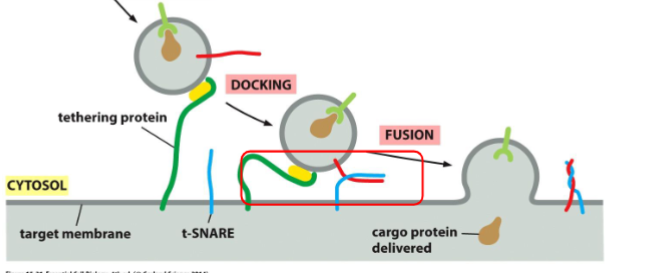
Recognition of Vesicles: two lipid bilayers intermix
fusion: the vescivle fuse w/ the target membrane and cargo protein is delivedto the interior of the organelle or secreted if at the plasma membrane. fusion of membranes is energetically unfavourable. fusion occurs when v-snares and t-snares wrap tightly around each other, whinching the vesicles closer to the membrane such tha two membranes are close enough for their lipids to intermix
modifying protein in the ER
formation of disulfide bonds: covalent bonds that link paris of cysteine side chains and stablize protein structures
glycosylation: covalent attachments of short branched oligosaccharides (glycoproteins). protects from degredation, hold protein in the er, recognition by proteins for packaging or cell-cell interactions, glycosylation is rare on the cytosolic side
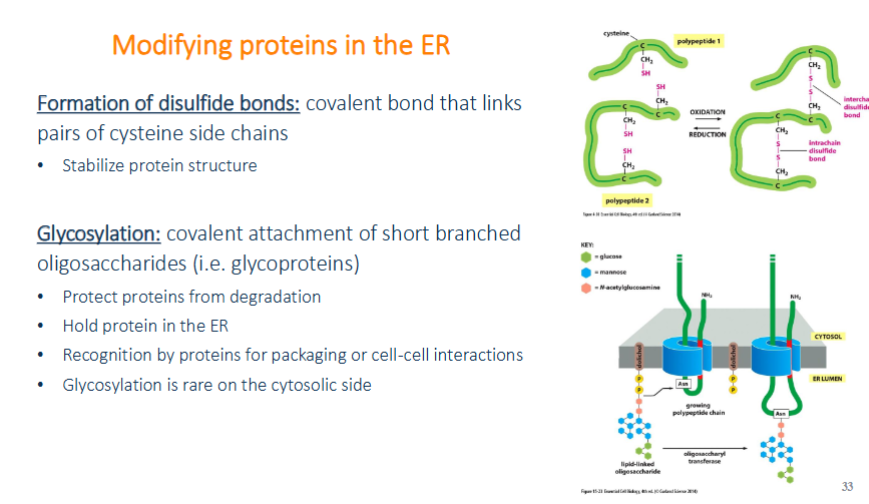
protein glycosylation in ER
oligosaccharides are not added one at a time but all together. 14 sugar oligosaccharide is originally attached to a specialized lipid dolichol in the ER membrane. then transferred onto the amino group of the asparagine side change as the peptide is translocated. because they are attached to an amino, they are said to be N-linked
exit from the ER is controlled
some proteins stay in ER andwill contain approprate retiontion signal sequence while if they escape, they will be recognized by receptors and sent back to ER.
exit from the er is highly selective: must be properly folded
misfolded or multimeric proteins that do not assemble properly are retained in the ER by the binding of chaperone proteins (prevent misfolding from aggregating). N-glycosylation is a sensor for whether a protein is folded properly. if it still fails ,it will be exported to cytosol where it will be degraded.
unfolded protein response
if too many unfolded proteins accumulate in ER, then the unfolded protein response (UPR) is triggered. more chaperones and quality control relaetd proteins are produced and inhibit protein synthesis. size of ER can be expanded to cope w/ the load but if it exceeds then it can be programmed to die.
further protein modification in the golgi
cisternae: flattened membrane enclosed sacks (cis = faces towards ER, trans faces plasma membrane, medial cisterna is in the middle)
enter from cis golgi, protein exit from trans, and modified in golgi.
