Exam 3 - Principles of Chemistry
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
what is a boiling point? high bp = ?
liquid turning into a gas ; high bp = higher melting point
what is vapor pressure? low vp = lower ?
gas leaving a liquid surface which breaks liquid - liquid IMFs ; volatility
what is viscosity?
how easily a liquid flows (high viscous liquids flow slowly)
what is apart of IMF’s? relative strength
stronger intermolecular attrations which means harder to break and higher heat/energy)
relative strength depends on: bond polarity, molecular polarity, polarizability
what are the 3 parts of stronger IMF’s? EXPLAIN boiling points / vapor pressure / viscosity
higher BP
lower vapor pressure:
higher viscosity
define intermolecular forces and examples
interactions between molecules arise from their charged regions (from electrons and protons) that ATTRACT or REPEL each other with forces that vary on the distance
hydrogen bonding, london dispersion
define intramolecular forces
hold atoms together within a single atom
covalent and ionic bonds
which are stronger? intra/inter
intra
what is the range for polar covalent bonds?
>2
more susceptible to being polarized = _____ dispersion forces
stronger
polarizability is generally larger with ____ electrons over a ____ volume and a ____ exposed area
more, larger, more
do all molecules have dispersion forces and induced dipole moments?
yes
which forces have the largest contribution to IMFs between atoms and molecules?
dispersion forces (induced dipoles)
which orientation would you expect dispersion interactions to be stronger?
the larger the contact area of interaction, the stronger the IMF
what are the two forces that make up total IMF’s?
dispersion and dipole dipole
Just because it is polar doesn’t automatically mean higher. You must count electrons. T/F
true
IMF polar bonding interaction is ____ (is/is not) a covalent bond
not
what effect do branches have on IMF?
the presences of branches reduces the effective contact area over which molecules can interact → decreasing IMFs
what happens in lower IMF? melting point, BP, viscosities, volatilities
lower
melting point, boiling point, viscosities
higher
volatilities (substance ability to transition)
what are ideal conditions for mixing?
lower PE, more configurations
Particles are ____ likely to adopt those states where the IMFs between them are stronger (lower PE)
more
Particles are ____likely to adopt states with more configurations.
more
explain exothermic vs endothermic
exothermic: energy released - endothermic: energy taken in
in a substance that mixes, what would you expect the heat of mixing to be greater than zero?
exothermic (lowers E)
Since mixing is exothermic, adding heat would ___ improve miscibility. (maybe
not
what is the effect of increasing or decreasing temperature on solubility and miscibility (mixing)?
generally - solubility and miscibility increases with increasing T (ususally true but are independent of eachother)
when mixing is favored, it is ____ (forward/backward)
forward
when UM and M are even, temperature (always/never) has an impact
never
define monomer
a repeating subunit used to make a polymer
define polymer
a large molecule made of repeating subunits
define macromolecule
a molecule with a very high molecular weight
what factors affect the properties of a polymer?
chain length, intermolecular forces, extend of chain branching
in a condensation reaction, what do you remove?
H2O (“water elimination”)
explain the importance of branching and chain length
branches create more structures within chains (disrupt IMFs)
longer chain length = higher density
what is density in regards to chain length and branching?
the amount of IMFs in a region
(more branches leads to less density)
what does a crosslink do to a polymer? what is the affect?
cross link (ionic, covalent, or hydrogen bonding) is a binder between polymer chains
increase strength, rigidity (decrease flexibility), and melting point
plasticizer contain _____ which are added to _____ (improve/decrease) flexibility
phthalates, improve
when proteins are synthesized inside a cell, they exist as _____ (folded/unfolded)
unfolded
what causes proteins to fold in intramolecular interactions?
interactions between amino acid residues
what causes proteins to fold in intermolecular interactions?
interactions with water molecules
why do proteins fold?
