Final Exam Study Guide Questions - Part II
1/57
Earn XP
Description and Tags
ONCOL 356 - Treatment Planning II. University of Alberta
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
What is the general radiotherapy (RT) planning workflow?
Patient imaging (CT/MRI) → contouring of targets and OARs → target definition (GTV, CTV, PTV) → prescription (dose and fractionation) → treatment planning (beam configuration and optimization) to achieve desired dose distribution while sparing OARs.
What defines a radiotherapy treatment plan?
A combination of machine parameters (collimator angles, MLC positions, beam energies, gantry angles, and monitor units) that together create the dose distribution.
Why is the planned dose not always identical to the delivered dose?
Due to factors like MLC leaf speed lag, patient setup variation, and limitations of dose calculation algorithms.
Why are verification tools needed in radiotherapy planning?
To assess how accurately the planned dose matches the actual delivered dose.
What is one-dimensional dose verification in radiotherapy?
Checking the dose at a single point (reference point) rather than across the full 3D distribution.
What is the reference point in dose verification?
A representative point where the dose is calculated and verified.
How is one-dimensional verification typically performed?
Using an independent manual MU calculation
What is a limitation of one-dimensional dose verification?
It does not assess dose distribution throughout the entire treatment volume.
How is dose at a reference point (Dref) obtained in treatment planning systems like Eclipse Treatment Planning System or Monaco Treatment Planning System?
Using advanced algorithms (AAA, Monte Carlo, Acuros) that perform detailed 3D dose calculations with heterogeneity correction.
What is the purpose of independent dose calculations in radiotherapy QA?
To verify the accuracy of the TPS-calculated dose at the reference point.
What tools can be used for independent dose calculation?
Vendor or in-house tools such as Excel-based calculations or DoseCHECK.
How do independent calculations differ from TPS calculations?
They use simplified 1D calculations and are independent of complex TPS algorithms like AAA or Monte Carlo.
Why is it important that independent calculations use a different method than the TPS?
To provide true validation and ensure the TPS calculation is accurate.
How is an in-house reference point dose calculation performed?
By taking a calibration dose and applying modifying factors (e.g., HSF, PSG, TPR, inverse square) to determine required MUs.
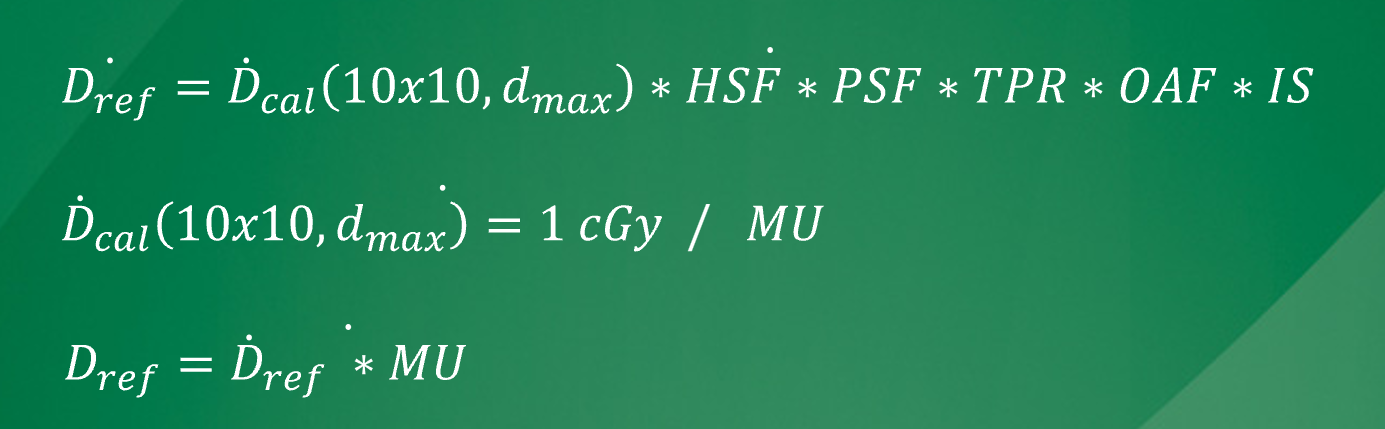
What is the purpose of modifying factors in MU calculations?
To account for clinical conditions affecting dose delivery.
How does plan complexity affect dose verification?
More complex plans require verification of additional factors.
What additional factors must be verified in complex plans?
Wedge factors, modulation factors (e.g., VMAT), and cutout factors (electrons).
How does spreadsheet (1D) calculation compare to TPS 3D calculation (e.g., Eclipse Treatment Planning System)?
Spreadsheet calculations are simplified, while TPS uses more sophisticated 3D algorithms
What level of discrepancy between 1D and 3D dose calculations is concerning?
Around 5–6% difference.
What should be done if a significant discrepancy is found between calculations?
Investigate the cause of the difference to ensure accuracy.