trans is the main sorting station
secretory proteins are released from the cell by exocytosis
exocytosis: vesicles from golgi fuse w/ plasma membrane
constitutive exocytosis pathway: supplied the plasma membrane w/ lipids and proteins. some proteins are secreted doesn’t need signal sequence other than ER enterance.
regulated exocytosis pathway: only operates in cells specialized for sections. hormones, mucus, digestive enzumes, etc. proteins ae sorted and packed in trans golgi which has conditions that cause proteins to aggregate (acidic pH and high Ca2+). stored in secretory vesicles waiting for a signal. aggregation allows secretory proteins to be at very high concentrations
endocytosis/endocytic pathways
The uptake of material through the invagination of the plasma membrane. Can be broken down into two types based on size:
Phagocytosis: involves the ingestion of large particles. Mainly performed by specialized phagocytic cells
Pinocytosis: ingestion of fluid and molecules via small vesicles. Performed by all cells. Macrophages removes the equivalent to 100% of its plasma membrane every 0.5 hours!
phagocytosis
uptake for food and defense against infections. aft particle is engulfed, they are enclosed in vesicles called phagosomes (fused w. lysosomes, digesting engulfed partical).
pinocytosis and receptor mediated endocytosis
occurs continuously, plasma membrane forms pinocytic vesicle and is mainly carried by clathrin-coated vesicles that pinch off and fuse with endosomes. indiscriminate, they just trap whatever.
receptor mediated endocytosis: selective uptake of macromolecules using specific receptors.
endosomes: sorting station for endocytic pathway
endolytic vesicles deliver material to and are sorted by endosomes. some are near plasma membrane mature into late endosomes by fusing with one another and are found near nucleus. endosomes maintain an acidic environment w/proton pump possible paths:
Recycling: returned to the PM
Degradation: sent to lysosomes
Transcytosis: move to a different domain of the PM
lysosome
principal site of intracellular digestion. Lysosomes are acidic and contain many hydrolytic enzymes involved in the degradation of macromolecules. Lysosomal membrane proteins are highly glycosylated on the luminal side – protects from degradation. Contains a proton pump as well as transporters for macromolecule subunits to enter the cytosol. Lysosome destined proteins receive a mannose 6-phosphate tag in the ER and Golgi
autophagy
process by which a cell digest molecules and organelles that are damaged (eats itself). organelle is enclosed by a double membrane, creating an autophagosome which then fuses with a lysosome for destruction
signal transduction
conversion of a signal or impulse from one form to another. signaling cells create extracellular signal molecules which are received by a target cell.
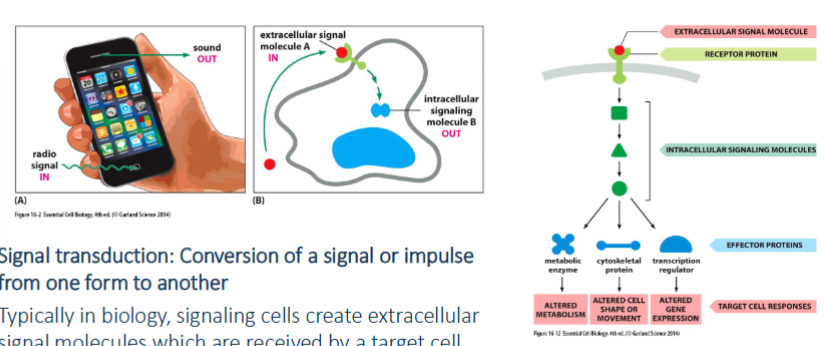
cell communication - general
varies in term of how a “public’ message is made. such as large varieties of forms like proteins, peptides, amino acids, nucleotides, steroids, fatty acids derivatives, and gases
cell communication - endocrine
the most public form of signaling.
