Quiz 10: Biomedical Sciences (Anderson - Pregnancy+Lactation and Hep Viruses)
1/114
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
115 Terms
What hormones are produced by the corpus luteum during the luteal phase?
estrogen and progesterone
What is the role of estrogen and progesterone during the luteal phase?
prepare and maintain the endometrium for implantation
What rescues the corpus luteum during pregnancy?
human chorionic gonadotropin (hCG) from the blastocyst/placenta
Why is hCG important early in pregnancy?
maintains progesterone and estrogen production until the placenta takes over
When does the morula stage occur?
at the 12-16 cell stage
When does a blastocyst form?
at ~50-60 blastomeres
Where does early embryo development occur before implantation?
during transit through the uterine tube to the uterus
What major event occurs during implantation?
maternal blood-filled lacunae intercommunicate around the implanted blastocyst
What begins forming around the primitive yolk sac during implantation?
extraembryonic endoderm
What is the function of decidual layers?
separate fetal tissue from maternal tissue
What substances freely pass across the placental barrier?
nutrients, O₂, CO₂, and waste products
What are the two fetal tissue layers surrounding the embryo?
amnion and chorion
What is the role of the amnion and chorion?
amnion secretes amniotic fluid; chorion forms the fetal placenta
What maternal tissue forms the maternal portion of the placenta?
decidua
What is the major function of the placenta?
exchange gases, nutrients, growth factors, and waste
Do maternal and fetal blood directly mix?
No
How does maternal blood interact with chorionic villi?
maternal blood flows through intervillous spaces around the villi for exchange
Which fetal vessels carry oxygenated vs deoxygenated blood?
umbilical arteries = deoxygenated; umbilical vein = oxygenated
Why are chorionic villi important?
provide massive surface area for placental exchange
What are the characteristics and functions of hCG?
placental peptide hormone, pregnancy marker, rescues corpus luteum progesterone production
What are the functions of human placental lactogen (hPL)?
alters maternal metabolism (lipolysis/glucose metabolism) and supports fetal angiogenesis
What is CRH involved in during pregnancy?
timing of parturition (birth)
What are progesterone's major roles in pregnancy?
prevents endometrial shedding and uterine contractions
When does placental progesterone production begin?
after ~6 weeks gestation
What are estrogen's major roles during pregnancy?
uterine growth, breast development, and stimulation of contractions near term
What tissue becomes a major endocrine organ during pregnancy?
placenta
What tissue synthesizes estrogens during pregnancy?
placental syncytiotrophoblasts
Where do placental estrogen precursors come from?
fetal adrenal gland, fetal liver, placenta, and maternal tissues
What does DHEAS stand for?
dehydroepiandrosterone sulfate
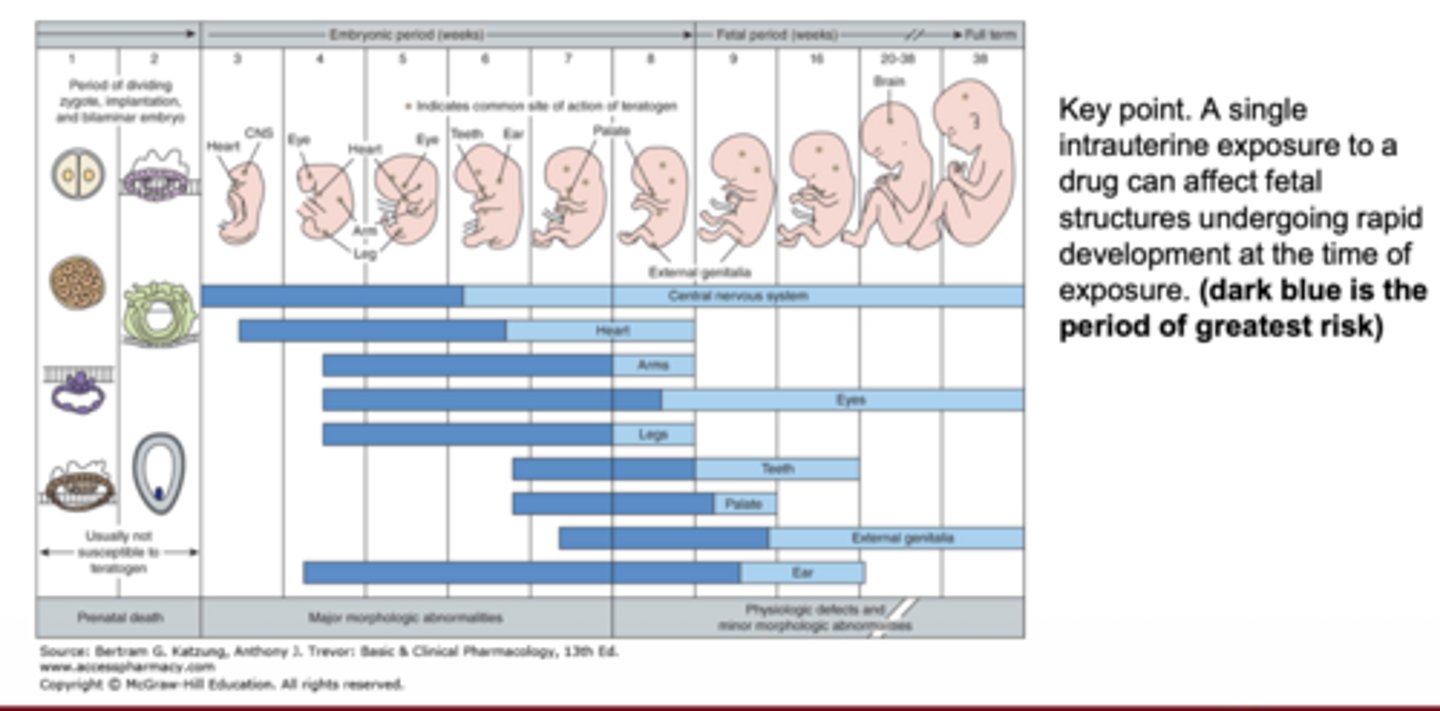
Why is timing of drug exposure important during pregnancy?
drugs can damage fetal structures undergoing rapid development at exposure time
What physicochemical property most favors placental crossing?
lipid solubility
How does molecular weight affect placental transfer?
MW 250-500 crosses easily; MW >1000 crosses poorly
Why do ionized drugs poorly cross the placenta?
charged drugs require transporters to cross membranes