they fold into the most structural conformation that is the most energetically stable in that enviornment
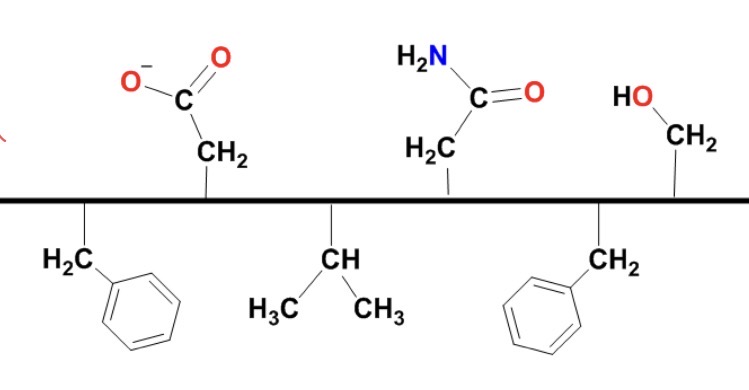
How would you expect this protein to fold if immersed in oil or water?
oil: hydrophobic - the bottom side prefers oil (interactions that are primarily dispersion force IMF’s)
the protein will fold to expose the hydrophobic side chains to interact w the lipid tails (dispersion)
water: hydrophilic - the top side prefers H2O; proximity for H-bonding and dipole-dipole IMFs
the protein will fold so that the hydrophobic (benzene rings, ect) are tucked into the inner core
define saturation and single vs double bond
refers to how many of the hydrocarbons are saturated (filled) with hydrogens
saturated: contain only a single bond are are “saturated” with the maximum amount of hydrogen
unsaturated: double or more bonds which means fewer hydrogen atoms
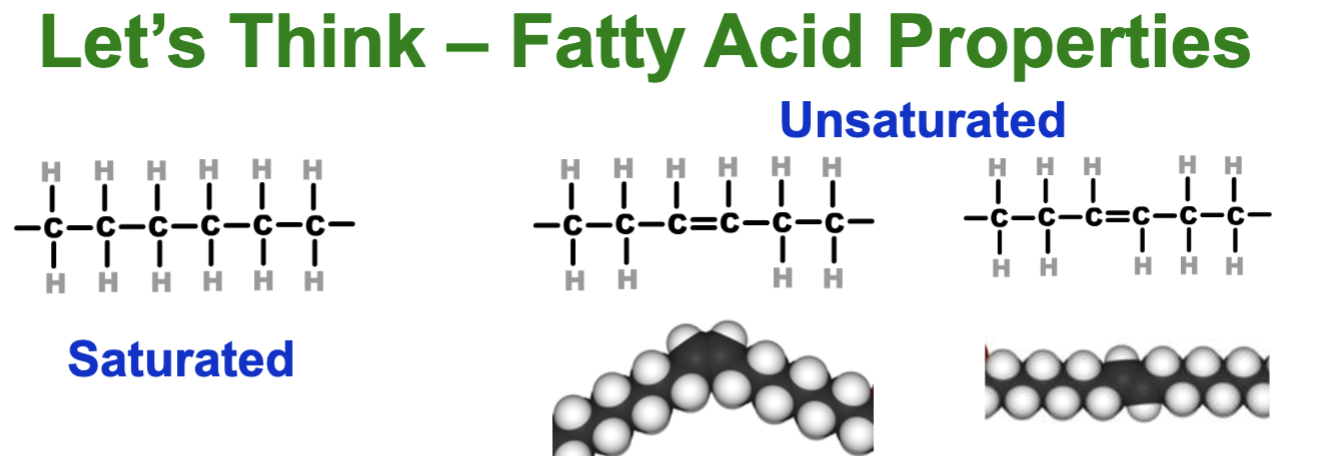
which is a trans or cis configuration?
cis (left, same side hydrogens) ; trans (right, different side hydrogens)
what are bonds and cis/trans related with fats?
if there are more double bonds - there is less effective dispersion (causes bend and liquid at room temp)
cis causes more bends in the chain so less effective dispersion IMF (h same side)
trans keeps the chain straight (hydrogen on opposite sides) - tightly packed (artificial butter solid at room temp)