Radioactivity
1/69
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
What is ionising radiation?
Radiation that can ionise atoms by removing an electron to leave a positive ion
Describe how a cloud chamber allows us to see tracks of ionising radiation
The cloud chamber contains air saturated with vapour at a very low temperature.
The radiation ionises air molecules in the chamber
Liquid condenses onto the ions to leave tracks of droplets marking the path of the radiation
What are the two types of beta radiation?
Beta minus
Beta plus
Gamma radiation can also be called …
Gamma rays
What does alpha radiation consist of?
Fast moving, positively charged alpha particles
Each alpha particle contains two protons and two neutrons (it is a helium nucleus)
What does beta minus radiation consist of?
Fast moving electrons
What does beta plus radiation consist of?
Fast moving positrons
What does gamma radiation consist of?
High energy photons with wavelengths less than ~10-13 m
What is the charge of an alpha particle?
+2e
What is the charge of a beta minus particle?
-e
Not the same as β+
What is the charge of a beta plus particle?
+e
Not the same as β-
What is the charge of a gamma ray?
0; they have no charge
What is the typical speed of alpha radiation?
~106 ms-1
What is the typical speed of beta radiation?
~108 ms-1
Same for both β- and β+
What is the typical speed of gamma radiation?
Speed of light
3.00 × 108 ms-1
What is the name of the unit u?
Unified atomic mass unit
What is 1u in kg?
1.661 × 10–27
What is the mass of alpha radiation in u?
4u
What is the mass of beta radiation in u?
1/2000 u
Same for both β- and β+
What is the mass of gamma radiation in u?
0
What is the range of alpha radiation in air?
Very short range - a few centimetres
What is the range of beta radiation in air?
a few metres
Same for both β- and β+
What is the range of gamma radiation in air?
Very long range
What is the penetration of alpha radiation?
Thin sheet of paper
What is the penetration of beta radiation?
4-5 mm of aluminium
Same for both β- and β+
What is the penetration of gamma radiation?
A few centimetres of lead
What force is responsible for beta decay?
Weak nuclear force
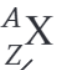
What is the nuclear decay equation for the alpha decay of the nucleus X?
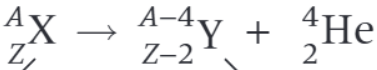
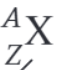
What is the nuclear decay equation for the beta minus decay of the nucleus X?
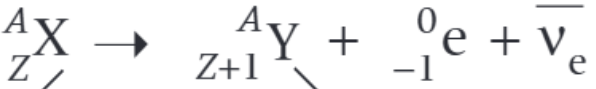
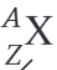
What is the nuclear decay equation for the beta plus decay of the nucleus X?
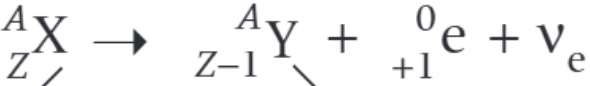
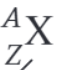
What is the nuclear decay equation for the gamma decay of the nucleus X?

What is the parent nucleus?
A nucleus before the occurence of radioactive decay
What is the daughter nucleus?
A new nucleus formed following a radioactive decay
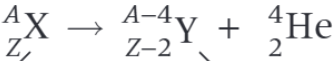
What is the parent nucleus in the decay equation shown?
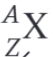
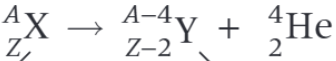
What is the daughter nucleus in the decay equation shown?
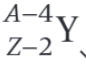
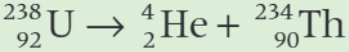
What is the parent nucleus in the decay equation shown?
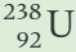
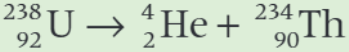
What is the daughter nucleus in the decay equation shown?
