Functional Groups and Carboxylic acid derivatives
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
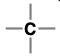
Alkane (Substituent: alkyl)
a hydrocarbon with no multiple bonds
nonpolar
tetrahedral (sp3 hybridized)
free radical reactions
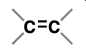
Alkene (Substituent: alkenyl)
a hydrocarbon with at least 1 C-C double bond
nonpolar
trigonal planar (sp2 hybridized)
addition reactions, oxidative cleavage
stability increases with # carbons attached
Alkyne (Substituent: alkynyl)
a hydrocarbon with at least 1 C-C triple bond
nonpolar
linear (sp hybridized)
addition reactions, oxidative cleavage, acid-base reactions
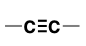
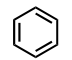
Benzene Ring
(Substituent: phenyl)
Alcohol
(Substituent: hydroxy)
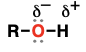
ether
(Substituent: alkoxy)
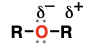
alkyl halide
(Substituent: haloalkyl)
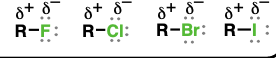
amine
(Substituent: amino)
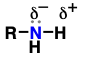
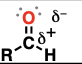
aldehyde
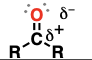
ketone
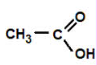
carboxylic acid
(suffix: -oic acid)
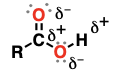
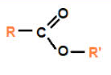
ester
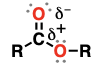
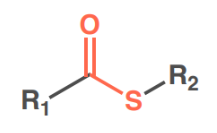
thiol
(Substituent: mercapto)
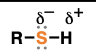
sulfide
(suffix: sulfide)
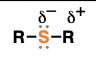
epoxide
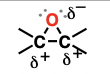
nitrile
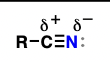
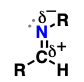
imine
nitrogen-containing analogues of ketones and aldehydes
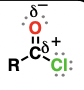
acyl halide
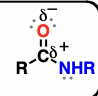
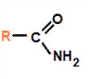
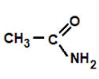
amide
anhydride
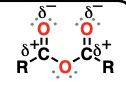
Nitro
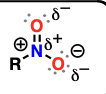
Enol
alkenes attached to a hydroxyl substituent
good nucleophiles, will perform addition reactions to aldehydes and ketones
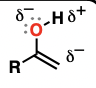
Enolate
the conjugate bases of enols
excellent nucleophiles
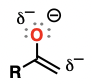
Enamine
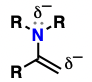
alkenes attached to an amino substituent
excellent nucleophiles
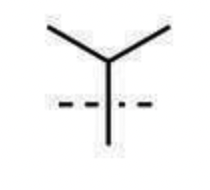
tert-butyl

isopropyl
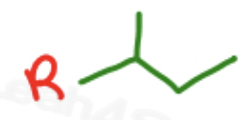
isobutyl

sec-butyl
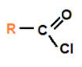
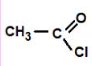
Acid Chloride
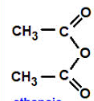
Acid Anhydride
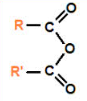
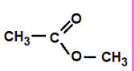
Ester
Amide
Nitrile
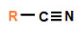
ethanoic acid
ethanoyl chloride
ethanoic anhydride
methyl ethanoate
ethanamide
ethanenitrile
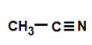
thioester