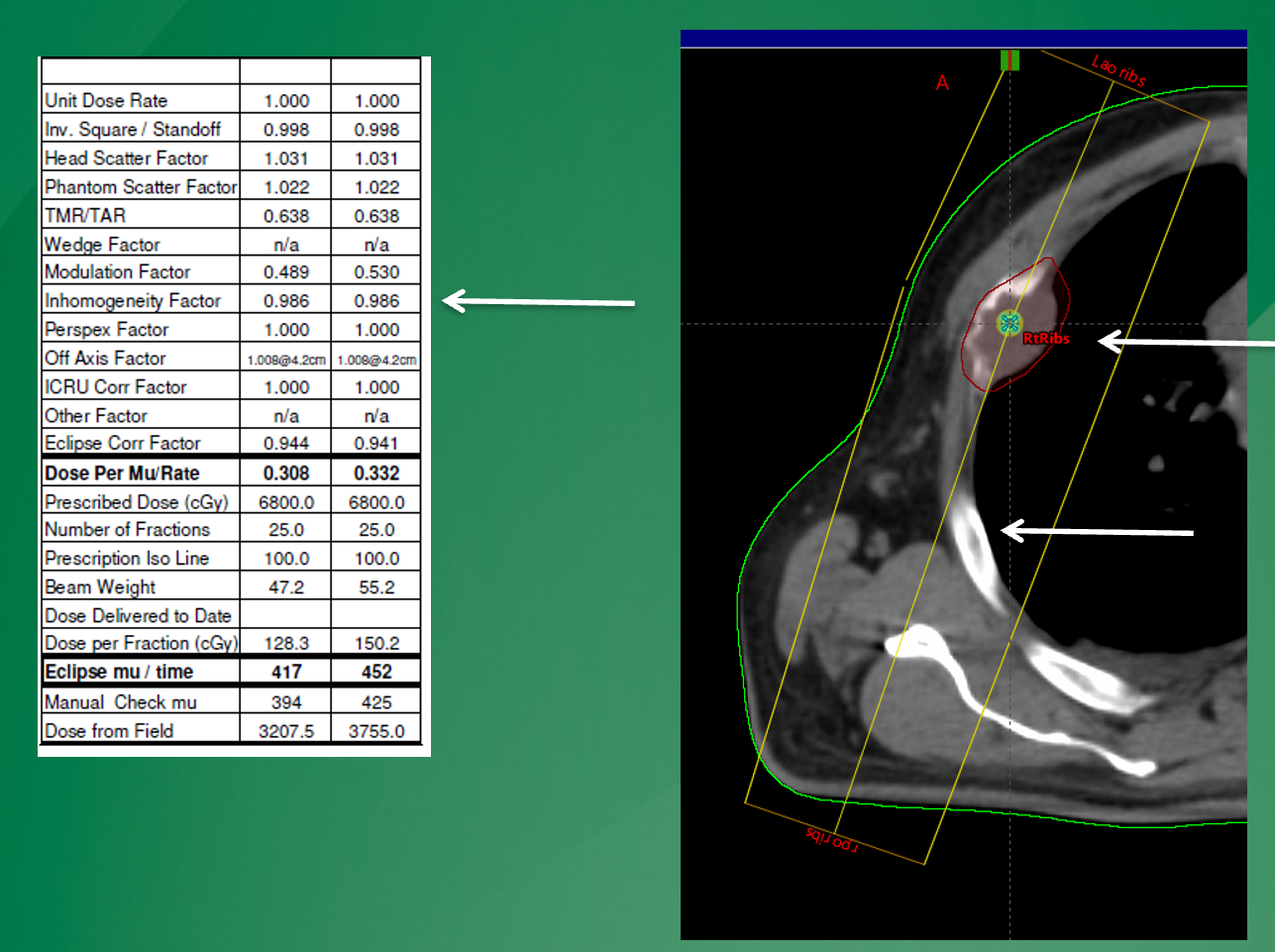
why may the 1D calculation (left) show a different MU value than the Eclipse calculation on the right?
simple 1D calculations will only account for heterogeneities lying along the central axis. in this case, the centra axis does not go through rib and through lung so there is not as significicant of a homogeneity calc than that seen in eclipse, that will account for the entire beam
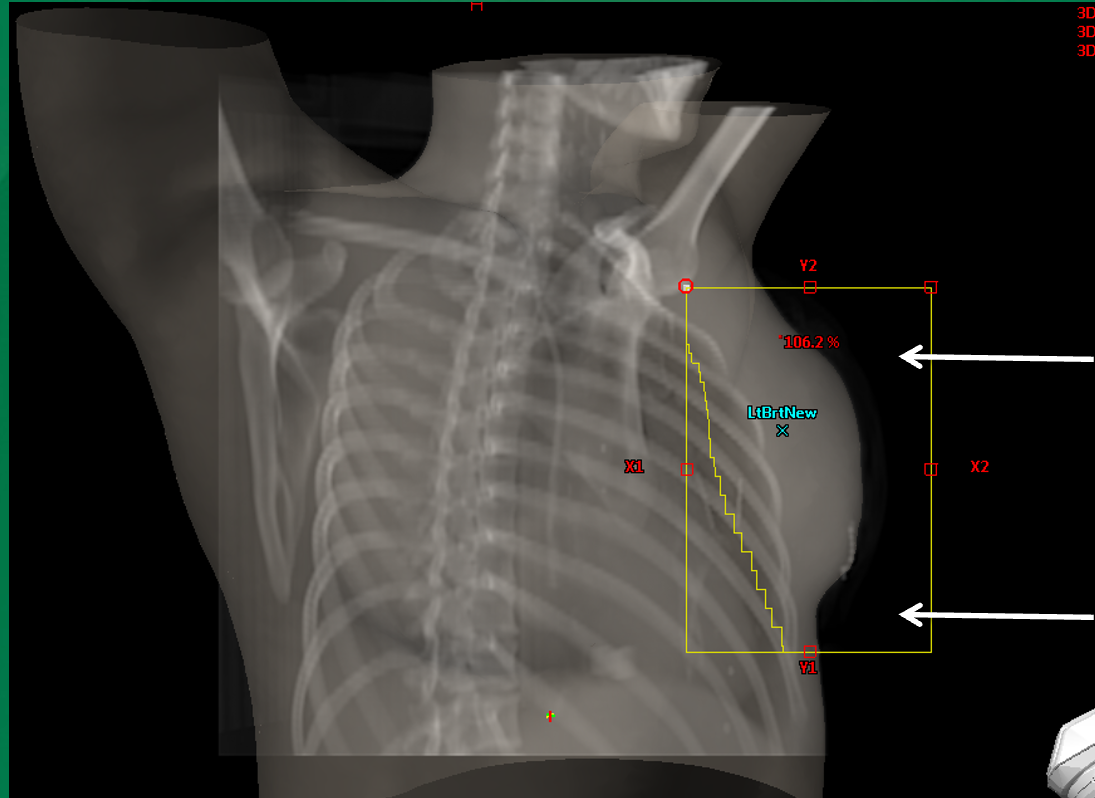
How does FLASH irradiation in air affect central axis dose calculations?
It alters the central axis conditions because part of the beam travels through air instead of tissue. 1D calculations assume tissue along the central axis, but in reality there may be air.
How does TPS (e.g., Eclipse Treatment Planning System) handle FLASH scenarios better than 1D calculations?
It accounts for heterogeneities like air vs tissue in its 3D dose calculation. 1D calculations do not accurately account for changes in scatter and attenuation
What is the modulation factor in radiotherapy?
Dose (modulated field) ÷ dose (open field).
represents the effect of beam modulation (e.g., MLC movement) on delivered dose
What is EPID portal dose verification used for?
QA in VMAT/IMRT to verify complex fluence patterns.