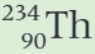
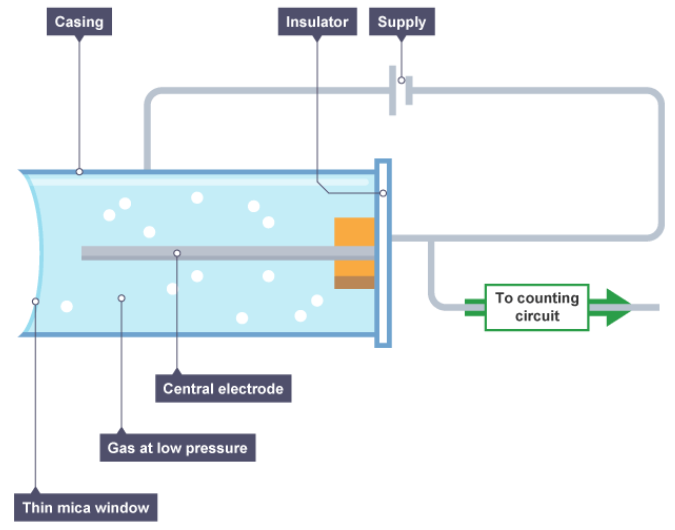
Draw a diagram of a Geiger-Müller tube
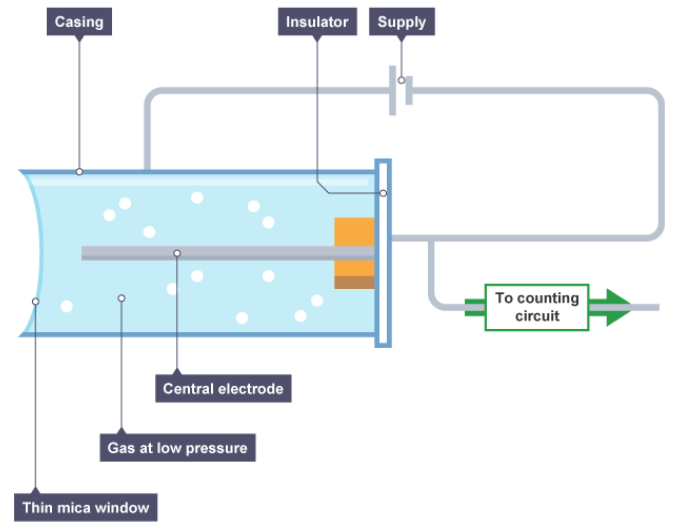
How is radiation detected by a Geiger-Müller tube?
When alpha, beta or gamma radiation enters the tube it produces ions in the gas.
The ions created in the gas enable the tube to conduct. A current is produced in the tube for a short time.
The current produces a voltage pulse. Each voltage pulse corresponds to one ionising radiation entering the GM tube.
The voltage pulse is amplified and counted.
Radioactive decay is a _____ and spontaneous event
Random
Radioactive decay is a random and _____ event
spontaneous
Radioactive decay is a _____ and _____ event
Random
Spontaneous
Why is radioactivity described as a random process?
We can’t predict when a particular nucleus in a sample will decay or which one will decay next
Each nucleus within a sample has the same chance of decaying per unit time
Why is radioactivity described as a spontaneous process?
Because the decay of nuclei is not affected by:
The presence of other nuclei in the sample
External factors such as pressure.
Define activity
The rate at which nuclei decay in a radioactive source
Define the Becquerel
One becquerel is an activity of one decay per second
What is the symbol for Becquerel?
Bq
What is the decay constant?
The probability of decay of an individual nucleus per unit time
What is the unit of the decay constant?
s-1 (NOT Bq)
What is the symbol for the decay constant?
λ
What is the relationship between activity and the decay constant? (Include meaning and units of each term)
A = λN
A is activity in Bq
λ is the decay constant in s-1
N is the number of undecayed nuclei
What is half-life?
The average time it takes for half the number of active nuclei in a sample of an isotope to decay
What is the corrected count (CCR)?
Measured count rate - background count rate
Draw a diagram of the apparatus used to measure the half-life of protactinium
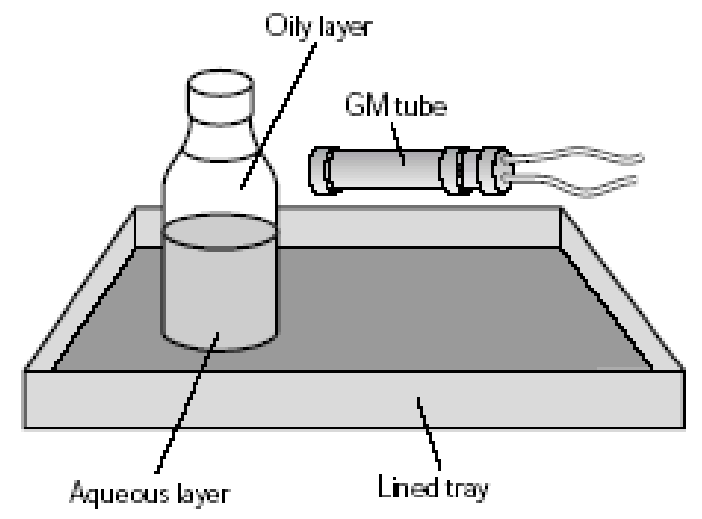
What equipment is used in the experiment to determine the half life of protactinium?
Geiger–Müller tube
Lined tray
Protactinium
Laptop
Clamp stand
Stopwatch
In the experiment to determine the half life of protactinium, how do we setup the plastic botle to get a fixed sample of protactinium?
Protactinium 234 is produced by the decay of 238U: 238U → 243Th by alpha decay, then 234Th → 234Pa by beta decay.
The 238U is in the form of uranyl nitrate dissolved in water and is contained in the sealed plastic bottle.
The bottle also contains an oily solvent that floats above the water.