How does protein binding affect fetal drug exposure?
free drug crosses more readily than protein-bound drug
What pregnancy-related factors affect fetal drug exposure besides drug properties?
amount reaching fetus, duration, tissue distribution, metabolism, transport rate, and developmental stage
What tissues significantly metabolize drugs during pregnancy?
placenta and fetal liver
What is the purpose of placental influx and efflux transporters?
bring nutrients in and keep toxicants/drugs out
What determines whether maternal drugs access fetal tissue?
drug physicochemical properties and transporter expression
Why can maternal drug responses change during pregnancy?
altered endocrine and physiologic states affect tissue responses
What maternal physiologic changes can alter drug effects?
changes in cardiac output and renal blood flow
Examples of predictable fetal drug toxicity.
opioid dependence, tetracycline tooth defects, thalidomide teratogenicity
Which fetal systems remain vulnerable for long periods during development?
CNS and external genitalia
What fetal toxicity is associated with tetracycline?
tooth staining during mineralization
What reproductive system changes occur during pregnancy?
uterus enlarges, cervix forms mucus plug, ovaries enlarge, vagina becomes more distensible
What is the role of the cervical mucus plug?
prevent microbial entry
How does the vagina adapt during pregnancy?
thickened lining, less connective tissue, more muscle tissue
What cardiovascular parameters increase during pregnancy?
blood volume, cardiac output, stroke volume, and heart rate
How does renal function change during pregnancy?
increased glomerular filtration rate and creatinine clearance
What contributes to increased GFR during pregnancy?
decreased renal vascular resistance
What are key breast anatomy and pregnancy-related changes important for lactation?
breasts contain lobes/lobules with alveoli producing milk; lactiferous ducts carry milk to nipple; estrogen causes duct/lobule proliferation during puberty and pregnancy

How does breast tissue appear before puberty?
mostly connective tissue with a few ducts
What effects does estrogen have on the breast during puberty?
stimulates duct branching, lobule/alveoli formation, and fat/connective tissue growth
How do menstrual hormones affect the breast?
fluctuating estrogen/progesterone alter fluid levels, circulation, and duct proliferation
What major breast changes occur during pregnancy?
significant duct proliferation and increased lobules/alveoli for milk production
What breast structures produce milk?
alveoli
Why are alveoli important pharmacologically?
drugs must cross the blood-milk barrier to enter milk within alveoli
What hormone primarily stimulates milk production?
prolactin
What hormone stimulates milk letdown/ejection?
oxytocin
What triggers oxytocin release during breastfeeding?
sensory stimulation of the nipple
Milk Let down Reflex Pathway
nipple stimulation → hypothalamus/posterior pituitary → oxytocin release → myoepithelial contraction → milk ejection
What major components are present in breast milk?
lipids, proteins, carbohydrates, vitamins, antibodies, and water
Why is breast milk important immunologically?
contains antibodies and immune-supportive factors for the infant
What cells synthesize milk in the lactating mammary gland?
alveolar epithelial cells
What physiologic changes support milk synthesis during lactation?