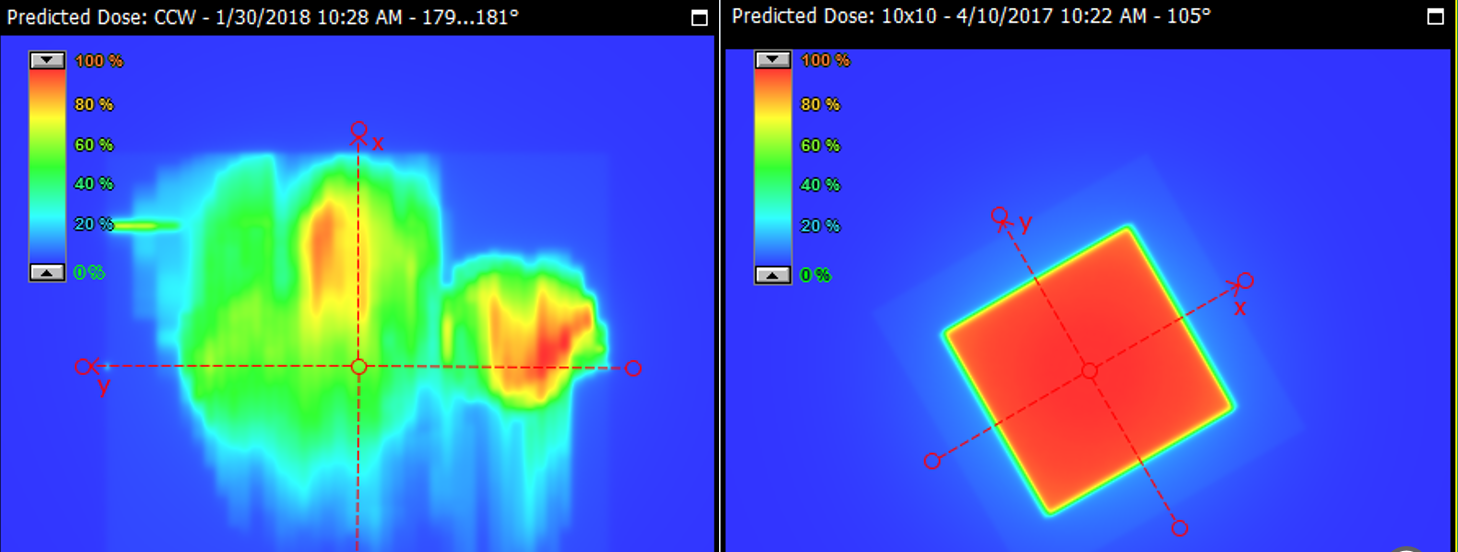
Why is EPID needed for VMAT/IMRT QA?
Because fields are highly modulated with hot and cold spots, not uniform.
Why are simple 1D hand calculations insufficient for VMAT/IMRT?
Because these techniques involve complex, non-uniform fluence distributions.
What is 2D dose verification in radiotherapy?
Projection of planned fluence onto an EPID to compare predicted vs measured dose.
What is the main purpose of 2D verification (EPID)?
To verify complex, modulated fluence fields in VMAT/IMRT.
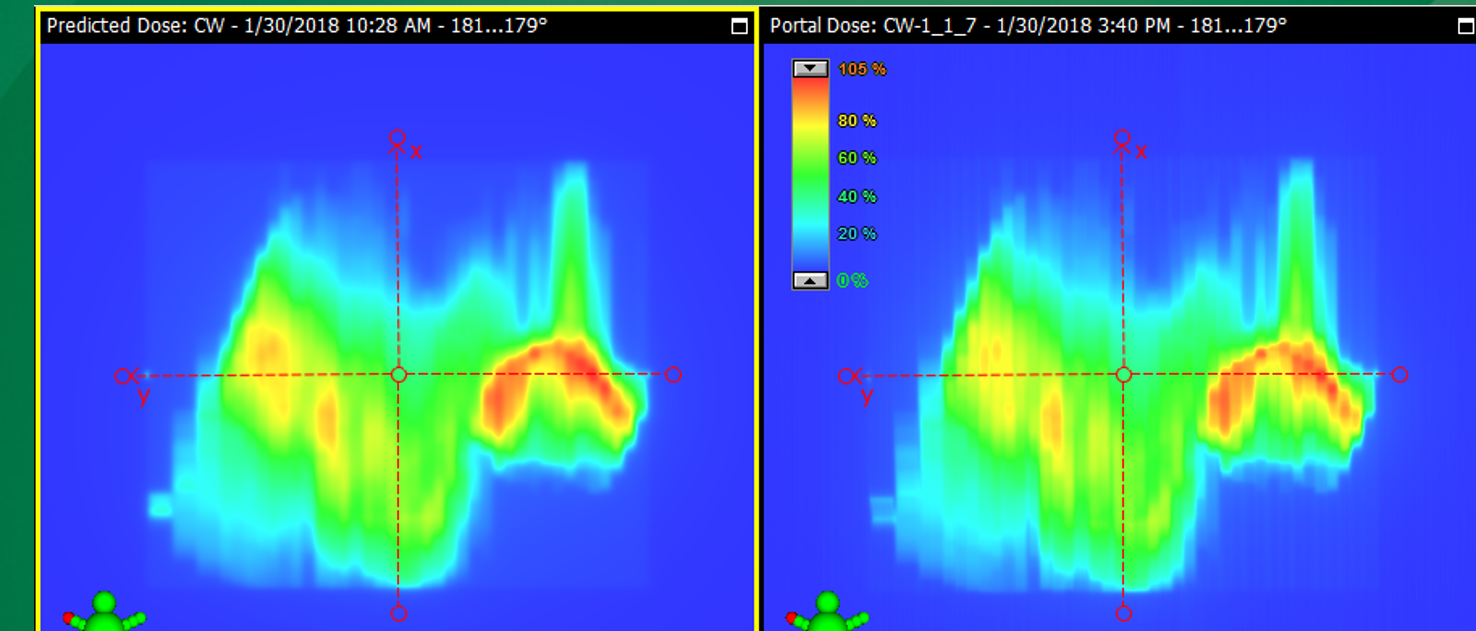
How is EPID QA performed?
Dose is measured without the patient and compared to the predicted portal dose.
What machine parameters can EPID verification assess?
MLC motion, gantry movement, and dose rate.