endocrine cells produce signal molecules: hormone switch are delivered thru the bloodstream. can signal through the body— very long range. insulin and glucagon are secreted to reduce blood sugar levels
cell communication - paracrine
signal cells produce local mediators which diffuse locally in extracellular fluid. signal is limited and can only be delivered to nearby cells. if the signaling cell response to their own signal = (paracrine) autocrine signaling. cancer cells secrete local mediators that promote their own survival
cell communication - synaptic
signals can be delivered very quickly over long distances but only to specific target cells. transmitted along a neuron in an action potential at nerve terminals. electric → chemical (neurotransmitter) → binds to target cell → converted back to electrical signal
cell communication - contact-dependent
short range, no signaling molecule is secreted. physical contact is made between molecules embedded in the plasma membrane of signal cell and receptors on target cell
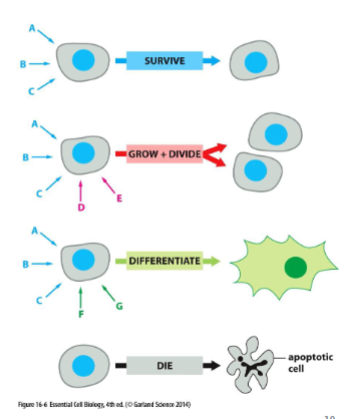
same signal, different response
cells can respond to a mix of signals depending on if it has the appropriate receptor. and even with the same receptor, it might not respond in the same way. the signal alone is not the message but the information is dependent on how the target cell receives and interprets the signal
multiple extracellular signals can dictate how a cell behaves
cells will contain limited set of receptor proteins that will respond to different extracellular signals. a combination of signals can create a response different from each individual signal. cells kill themselves w/o signals
extracellular signal molecule binds either to cell surface receptors or intracellular receptors
can fall into two categories:
molecules that do not cross the plasma membrane and bind to surface receptors (large and hydrophilic)
molecules that cross plasma membrane and enter cytosol and bind to intracellular receptors (small, hydrophobic)
steroid hormones
rely on intracellular receptors. they are hydrophobic molecules that cross the plasma membrane. bind to nuclear receptors that when bound to ligand can enter the nucleus and imitate transcription.
nitric oxide
a gas that can diffused across plasma membrane and bind to proteins like guanylyl cyclase forming cyclic GMP (cGMP). only works because it converts nitrates and nitrites. produced in endothelial cells cause smooth muscle cell to relax = blood vessels dilate. VIAGRA is an example
cell surface receptors
most extracellular signals bind to cell surface receptors which generates intracellular signaling response with intracellular signaling molecules. intracellular molecules activate effector protein → cellular response
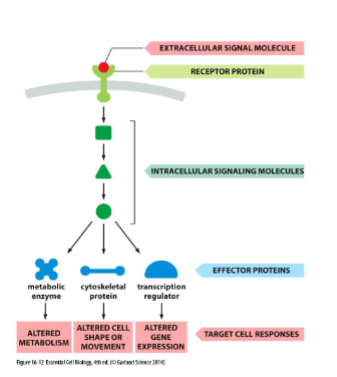
cell signaling pathways function
Relay: signal forwards throughout cell
Amplify: signals making it stronger
Detect signals from 1+ intracellular signaling pathway and integrate them
they can distribute the signal to one or more effector proteins causing a complex response
molecular switches
signaling protein that toggles between active and inactive states in response to a signal. It is important to be able to control both the activation and inactivation
Two classes of molecular switches:
1. Proteins activated or inactivated by phosphorylation
2. GTP-binding proteins
signaling by protein phosphorylation
largest class of molecular switches and involve protein kinases which phosphorylate proteins, and protein phosphatases, which desphosphorylate proteins.
