electron configuration and ionisation energies
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
define first ionisation energy (2 marks)
the energy required to remove one mole of electrons from one mole of gaseous atoms
to form 1 mole of gaseous +1 ions
give the equation for first ionisation
X (g) → X+ (g) + e-
give the definitions for third ionisation energy
the energy required to remove one mole of electrons from one mole of gaseous 2+ ions
to form 1 mole of gaseous +3 ions
give the equation for third ionisation
X+2 (g) → X+3 (g) + e-
give the electron configuration of chromium
1s2 2s2 2p6 3s2 3p6 4s1 3d5
stability for having a half filled 3d orbital
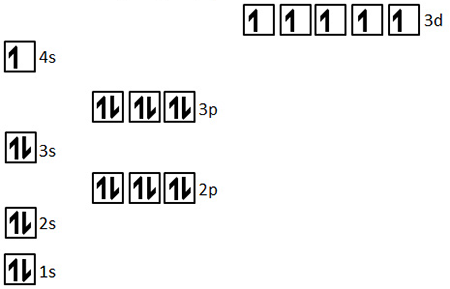
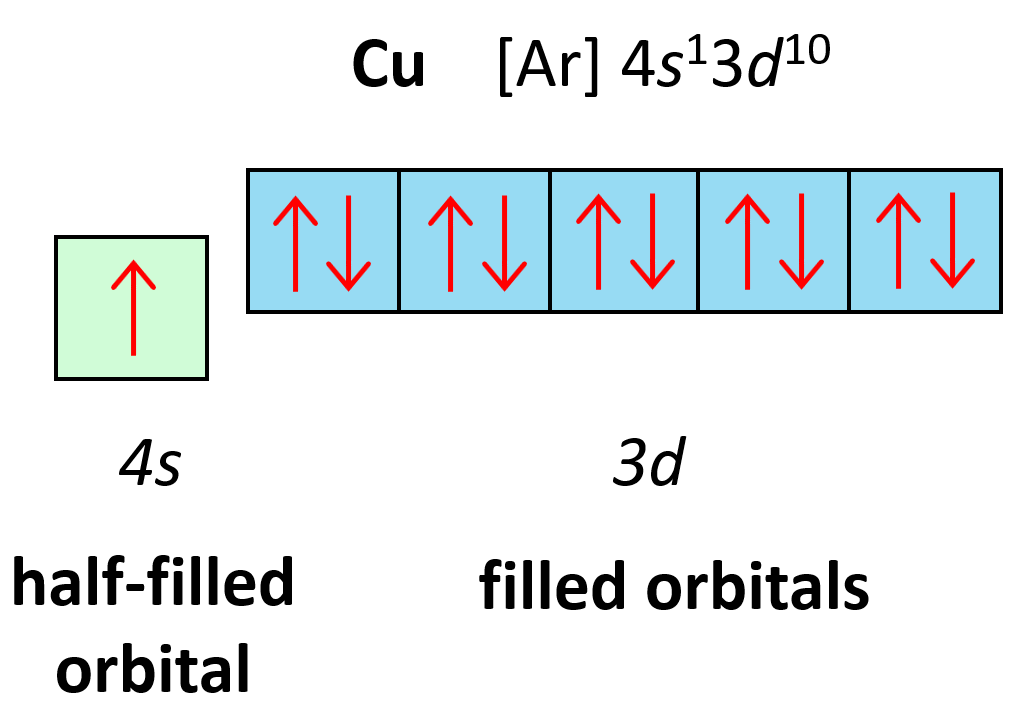
give the electron configuration of copper
1s2 2s2 2p6 3s2 3p6 4s1 3d10
stability for having a full 3d orbital
which are the 2 exceptions in electron config?
chromium
copper
what phrase should you use to answer exam questions on explaining trends in ionisation energies?