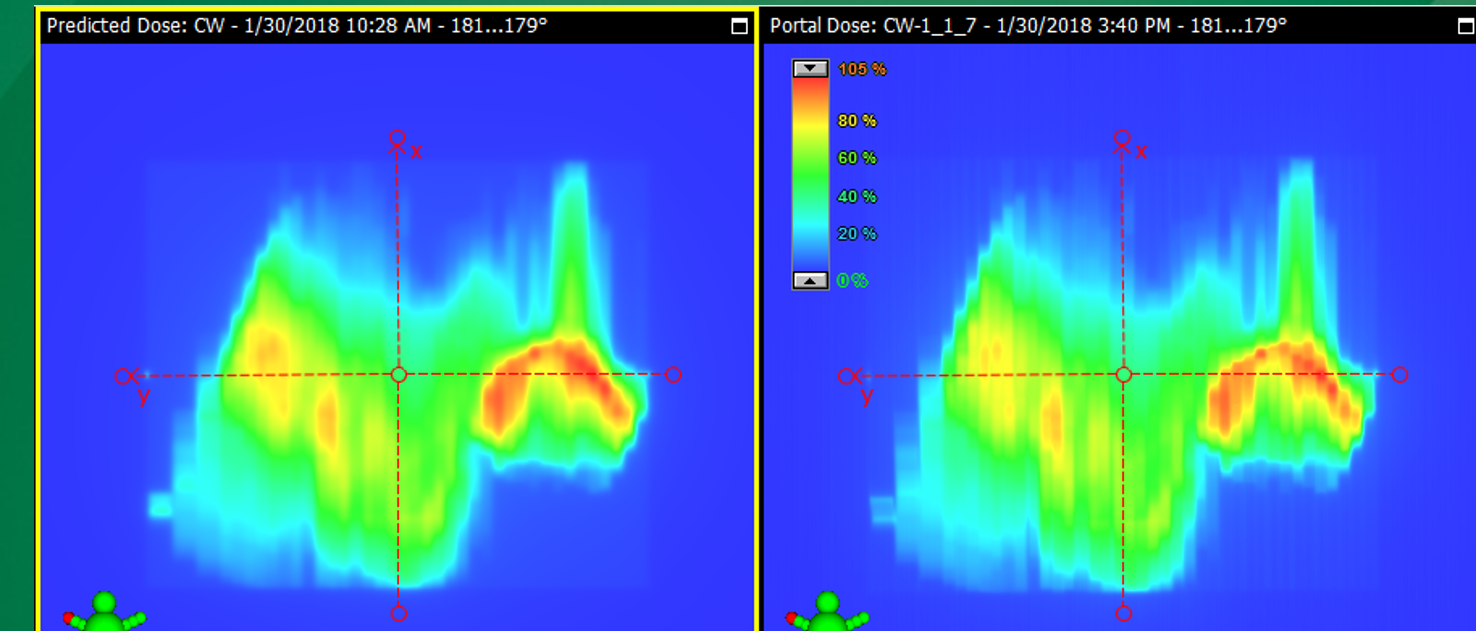
Why is it called “2D” verification?
It verifies the fluence field, not the actual 3D dose inside the patient.
Why is visual comparison alone insufficient in EPID QA?
Differences may not be obvious, so quantitative analysis is required.
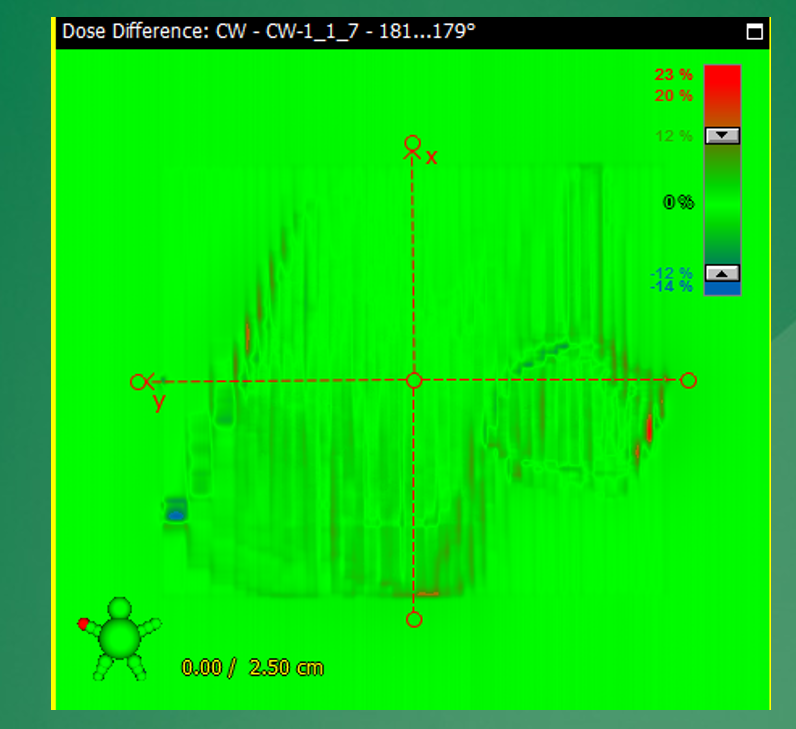
What is a dose difference map in 2D verification?
A subtraction of predicted and measured dose images to identify discrepancies
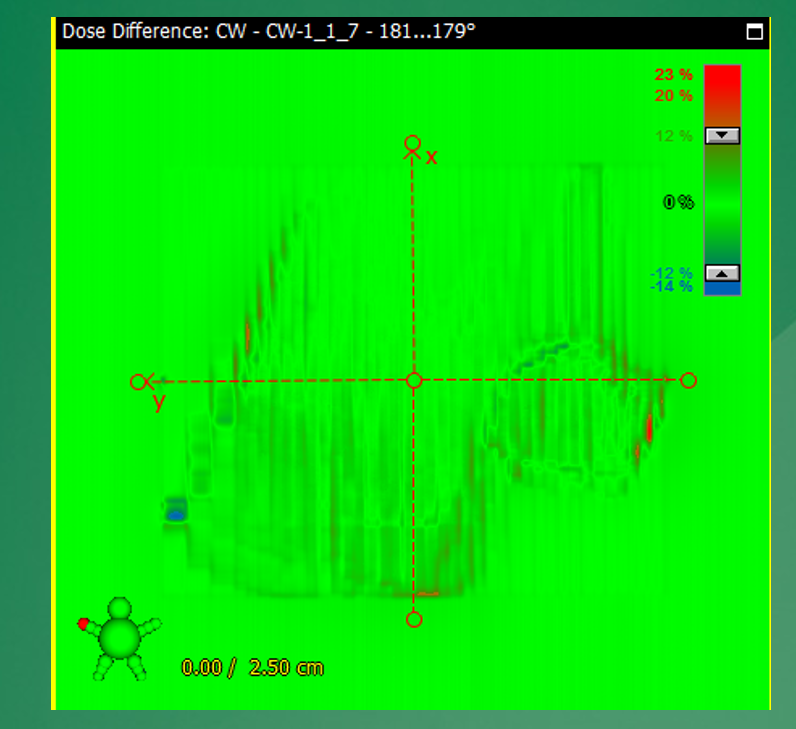
What does an ideal dose difference map look like?