Phosphorylation can either activate or inactivate a protein
• Two main types:
1. Serine/threonine kinases
2. Tyrosine kinases
phosphorylation cascades
The phosphorylation of one molecular switch causes it to phosphorylate another molecular switch allowing the transmission (sends far), amplification (stronger), distribution(branches out) and regulation of signals. essentially its a lot more convenient and affective
GTP-binding proteins
toggles between active and inactive depending on whether they have GTP or GDP bound. GTP-binding proteins posses GTP-hydrolyzing (GTPase) activity
Two main types of GTP-binding proteins:
1. Large, trimeric GTP-binding proteins
(Aka: G-Proteins, more later)
2. Monomeric GTPases
monomeric GTPases
Small GTP-binding proteins that are aided by two sets of regulatory proteins:
1. Guanine nucleotide exchange factors (GEFs): which activate proteins by exchanging GDP for GTP
2. GTPase-activating proteins (GAPs): which inactivate proteins by promoting GTP hydrolysis
Cell-surface receptors
All cell-surface receptors proteins bind to an extracellular signal molecule and transduce its message into one or more intracellular signaling molecule that alter cell’s behavior.
Three major classes:
1. Ion-channel-coupled receptors
2. G-protein-coupled receptors
3. Enzyme-coupled receptors
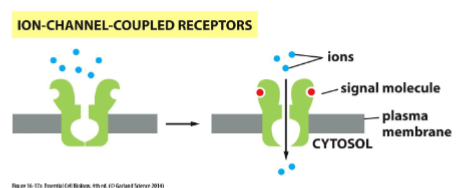
ion channel-coupled receptors
how a postsynaptic cell can receive a chemical signal (a neurotransmitter) and transduce it into an electrical signal by opening ion channel and causing a change in the membrane potential. important in neurons and electrically excitable cells like muscle cells
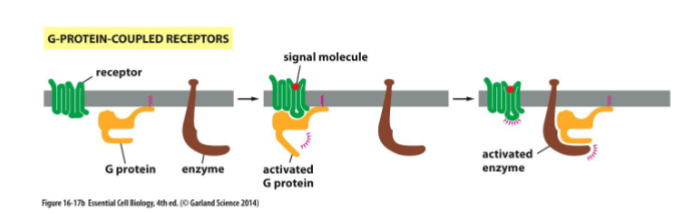
g-protein coupled receptors
G-Protein-coupled receptors activate membrane-bound, trimeric GTP-binding proteins causing the activation (or inactivation) of an enzyme or an ion channel in the plasma membrane
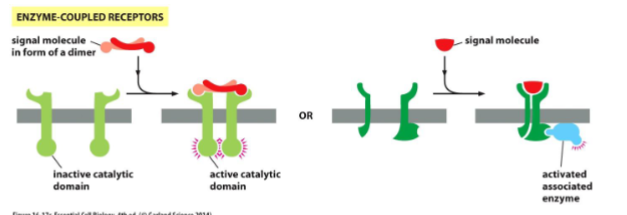
enzyme coupled receptors
The receptor itself can act as an enzyme or associate with enzymes in the cell.
g-protein coupled receptors (GPRCs)
are the largest family of cell surface receptors and mediate the response to an enormous diversity of extracellular signaling molecules including hormones, local mediators, and neurotransmitters. 1/3 of drugs used today work via GPCRs and are composed of a single polypeptide chain that spans the 3 lipid bilayers 7 times
Activation of a GPCR
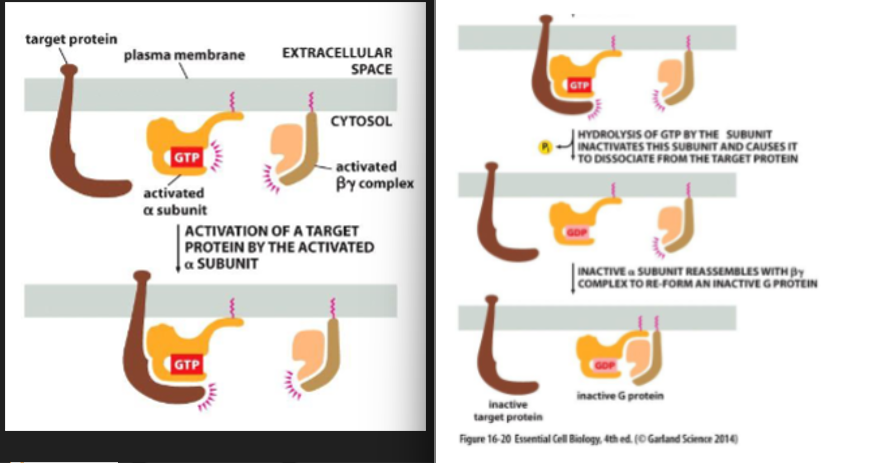
binding of an extracellular signal molecule to a GPCR causes it to change conformation. this in turn activates a trimeric G-proteins which results in the transmission of a signal. there are several G-proteins, each is a specific set of receptors and target enzymes/ion channels. upon stimulation by a signal molecules, the GPCR changes conformation that facilitates binding of the trimeric G protein complex. this leads to a decreased affinity of G(alpha) for GDP and increased affinity for GTP. GDP dissociates and is exchanged for GTP and are switched “on”. G-proteins interact w/ target enzymes or ion channels.