↑ blood flow, nutrient availability, protein synthesis, lipogenesis, and lactose synthesi
What forms the blood-milk barrier in alveoli?
tight junctions between alveolar cells
What pathways move substances into milk?
exocytosis/secretory pathways, transcellular transport, paracellular pathways, and transport proteins/channels
What substances are secreted into milk by alveolar cells?
proteins, lipids, sugars, salts, vitamins, minerals, antibodies, and nutrients
Are most maternal drugs detectable in breast milk?
Yes, but concentrations are usually low
What factors determine how much drug enters milk?
lipid solubility, transport mechanism, ionization, protein binding, molecular weight, and plasma concentration
How can nursing timing reduce infant drug exposure?
taking drugs 3-4 hours before nursing lowers circulating drug levels during feeding
What drug properties increase transfer/accumulation in breast milk?
low MW, weak bases, high lipid solubility, long half-life, and low protein binding
How do tight junctions affect drug entry into milk?
tight junctions help keep toxicants and many drugs out of milk; contamination depends on permeability and transport proteins
What are the major physiologic functions of the liver?
1. removes toxins/drugs
2. metabolizes nutrients
3. produces bile
4. breaks down bilirubin
5. produces cholesterol
6. converts ammonia to urea
Why is liver dysfunction dangerous during hepatitis?
the liver is essential for detoxification, metabolism, bile production, clotting factors, and vitamin storage
Which hepatitis viruses are RNA vs DNA viruses?
HAV, HCV, and HDV are RNA viruses; HBV is a DNA virus
What are the pre-icteric vs icteric symptoms of hepatitis?
pre-icteric = malaise, anorexia, nausea (IFN-mediated)
icteric = jaundice, dark urine, RUQ pain
Are hepatitis viruses directly cytopathic?
No, immune-mediated inflammation damages hepatocytes
What is fulminant viral hepatitis?
massive liver necrosis leading to liver failure and possible death
What causes jaundice in hepatitis?
incomplete elimination of bilirubin by the damaged liver
What are clinical signs of bilirubinemia?
yellow skin/sclera and tea-colored urine
How is bilirubin normally formed and eliminated?
heme from old RBCs → broken down to bilirubin → kidney conversion to urobilin → elimination in bile/feces or urine
Why does bilirubin accumulate during viral hepatitis?
damaged hepatocytes cannot properly uptake/conjugate bilirubin
What histologic changes occur in chronic hepatitis?
mononuclear inflammatory infiltration and hepatocyte necrosis
Why can chronic hepatitis go undiagnosed?
many chronic infections are asymptomatic and only detected by blood testing
What symptoms may occur in chronic hepatitis?
fatigue, anorexia, malaise, and RUQ pain
What severe complications can chronic hepatitis cause?
cirrhosis, variceal bleeding, encephalopathy, coagulopathy, hypersplenism, and ascites
What causes cirrhosis during chronic hepatitis?
chronic inflammation causes fibrosis and abnormal liver architecture
How does cirrhosis alter liver blood flow?
fibrosis and endothelial changes impair sinusoidal blood flow and hepatocyte access to blood
What are major consequences of portal hypertension?
ascites, varices, and splenomegaly
What complications result from liver cell failure in cirrhosis?
encephalopathy, coagulation defects, edema, bleeding tendency, and excess estrogen effects
Why are esophageal varices dangerous?
rupture can cause catastrophic bleeding and death
How do hepatitis viruses contribute to HCC?