Values close to zero across the entire field.
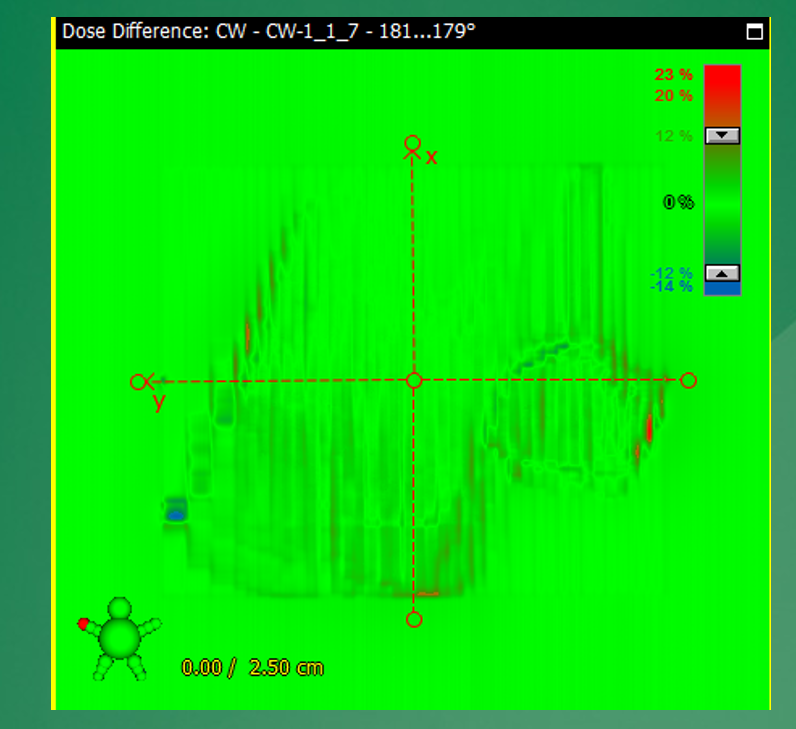
What does it indicate if measured dose is “colder” than predicted?
The delivered dose is lower than expected in those regions.
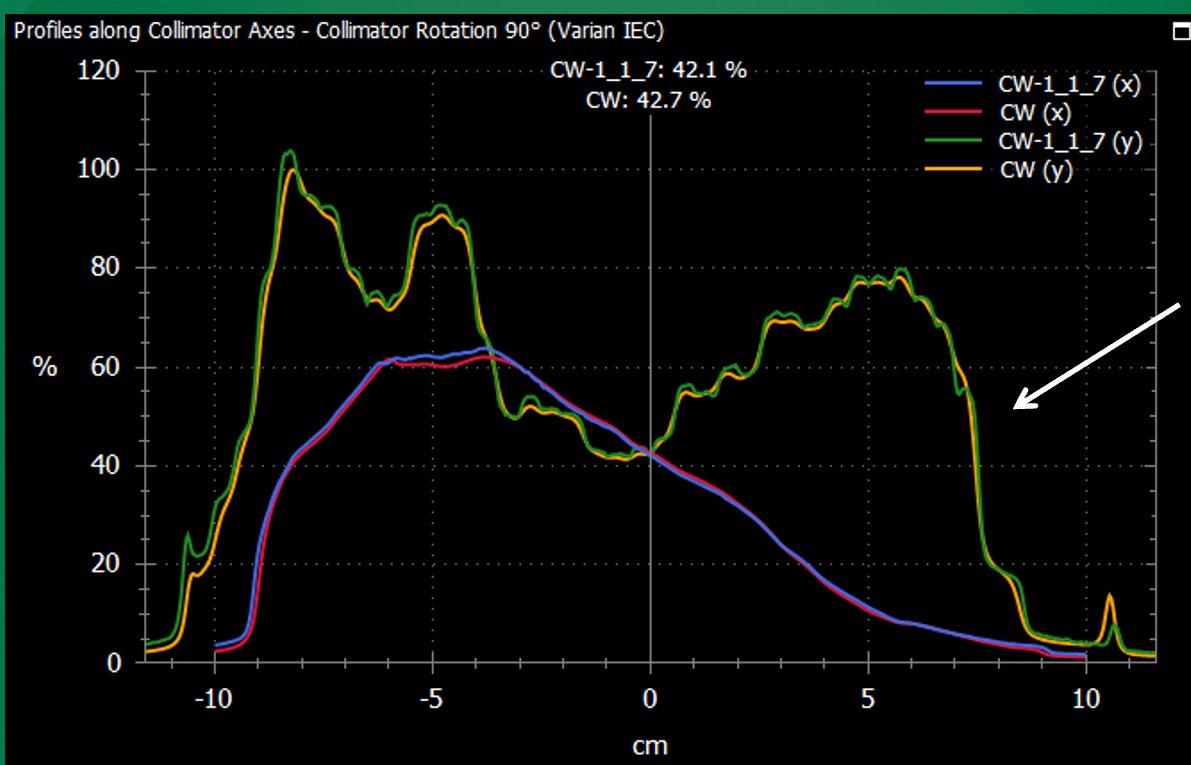
What are dose profiles used for in radiotherapy QA?
To compare measured vs predicted dose along a line through the field.
What do dose profiles help assess?
Agreement in dose distribution, especially across gradients and field edges.