trimeric G-proteins
made up of alpha, beta, and gamma subunit. when unstimulated, alpha is bound to a GDP
switching off
the amount of time that the G-proteins subunits are “switched on” dictates the length of response. the subunit will remain on when GTP is bound to a alpha subunit. the alpha subunit contains GTPase activity which can hydrolyze the GTP to form GDP. alpha subunit reassembles w/ the betagamma complex and returns to it’s original inactive state.
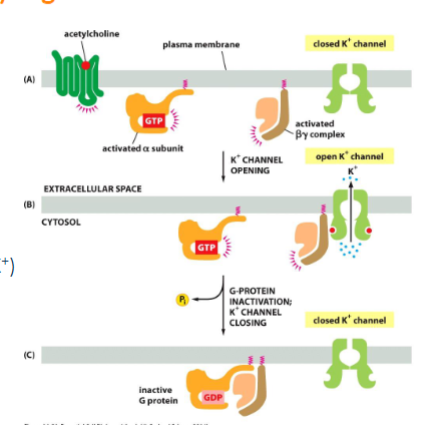
some g-proteins directly regulate ion channels
ie regulation of heart rate - slowing down. acetylcholine binding to GPCRs of heart pacemakers cells activate the the G-protein Gi. the betagamma subunit binds to K+ ion channel causing it to open (slowing heart rate by increasing membrane permeability to K+). Channel closes when GTP is cleaved and the subunit re-associates w/ one another
many g-proteins activate membrane bound enzymes that produce smaller messenger molecules
2 most common enzyme targets:
Adenylyl cyclase - produces cyclic AMP (cAMP)
Phospholipase C - produces inositol triphosphate (IP3) and diacylglycerol (DAG)
are activated by different G-proteins, these three are called SECOND MESSENGERS
cAMP signaling pathway can activate enzymes and turn on genes
denylyl cyclase synthesizes cAMP:
• Generates cAMP from ATP, releasing PPi
• The α subunit of the G-protein Gs is responsible for
the activation of adenylyl cyclase
– “S” is for “stimulate”
cAMP phosphodiesterase degrades cAMP:
• Converts cAMP to AMP using water
cAMP glycogen breakdown
cAMP activate cAMP-dependent protein kinase (PKA). Normally inactivated by binding to a regulatory protein. binding of cAMP ot PKA releases regulatory protein. PKA can then phosphorylate other proteins like glycogen phosphorylase in skeletal muscle.