persistent inflammation and liver regeneration increase DNA mutations and carcinogenesis
Hepatitis Virus Summary
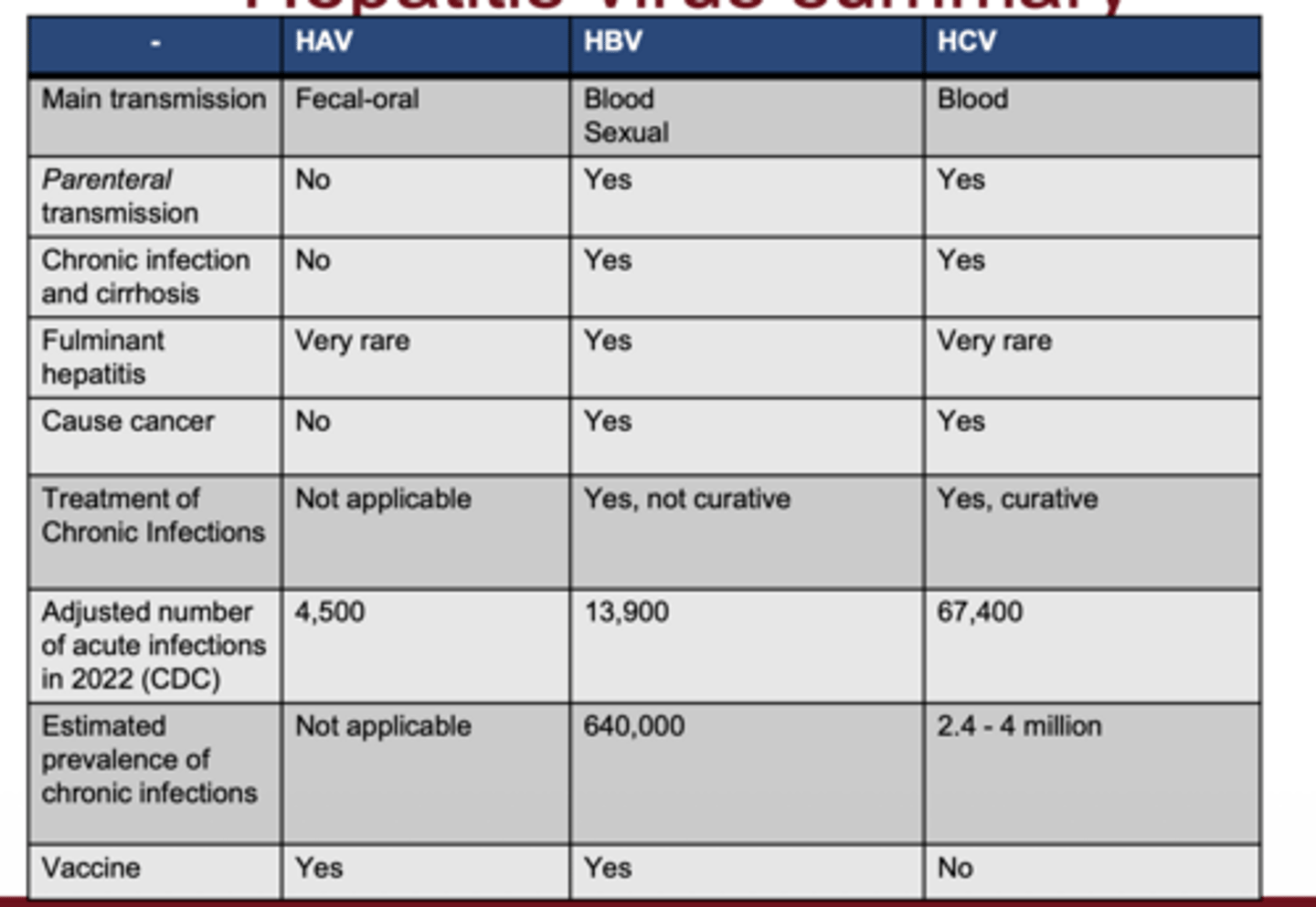
Which hepatitis viruses are transmitted fecal-orally vs through blood?
HAV = fecal-oral; HBV/HCV = bloodborne
Which hepatitis viruses have vaccines?
HAV and HBV have vaccines; HCV does not
What are key properties of HAV?
picornavirus, ssRNA, non-enveloped, enterovirus genus
How is HAV transmitted?
fecal-oral route through contaminated food/water, poor hygiene, or unsanitary sexual activity
Why is HAV easily spread environmentally?
stable to mild heat and survives on surfaces for weeks
HAV pathogenesis
replicates in GI mucosal cells → spreads via blood → infects hepatocytes → cleared by cell-mediated immunity
Does HAV cause chronic hepatitis?
No chronic hepatitis or carrier state