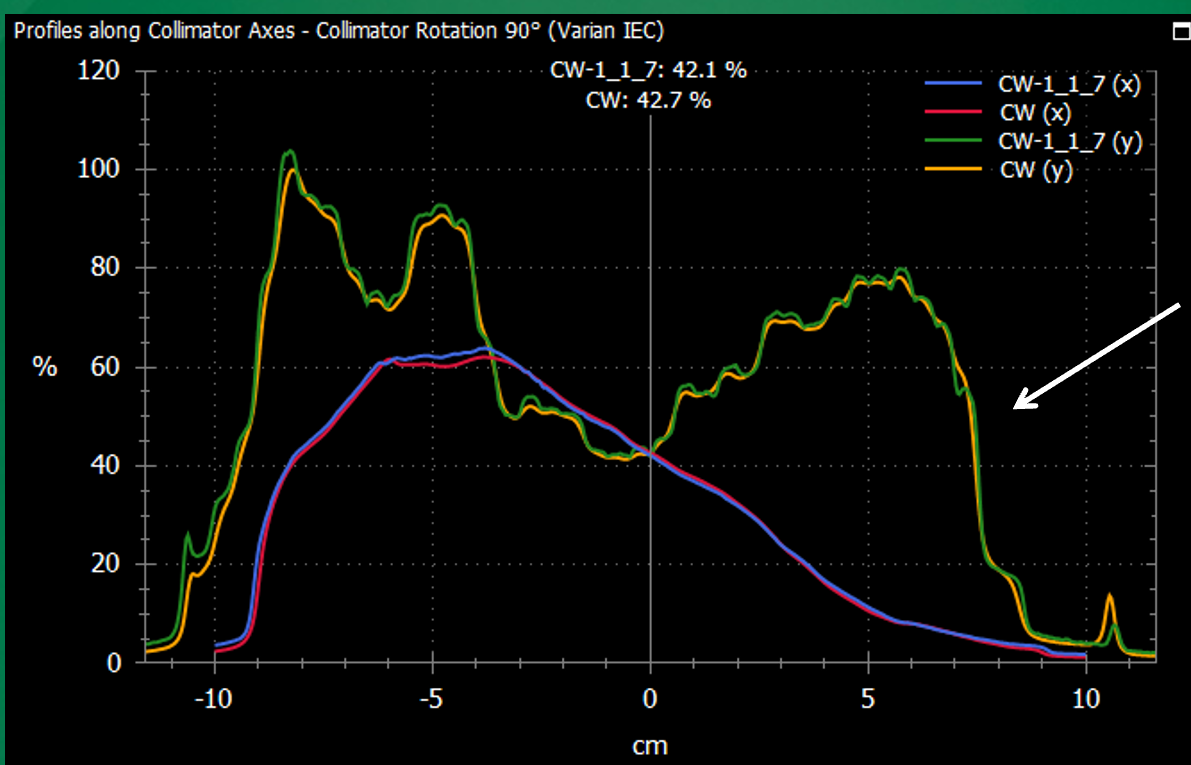
Why can large dose differences appear in high-gradient regions?
Small spatial misalignments can cause large dose differences.
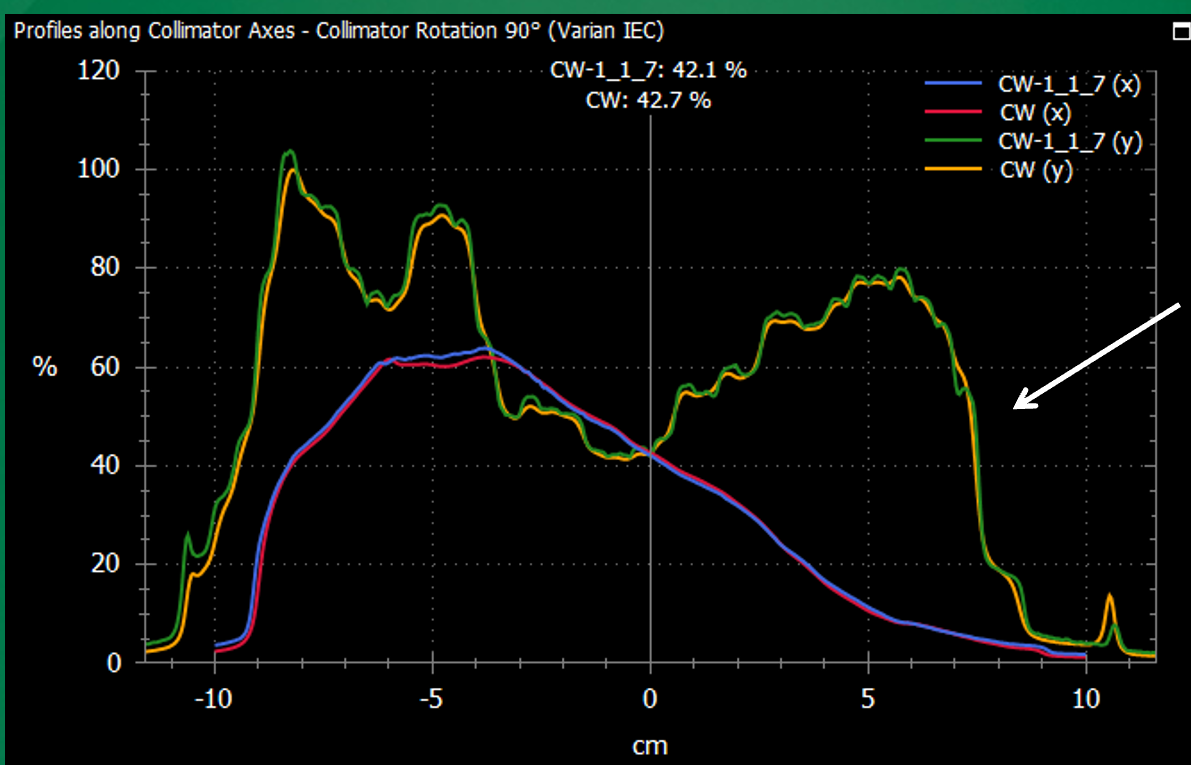
How can apparent discrepancies in dose profiles sometimes be resolved?
By recognizing small positional offsets that, when adjusted, improve agreement.