When the bottle is shaken some of the 234Pa in the watery layer dissolves into the oily layer.
Once the two layers have separated out no more 234Pa moves into the oily layer, so we have a fixed sample of 234Pa in the oily layer.
In the experiment to determine the half life of protactinium, how can we measure only the radiation from the protactinium and not the radiation from the uranium or thorium?
The 238U emitts alpha radiation which is stopped by the plastic bottle, so wont reach the Geiger-muller tube.
234Pa emits energetic beta radiation, which can penetrate the plastic bottle and travel some distance in air and hence will reach the Geiger-muller tube
234Th also emitts beta radiation which can reach the Geiger-muller tube.
However, only protactinium dissolves in the oily layer. Since we point the Geiger-muller tube towards this oily layer (and not the aqueous solution), so we only measure the beta radiation from the 234Pa
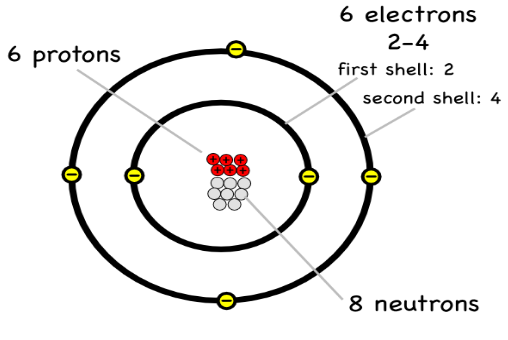
Describe the structure of carbon 14
6 protons
8 Neutrons
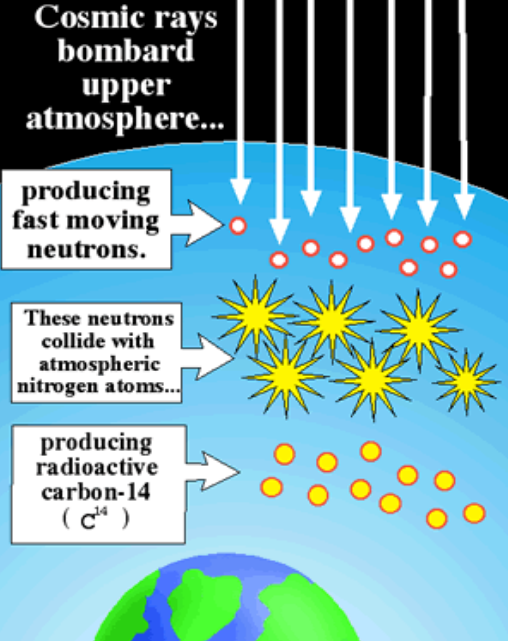
How is carbon 14 formed?
What sort of materials can be Carbon dated?
Organic matter
What are the decay products of Carbon-14 decay?
Nitrogen-14
Electron
Electron anti-neutrino
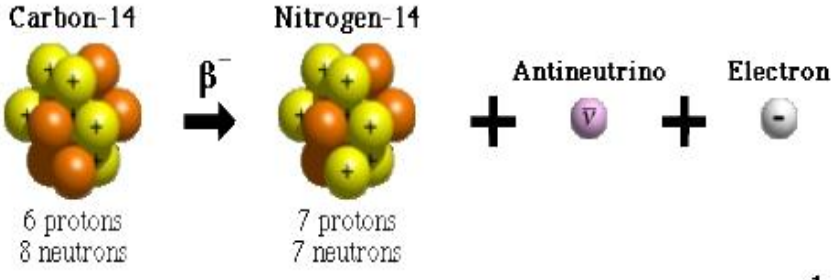
What type of radioactive decay does carbon-14 undergo?
Beta minus decay
What is the half life of Carbon-14?
5730 years
What are the limitations of the carbon dating process?
Can only date organic matter
Can only date matter less than 50,000 years old
Explain the method of Carbon dating
Living plants / animals absorb carbon-14.
Once dead, the plant does not take in any more carbon-14.
The number of C-14 nuclei is measured in dead and living sample.
x = x0e-λt used with data above to estimate the age.
What is an assumption made in carbon dating?
The ratio of carbon-14 to carbon-12 (in a living sample) has remained constant
What equation is used to find the activity after a period of time? What is the meaning and units of each term?
A = A0 e-λt
A is the activity at time t in Bq
A0 is the activity when t=0 in Bq
λ is the decay constant in s-1
t is time in seconds
What equation is used to find the number of undecayed nuclei after a period of time? What is the meaning and units of each term?
N = N0 e-λt
N is the number of undecayed nuclei at time t
N0 is the number of undecayed nuclei when t=0
λ is the decay constant in s-1
t is time in seconds
What equation links the decay constant and half-life?
λ t1/2 = ln(2)
How can you safely handle radioactive samples?
Keep as far from your body as possible - use tongs with long handles
Store samples in lead boxes when not in use
Never handle with your bare hands