Super Mineralogy Midterm (copy)
1/120
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
121 Terms
True or False?
Most minerals have a single cation or cationic group, while they often have more than one anion.
False.
Most minerals have a single anion or anionic group, while they often have more than one cation.
Crystal Chemistry
relationship of chemical composition to atomic structure
Mineral
A mineral is a naturally occurring solid with a highly ordered atomic arrangement and a definite (but not fixed) chemical composition. It is usually formed by inorganic processes
Steno’s Law
"In crystals of the same species (different specimens), under equal conditions of pressure and temperature, the corresponding dihedral angles are always the same, the number, shape and size of the faces being variable"
The 5 chemical bonds
ionic
covalent
metallic
van der waals
hydrogen
Compounds ending in–ide…
are simple binary compounds containing 2 elements even if there is no metal
Compounds ending in–ate…
means oxygen is present
Ending in –ite…
have less oxygen present than in –ate compounds
4 Crystal Bonding Types
Molecular Crystal
Covalent Crystal
Metallic Crystal
Ionic Crystal
radius ratio
is the ratio of the cation radius to the anion radius
symmetry
Property that makes an object (motif) coincide with another identical one through a movement called the symmetry operation
4 Basic Symmetry Operations
Translation
Rotation
Reflection
Inversion
6 Crystal Systems
Triclinic
Monoclinic
Orthorhombic
Tetragonal
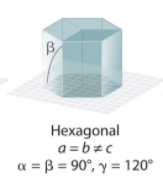
Hexagonal
Isometric
Crystal shape is determined by _______
Atomic arrangement
THE BECKE LINE WILL ALWAYS MOVE ONTO THE MATERIAL WITH THE________ AS THE STAGE IS LOWERED
HIGHER REFRACTIVE INDEX
When the RI of the mineral and the RI of the mounting material are equal, the Becke line…
splits into two lines, a blue line and an orange line.
Anisotropic minerals extinct _____ times
4
Isotropic minerals extinct ___ times
Isotropic minerals are always extinct. This is because they cannot reorient light
What causes extinction?
The direction of vibration of light in the mineral is parallel to the direction of polarization
Uniaxic crystal systems
Hexagonal
tetragonal
biaxic crystal systems
rhombic, monoclinic, triclinic
C axis refers to the…
Optic axis
All uniaxial minerals show ____ extinction
parallel
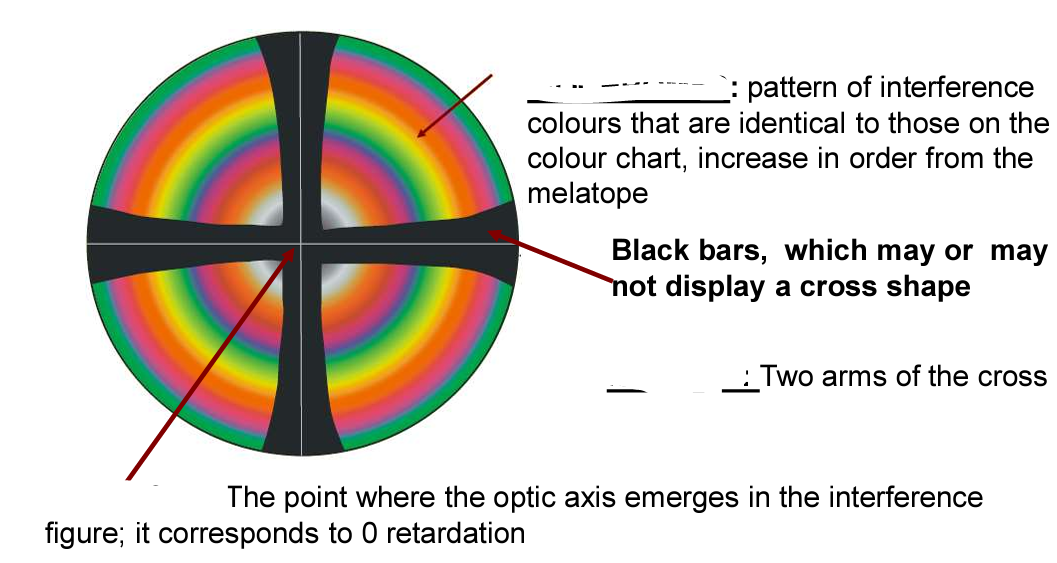
What type of interference figure is this + label the elements
Centered Uniaxial Interference Figure
Isochrome: pattern of interference colours that are identical to those on the colour chart, increase in order from the melatope
Isogeres: Two arms of the cross
Melatope: The point where the optic axis emerges in the interference figure; it corresponds to 0 retardation
The 3 Uniaxial Interference Figures
Optic Axis Figure-OA vertical
Off Centred Optic Axis Figure- OA inclined
Flash Figure- OA horizontal
Which quadrant helps you determine the sign?