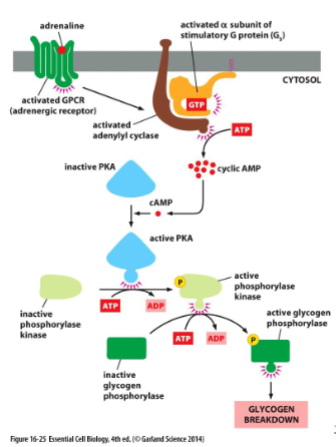
cAMP slow responses
can also activate gene expression - a relatively slow process. PKA phosphorylate transcriptional regulators which can initiate transcription.
inositol phospholipid pathway
Some GPCRs activate the membrane bound enzyme phospholipase C (instead of adenylyl cyclase)
• Phospholipase cleaves an inositol phospholipid
(found in the plasma membrane) into:
inositol 1, 4, 5-triphosphate (IP3): Released into the cytosol
Diacyglycerol (DAG): Remains embedded in the membrane
Both products are important in signaling
Phospholipase C
IP3 binds to and opens Ca2+ channels embedded in ER membrane which free Ca2+ is released into the cytosol which can act on other proteins. DAG recruits a cytosolic protein to the plasma membrane, protein kinase C (PKC)
Activation of PKC requires the binding of Ca2+
PKC phosphorylates several intracellular proteins
Calmodulin
Ca2+ binds to specific proteins in order to exert their effects. The most common of which is calmodulin. Calmodulin binds to four Ca2+ ions, inducing a conformational change allowing it to interact with other proteins like Ca2+/calmodulin- dependent protein kinases (CaM-Kinases)
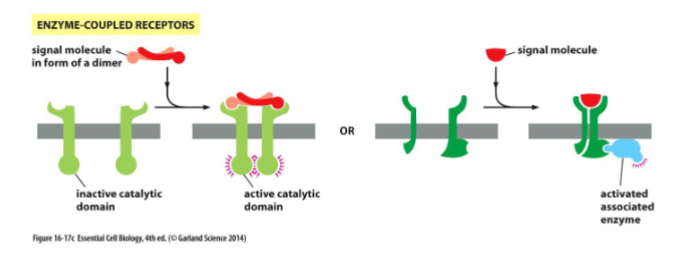
Enzyme-Coupled Receptors
Transmembrane proteins that either:
1. act as enzymes themselves or
2. associate with another protein that acts as an enzyme
• Responses can be fast (eg. reconfigurations of the cytoskeleton) slow (eg. result in
changes in gene expression)
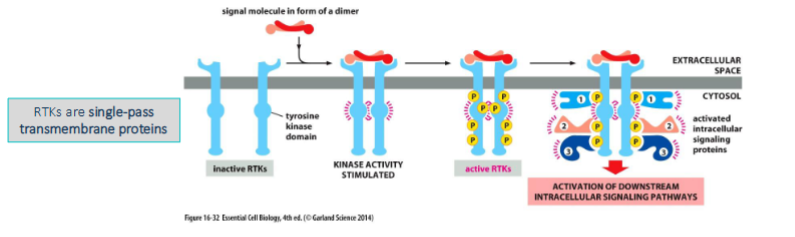
Receptor Tyrosine Kinases:
The receptors often form dimers upon binding of an extracellular signaling molecule
• Each receptor protein possesses a tyrosine kinase domain which allows each receptor to phosophorylate tyrosines on the other
– Tyrosine residues on the cytoplasmic tail are phosphorylated which serve as docking sites for other proteins
The phosphorylated tyrosines serve as a dock for many other proteins.