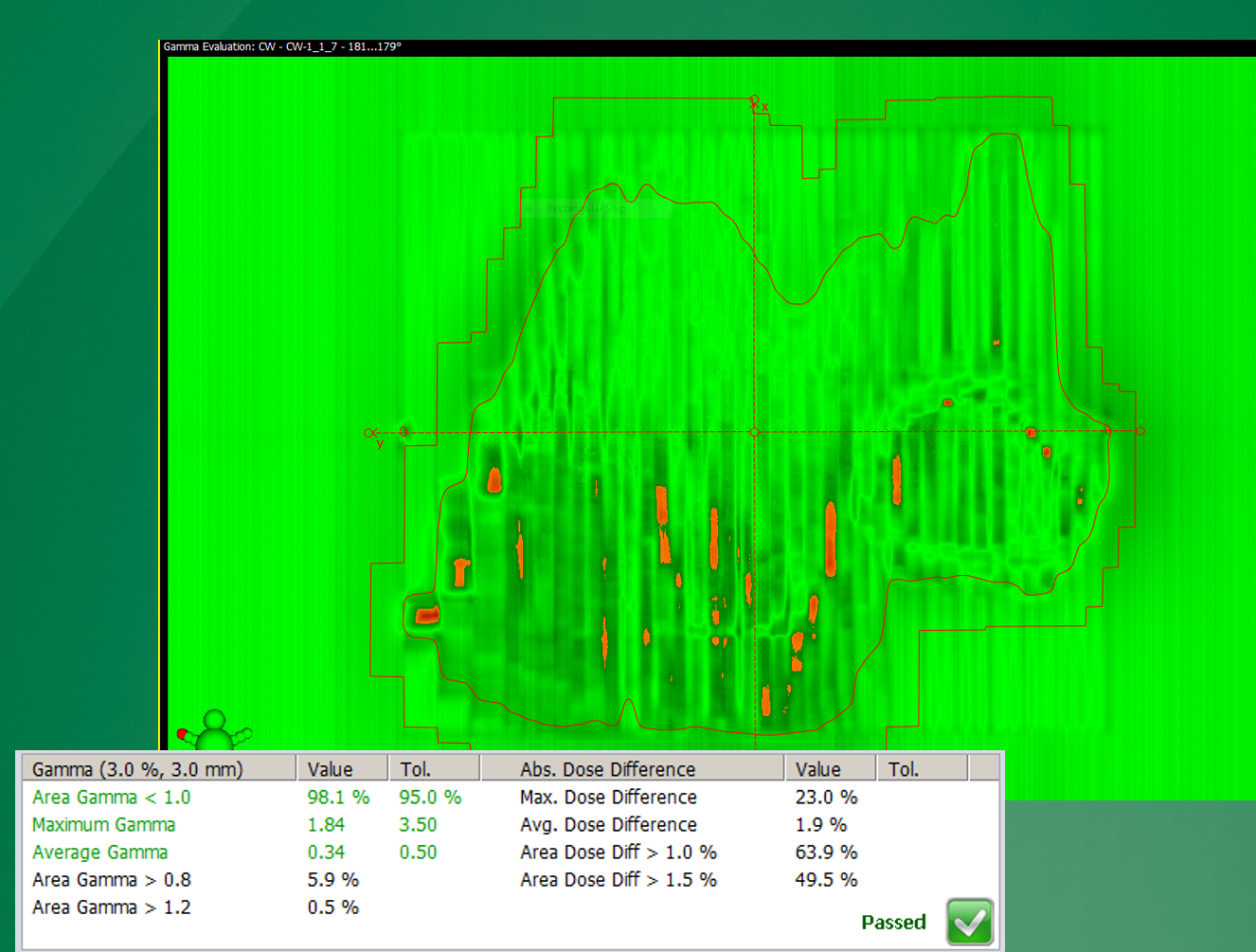
What is a gamma map used for in radiotherapy QA?
To quantitatively compare measured vs predicted dose using both dose and spatial agreement.
What does gamma evaluate?
How much dose difference and positional shift are needed for two distributions to match.
What is a typical gamma criterion?
3% dose difference and 3 mm distance-to-agreement.
What is a common passing threshold for gamma analysis?
≥95% of points must have gamma ≤ 1.
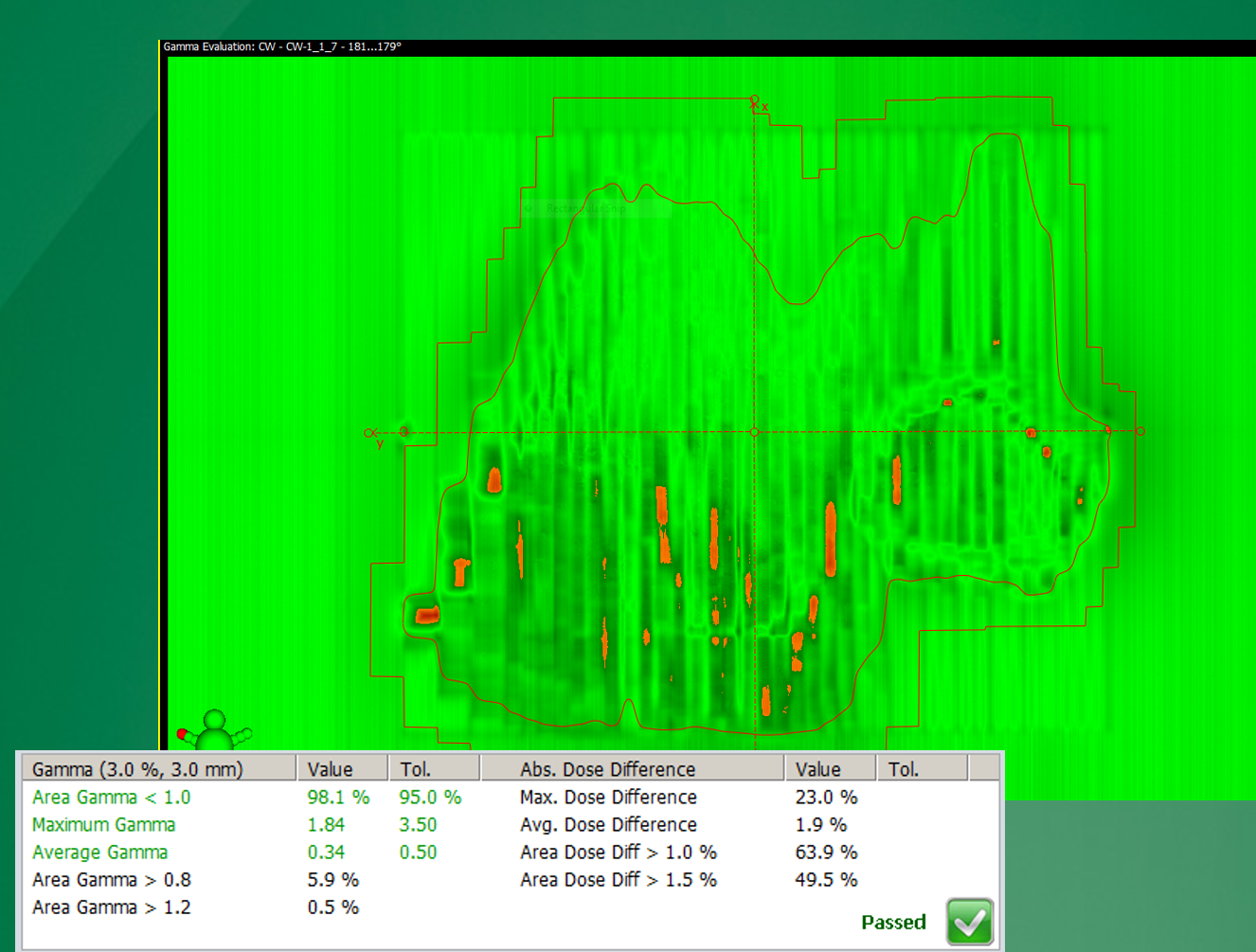
Why is gamma analysis preferred over simple dose difference?