The NE quadrant
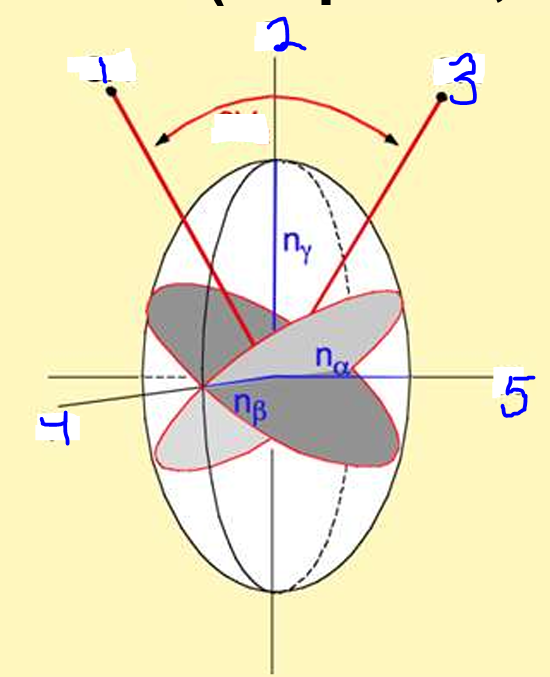
Label 1-5
Optic Axis
Z axis
Optic Axis
Y
X
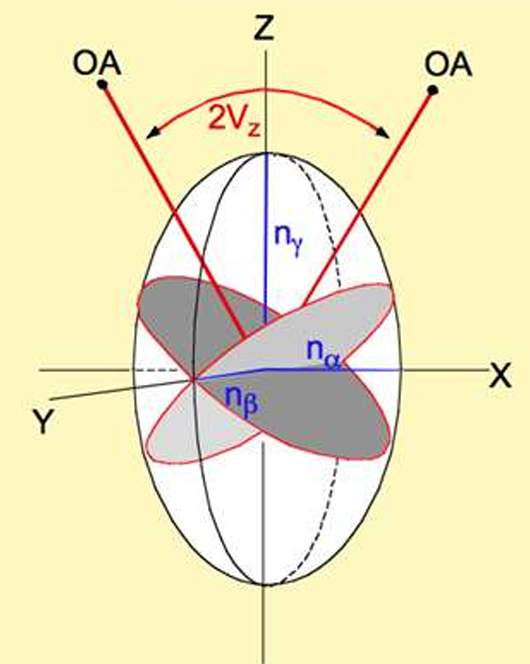
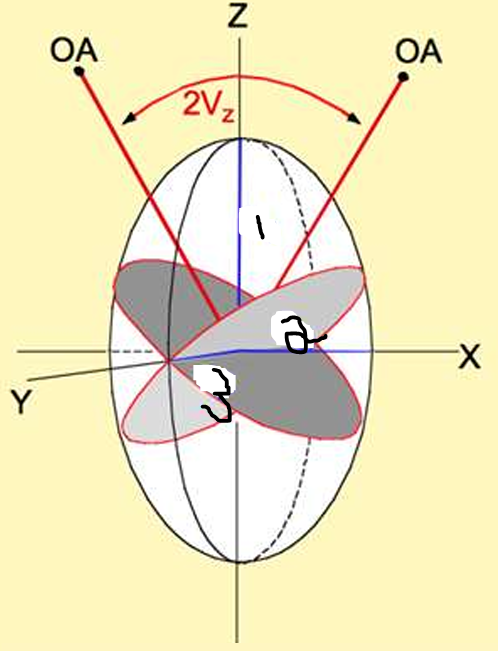
direction X = ?