• Some known as adaptor proteins which act as a scaffold so that other proteins can bind while others propagate the signal
– Each contains an interaction domain which recognizes phosphorylated tyrosines on the tail
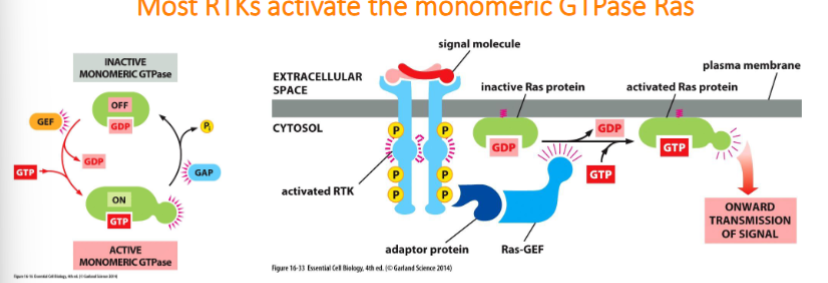
Most RTKs activate the monomeric GTPase Ras
Ras is a small GTP-binding protein that is bound to the cytoplasmic face of the plasma membrane
• Ras-GEF encourages Ras to exchange GDP for GTP, which activates Ras
• Ras-GAP promotes the hydrolysis of GTP to GDP which inactivates Ras 42
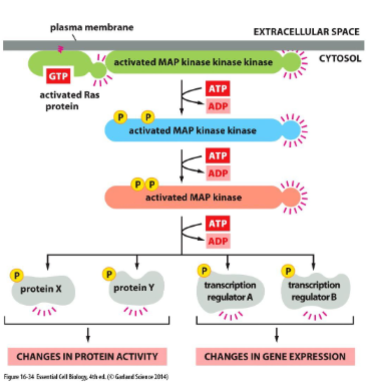
Ras activates a phosphorylation cascard: MAP-kinase signaling
Ras activates a series of serine/threonine protein kinases
• Example: Mitogen-activated protein kinase (MAP kinase) pathway
• Each member of the cascade is a molecular switch that when activated, phosphorylates the next member of the cascade.
• Mitogen: extracellular signaling molecule that stimulates cell proliferation.
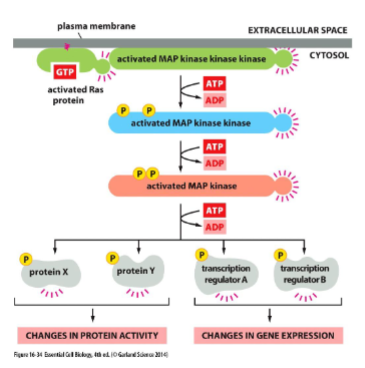
Ras and cancer
MAPK pathway is involved in cell proliferation, survival and differentiation
• 30% of human cancers involve a mutation in Ras which inactivates the GTPase activity and so keeps Ras in its GTP-bound “ON” state (the others have mutations in genes that encode proteins that function in the same signaling pathway as Ras)
• This prevents the signal from being turned off and results in uncontrolled cell proliferation
• Oncogene: A gene that when activated can potentially make a cell cancerous (e.g. Ras)
Some RTKs create lipid docking sites
RTKs also work through the phosphoinositide 3-kinase (PI 3-kinase) signaling pathway involved in cell growth and survival
• PI 3-kinase phosphorylates inositol phospholipids in the plasma membrane which then serve as docking sites for other proteins
– Same inositol phospholipid as the Phospholipase C substrate
– Converts to Phosphatidylinositol triphosphate (PIP3)
• These other proteins are recruited to the PM from
the cytosol where they can activate one another
• Example: Akt, promotes growth and survival
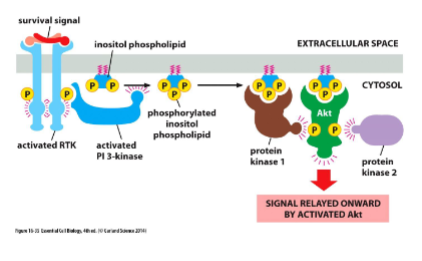
Activated Akt promotes cell survival and cell growth
PI 3-kinase-Akt pathway promotes cell survival:
• Akt (Protein kinase B) is a cytosolic protein that binds to phosphorylated inositol
phospholipids
• Phosphorylated Akt phosphorylates proteins and, in this way, prevents cell death
PI 3-kinase-Akt pathway promotes cell growth:
• Akt can also activate the serine/threonine kinase called Tor
• Tor enhances protein synthesis and inhibits protein
degradation
• Overactive Tor may play a role in cancer
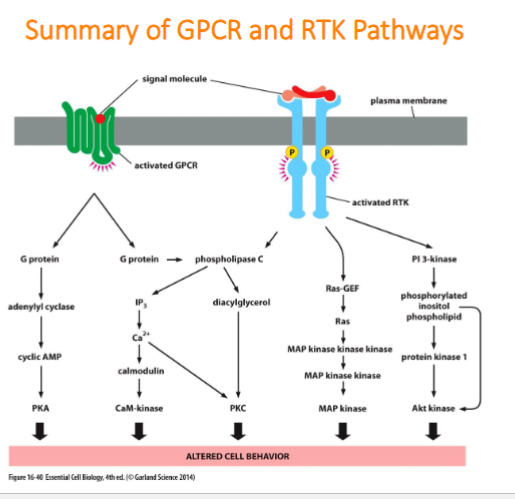
summary of GPCR and RTK pathways
the cytoskeleton
Network of protein filaments that gives the cell shape and capacity for directed movement. not only the skeleton but the muscles too. helps dictate location of organelles and transport between them. they are dynamic.
