AP Chem Electron and molecular geometries.
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
Pattern for Increasing Electron Geometries
Linear (1/diatomic)
Linear (2)
Trigonal Planer (3)
tetrahedral (4)
Trigonal bipyramid (5)
Octahedral(6)
Trigonal Planar Molecular Geometries as Lone Pairs increase
Trigonal Planar (0)
Bent (1)
Tetrahedral Molecular Geometries as Lone Pairs increase
Tetrahedral(0)
Trigonal Pyramid(1)
Bent(2)
Trigonal Bipyramid Molecular Geometries as Lone Pairs increase
Trigonal Bipyramid (0)
See-Saw(1)
T-Shaped(2)
Linear(3)
Octahedral Molecular Geometries as Lone Pairs increase
Octahedral(0)
Square pyramid(1)
Square Planar (2)
T-shaped(3)
Linear(4)
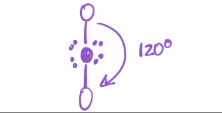
1 bond; 0 lone pairs
electron geometry — linear
molecular geometry — Linear
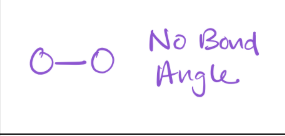
2 bonds; 0 lone pairs
electron geometry — linear
Molecular Geometry — Linear
*180 Degrees
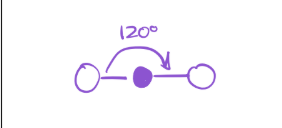
3 bonds; 0 lone pairs
electron geometry—trigonal planar
Molecular Geometry—trigonal planar
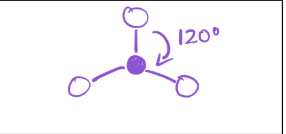
2 bonds; 1 lone pair
electron geometry—trigonal planar
molecular geometry—Bent
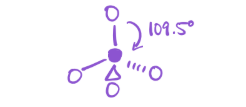
4 bonds; 0 lone pairs
electron geometry—tetrahedral
Molecular geometry—tetrahedral
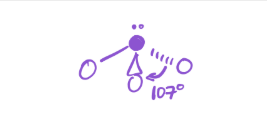
3 bonds; 1 lone pair
electron geometry—tetrahedral
molecular geometry—trigonal pyramidal
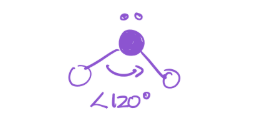
2 bonds; 2 lone pairs
EG—tetrahedral
MG—Bent
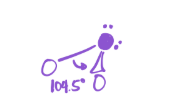
5 bonds; 0 lone pairs
EG—trigonal bipyramid
MG—Trigonal Bipyramid
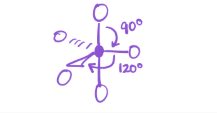
4 bonds; 1 lone pair
EG—trigonal bipyramidal
MG—See-saw
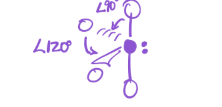
3 bonds; 2 lone pairs
EG—trigonal bipyramidal
MG—T-shaped
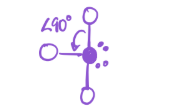
2 bonds; 3 lone pairs
EG—trigonal bipyramidal
MG—Linear
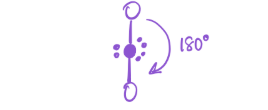
6 bonds; 0 lone pairs
EG—octahedral
MG—Octahedral
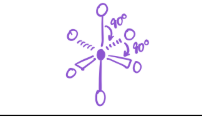
5 bonds; 1 lone pair
EG—octahedral
MG—Square Pyramid
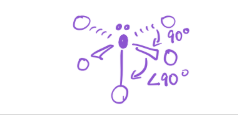
4 bonds; 2 lone pairs
EG—octahedral
MG—Square Planar

3 bonds; 3 lone pairs
EG—octahedral
MG—T-Shaped
*<90
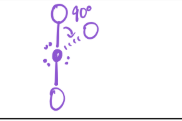
2 bonds; 4 lone pairs
EG—octahedral
MG—Linear