direction Y = ?
direction Z = ?
direction Z = nγ (highest)
direction X = nα(lowest)
direction Y = nβ (intermediate; ray of circular section)
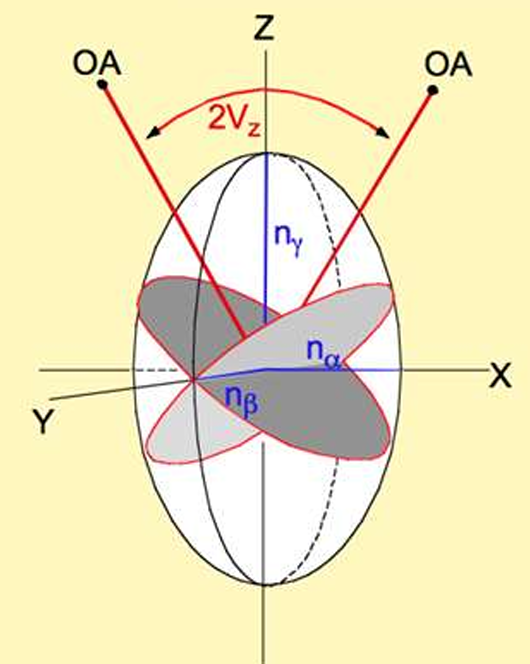
How is 2V measured?
Using the interference figures
Bxa stands for…
Acute Bisectrix
OA stands for…
Biaxial Optic Axis
Bxo stands for…
Obtuse Bisectricx
ON stands for…
Biaxial Optic Normal or Flash Figure
The appearance of the interference figure is dependent on the ____ of the mineral grain and its corresponding______
orientation ; indicatrix
Isotropic
speed of light is the same in any direction.
Vibration direction will always be perpendicular to the propagation direction
only ONE index of refraction
Anisotropic
speed of light varies depending on the direction the light is traveling in
more than one index of refraction
“Cross Nichols” refers to…
adding the analyzer (XPL)
Pleochrism
minerals ability to change its color when its position is changed on the stage
The slow ray in an anisotropic mineral will have a _____ than the fast ray
higher index of refraction
The fast ray is the ______ and is _______ when going through the mineral
extraordinary ray ; not deviated
The slow ray is the ______ and is _______ when going through the mineral
ordinary ray ; slowed down + deviated
the sign of elongation is POSITIVE if the ____ beam vibrates ____ to the maximum length of the crystal
slow ; parallel
the sign of elongation is NEGATIVE if the ____ beam vibrates ____ to the maximum length of the crystal
fast ; parallel

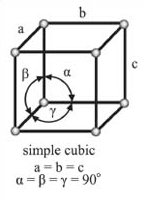
Isometric Crystal System Angles?
a=b=c α=β=γ=90
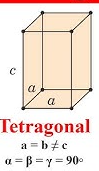
Tetragonal Crystal System Angles?
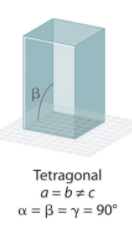
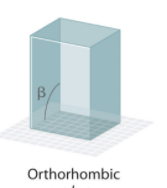
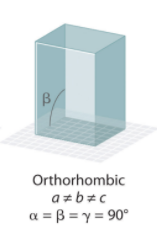
Orthohombic Crystal System Angles?
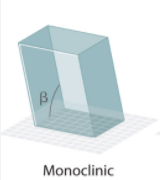
Monoclinic System Angles?
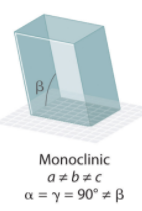
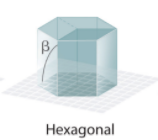
Hexagonal System Angles?
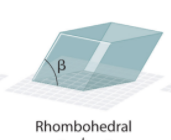
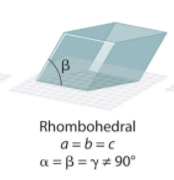
Rhombohedral System Angles?