intermediate filaments (IF) function and location
function: great tensile strength which enabled cells to withstand mechanical stress. strong and durable found in most but not all eukaryotes. provide internal reinforcement
location: surrounding nucleus. often anchored to the plasma membrane of cell-cell junction. within nucleus forming nuclear lamina. prominent in cytoplasm of cells that are subject to mechanical stress (axons, muscle cells, epithelial cells). they distribute the effects of a locally applied force to prevent membranes from tearing.
IF structure features
several types but they all have:
1. α-helical central rod domain, they all contain similar AAs
2 monomers wrap around each other to form a coiled-
2 coiled-coil dimers associate to form a staggered tetramer
Each dimer runs in opposite directions (“head-to-head”)coil dimer
IF structure 3D
8 tetramers associate with each other side by side
The 8 tetramers add to a growing, overlapping filament
Each end is the same! The N-termini of the dimers are on the ends
Noncovalent binding holds the filaments together
4 classes of intermediate filaments
keratin, vimentin, and neurofilaments found in cytoplasm - form ropelike structure
nuclear lamins found in nucleus - forms 2D mesh
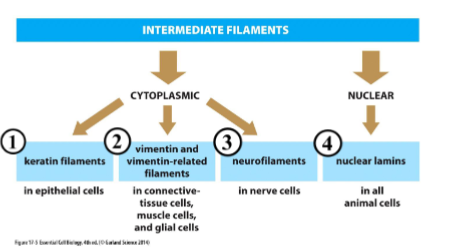
keratin filaments
found in every epithelial cell, specialized for hair, feathers, and claws. anchored to cell-cell junctions (desmosomes) and spans the interior of epithelial cell from one side to another.
ie Epidermolysis bullosa simplex is a disease where the formation of keratin is impaired. The skin is vulnerable to mechanical injury causing skin to blister
nuclear lamina
ifs that forms the nuclear lamina. strengthen the inside of the nuclear envelope and provides attachments sites for DNA binding proteins. Must disassemble and reassemble during mitosis (controled by phosphorylation and dephosphorylation of lamins.
Progeria: Disease with a defect in a particular nuclear lamin. Causes premature aging
microtubules
organization of eukaryotic cells. long and relatively stiff hollow tubes. important for transporting and positioning of membrane-enclosed organelles. forms mitotic spindle during mitosis. allow cell motility by forming cilia and flagella
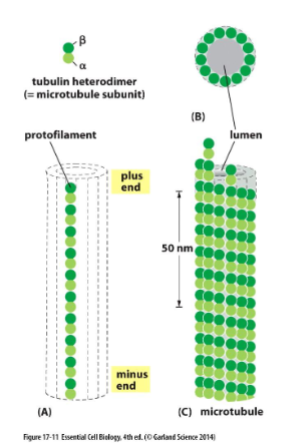
microtuble structure
microtubules are made from subunits called tubulin. each subunit is comprised of a globular α- and β-tubulin dimer bound by noncovalent interactions. protofilament: linear chain of tubulin dimers
13 protofilaments form the hollow tube of microtubule