It accounts for both dose and spatial discrepancies.
What can consistent gamma failures across a region indicate?
Mechanical issues such as MLC leaf speed or positioning errors.
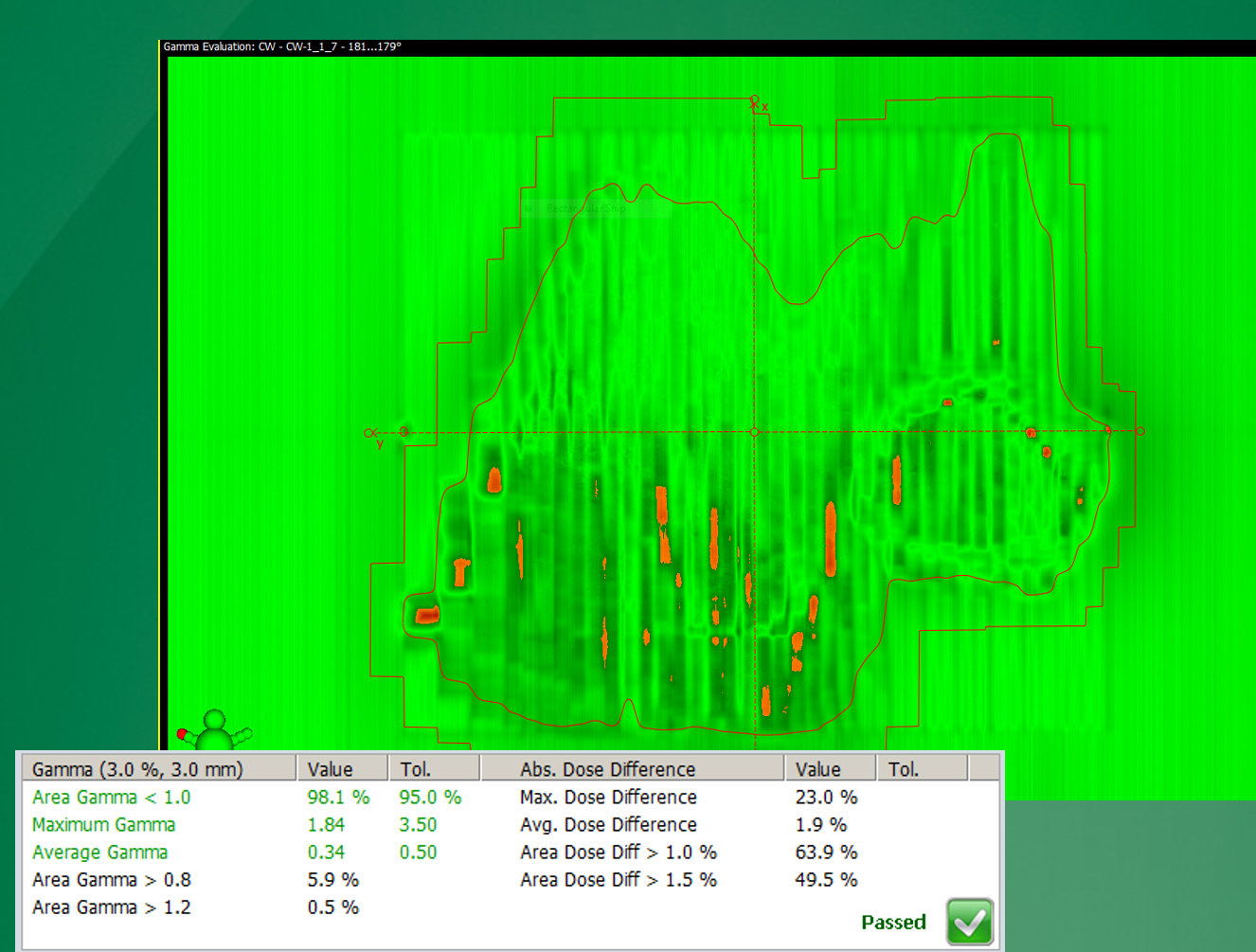
In what treatments is gamma analysis commonly used?
IMRT and VMAT QA with EPID verification.
What is 3D dose verification in radiotherapy?
Projecting planned fluence onto a 3D dataset (e.g., CT) to evaluate full dose distribution.
What is a key advantage of 3D verification over 2D (EPID)?
It assesses dose within a volume (patient/phantom), not just fluence.
How can 3D verification account for anatomical changes?
By recalculating dose on updated CTs (e.g., weight loss, tumor shrinkage).

How are water tanks used in 3D verification?
As simple phantoms to measure dose and compare with TPS predictions.
measure PDD and modulation factor verification
What detectors are used in 3D verification?
Ion chambers, film, TLDs, and diode arrays.

What is an end-to-end phantom test?
A full workflow test (imaging → planning → delivery → measurement) using a phantom with simulated targets and OARs.

Why are phantoms important for clinical trials or new techniques (e.g., SRS)?
To verify accuracy and meet required standards before treating patients.

How can motion be simulated in 3D QA?
Using moving phantom inserts to mimic tumor motion (e.g., lung motion, gating).

What is a 3D diode array phantom used for?
Measuring dose at many points simultaneously in 3D for IMRT/VMAT QA.

When is 3D verification preferred over EPID QA?
When more detailed volumetric dose information is needed or EPID results are uncertain.
Why is QA and portal dose verification essential in radiotherapy?
Although most plans pass, failures can lead to severe errors—e.g., a head & neck IMRT case with a replan under time pressure and no EPID QA resulted in a computer/MLC error, delivering ~7× the prescribed dose over 3 fractions with poor OAR sparing—highlighting QA’s critical role in patient safety.