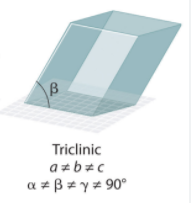
Triclinic System Angles?
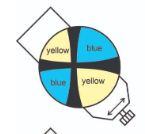
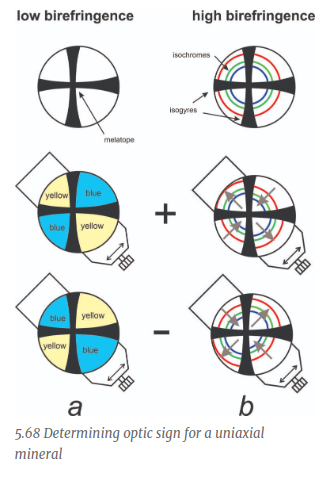
When the NE Quadrant is Blue, what is the sign?
Positive
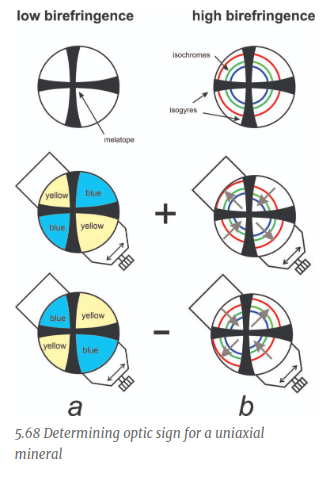
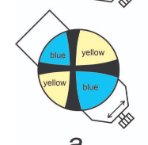
When the NE Quadrant is Yellow, what is the sign?
Negative
How much symmetry does the isometric/cubic system have?
High
How much symmetry does the Hexagonal system have?
Medium
How much symmetry does the Tetragonal system have?
Medium
How much symmetry does the Trigonal system have?
Medium
How much symmetry does the Rhombic system have?
Low
How much symmetry does the Monoclinic system have?
Low
How much symmetry does the Triclinic system have?
How much symmetry does the isometric system have?
Name the four basic symmetry operations
Translation, Rotation, Reflection, Inversion
Name the three composite symmetry operations
Rotation+Inversion (Rotoinversion), Rotation+Translation (Screw Axis), Reflection+Translation (Slip Plane)
Define Motif
fundamental part of a symmetrical design, which when repeated, creates the entire pattern
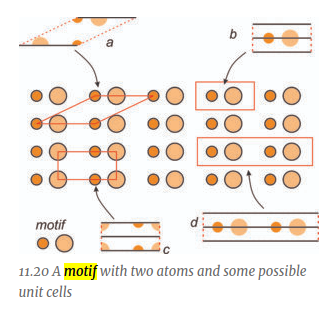
Define Operation
some act that reproduces the motif to create a pattern
Define Symmetry Element
an operation located at a particular point in space
Define Symmetry Plane
"Imaginary plane that divides the body into two identical parts"
Define Axis of symmetry
Imaginary line that crosses a body, around which, when the body is rotated, it presents the same appearance multiple times in a complete rotation
Define Center of Symmetry
“A part of a perfect body is diametrically opposite to another similar face located on the opposite side of the body ”
When a mineral shows nearly black interference colors, what type of indicatrix section is in the plane of the mineral?
CIRCULAR SECTION
The interference colors shown by a mineral under cross-polars depends on the thickness of the mineral and what else?
BIREFRINGENCE OR DIFFERENCE IN 2 INDICES OF REFRACTION IN SECTION
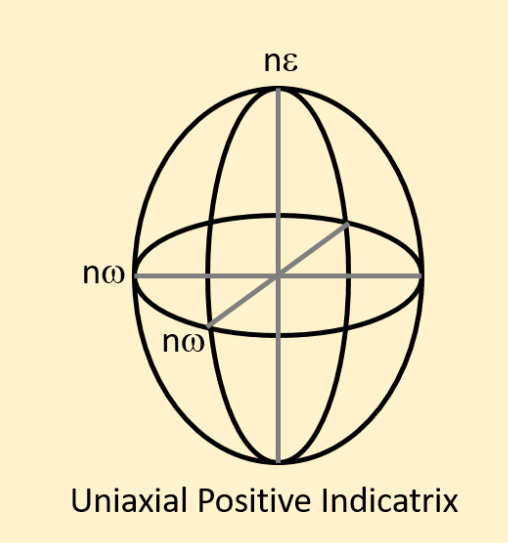
In a positive uniaxial indicatrix with light vibrating in the omega direction would it be the fast or slow ray?