Never-number of protons and nuclear charge
Stop- shielding and shells
Dancing-distance from nucleus- atomic or ionic radius
Around- nuclear attraction
Earth- energy
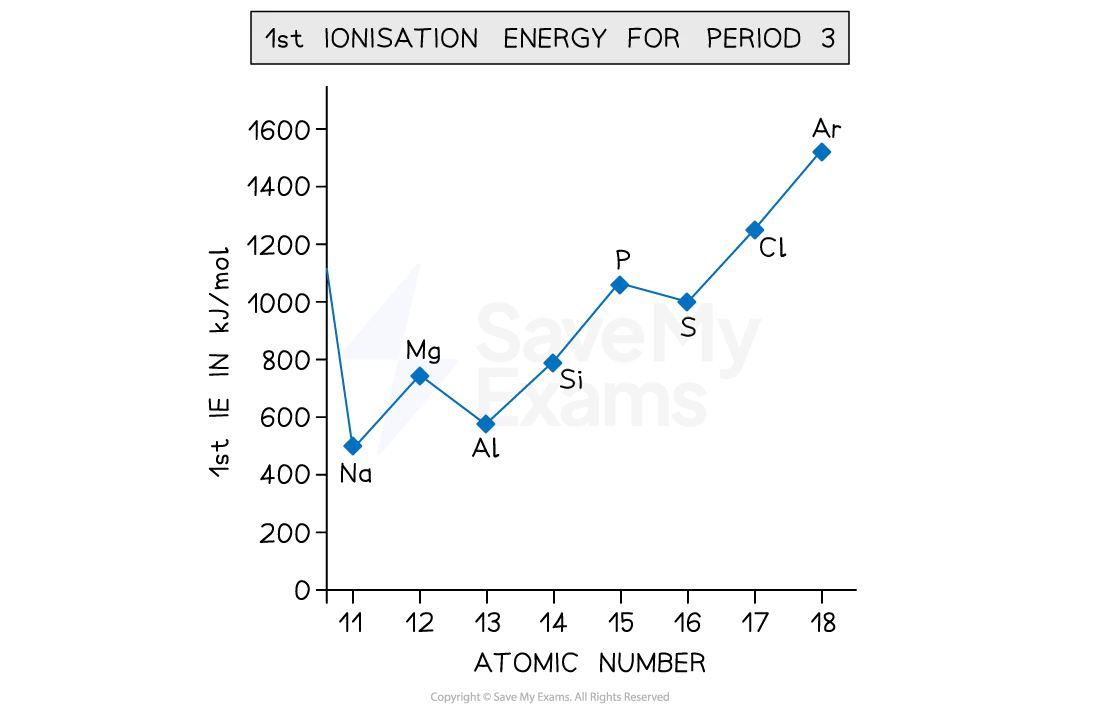
explain the general trend in first ionisation energy across period 3
across period 3
the number of protons increases
shielding stays the same
so the attraction between the nucleus and the outermost electrons increases
so the atomic radius decreases
so more energy is needed to remove an electron
so the first IE increases
give the electron configuration of Sc 2+
1s2 2s2 2p6 3s2 3p6
give the electron configuration of Mn 2+
1s2 2s2 2p6 3s2 3p6 3d5
Model answer for explaining trends in ionisation energies
the nuclear charge (increases/decreases)
shielding (increases/decreases/stays the same)
the distance between the nucleus and outermost electrons (increases/decreases)
nuclear attraction for the outermost electrons (increases/decreases)
the energy needed to remove an electron (increases/decreases)
compare the first ionisation energies of period 2 and 3
period 3 has a lower first ionisation energies than period 2
because shielding increases, which out weighs the increase in nuclear charge
therefore there is an increase in distance between the nucleus and outermost electrons
therefore nuclear attraction decreases
and the energy to remove one electron decreases
give the model answer for describe and explain the changes in first IE across a period
general trend NSDAE
1st dip
2nd dip (due to repulsion in an orbital or subshell due to each of them having different energies)
order the subshells from highest to lowest energy
f > d > p > s
describe and explain the changes in first IE across period 2 (7 marks)
the nuclear charge increases
shielding stays the same
the distance between the nucleus and outermost electrons decreases
nuclear attraction for the outermost electrons increases
the energy needed to remove an electron increases
from Be to B there is a decrease due to the P subshell having higher energy than the S subshell
from N to O there is a decrease due to repulsion due to the occupied P orbital
which of these have a higher 2nd IE? and explain why
Na+ Mg+ Al+
Na+ because the electron in the 2p subshell which is a higher energy subshell
therefore requires more energy to remove
an element in period 3 has the following succesive first IE
1681, 3374, 6050, 8408, 11203, 15164, 17868, 92038, 106434
identify which element and explain why (2 marks)
there is a large increase in energy from the 7th and 8th IE
therefore must have 7 electrons in it’s outershell
so must be chlorine
sketch a graph for the first 5 Ionisation energies of Na
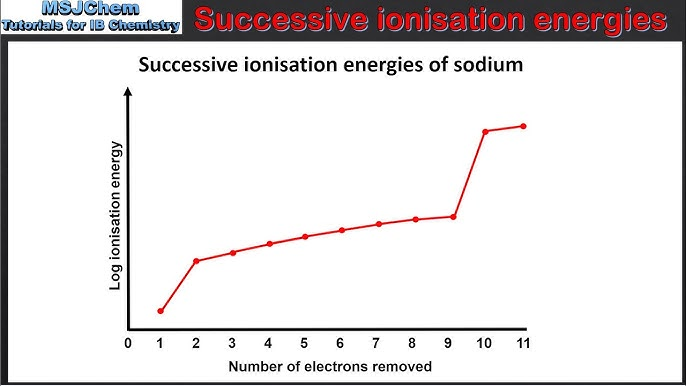

and explain your answer
D because the furthest electron has the least nuclear charge from the nucleus.
and it is an even number. (Doesn’t want to gain electrons)