FAST RAY (LOWEST N)
What is an indicatrix?
The indicatrix is a 3-dimensional visualization of the indices of refraction of a mineral.
What’s another name for the C axis in uniaxial indicatrix?
OPTIC AXIS
Define Uniaxial Positive
Uniaxial birefringence is classified as positive when the extraordinary index of refraction ne is greater than the ordinary index no
Define Uniaxial Negative
For uniaxial minerals these two extreme values of refractive index are defined as ω (or No) and ε (or Ne). Values between ω and e are referred to as ε'. Uniaxial minerals can be further divided into two classes. If ω > ε the mineral is said have a negative optic sign or is uniaxial negative.
A mineral in which the index of refraction (n) is the same in all vibration directions is said to portray what kind of optical properties?
ISOTROPY/ISOTROPIC
Define Relief
The degree to which the mineral grain stands out from the surrounding material in thin section.

How much relief is shown?
Strong relief
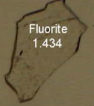
How much relief is shown?
Moderate to strong
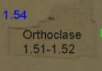
How much relief is shown?
Low
Define positive relief
Relief is said to be positive when the refractive index of the mineral is higher than the refractive index of the medium,
Define negative relief
Relief is said to be negative when the refractive index of the mineral is lower than the refractive index of the medium
When the angle of incidence exceeds the critical angle of refraction, what will happen?
THE LIGHT WILL REFLECT or Total Internal Reflection
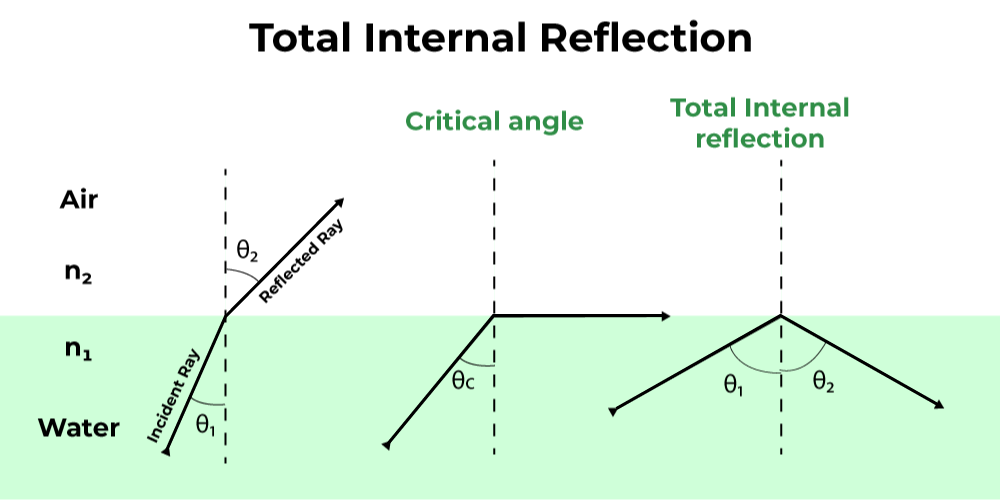
Index of refraction is a measure of?
HOW MUCH LIGHT IS SLOWED DOWN
What property of light does amplitude control?
INTENSITY or BRIGHTNESS
What type of energy is light?
ELECTROMAGNETIC RADIATION
What property light does wavelength control?
Color
What property of light does the index of refraction control?
Speed
define Birefringence
when light is refracted and split into 2 different rays (slow and fast) and it depends on the difference in the index of refraction
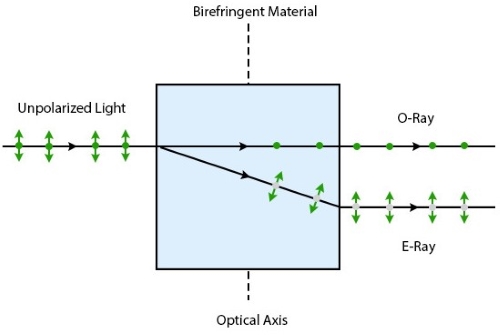
Define Diffraction
the random scattering of light waves
Define Dispersion
Dispersion of light is the process of separating white light into its individual colors

Define Pleochroism
Pleochroism is an optical phenomenon in which a substance has different colors when observed at different angles, especially with polarized light.

Define Refraction
the bending of light, or changing the path angle of light

What is Snell’s Law?
Snell's law is a formula used to describe the relationship between the angles of incidence and refraction
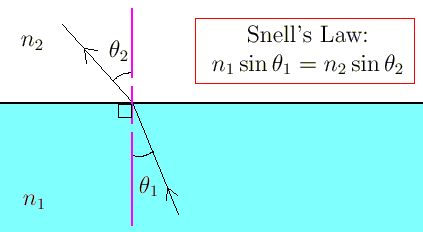
What is the atomic number defined by?
The number of protons in an atom
Define Iridescence
Iridescence is a optical phenomenon that causes a mineral to display a play of colors. It is caused by interference of light waves reflected from thin layers within the mineral.
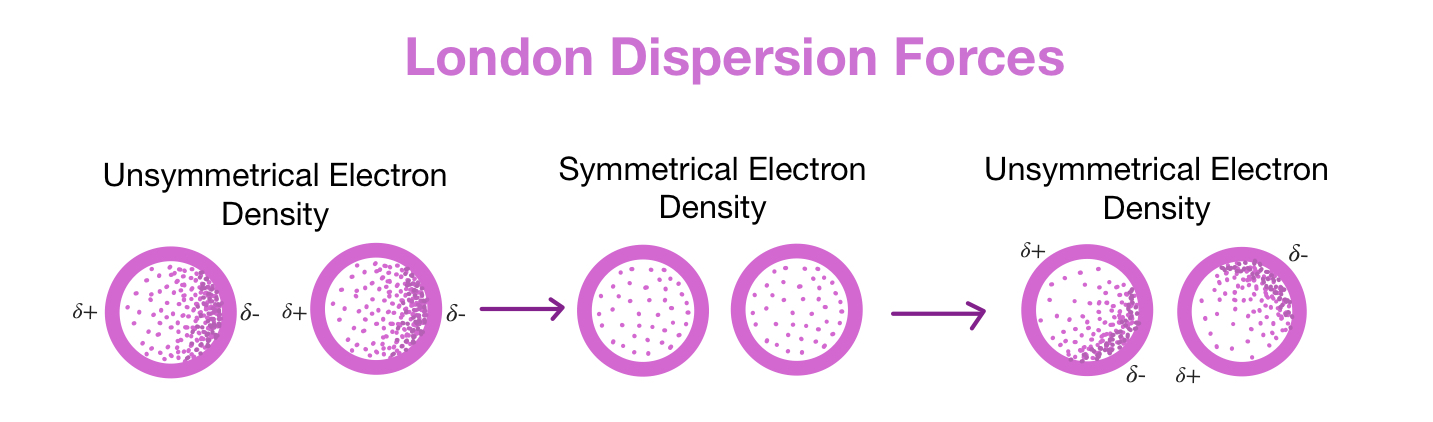
T/F, Dispersion forces are only experienced in molecules with permanent dipoles?
True , dispersion forces (also known as London dispersion forces) are experienced by all molecules, regardless of whether they have permanent dipoles or not, as they arise from temporary fluctuations in electron distribution within the molecule
T/F, Ionic, covalent and metallic bonds are examples of intermolecular bonds.
False. They are interatomic bonds
T/F, Ionic radii increase with increasing coordination number.
True. An atom's coordination number is the number of other atoms that it bonds to
T/F, Minerals whose bonds are at least partially metallic are transparent in thin sections
False. metallic bonds are not transparent even in a thin section because the free electrons within the metal readily absorb visible light, making metals inherently opaque
T/F, The most common type of phase diagram for binary systems is a temperature-composition diagram
True