A&P Final (CH. 1-4)
1/150
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
151 Terms
anatomy
study of structure/form
physiology
study of function (of those structures)
levels of the hierarchy of life
“All Men Own Cats, They Often Own One, Paws Catch Every Bug.”
atom, molecule, organelle, cell, tissue, organ, organ system, organism, population, community, ecosystem, biosphere
atom example
molecule example
(hierarchy of life)
carbon atom, water molecule
organelle example
tissue example
organ example
(hierarchy of life)
ribosome, simple squamous epithelium, liver
About one of every thousand people is born with extra digits.
anatomical variation
Missing muscles, extra vertebrae, renal arteries
ex.) Most people have 5 lumbar vertebrae, but I was born with 6.
anatomical variation
Because of differences in enzyme function, children must be given a smaller dose of Tylenol.
Physiological Variation
Some hypothesize that differences in COVID-19 symptoms is due to difference in patient immune system function.
Physiological Variation
feeling structures inside the body by applying slight pressure with the fingers
Palpation
Uses X-rays (radiation) to produce a 3-dimentional image of soft tissues
CT Scan (Computer Topographic Scan)
X-rays =
bone structure
MRI =
soft tissue
Uses magnets to producing 3-dimentional images of soft tissue. Less damaging to healthy tissue
MRI (Magnetic Resonance Image)
magnetic field - uses increases in metabolic activity or blood flow to infer which areas of the body are associated with a function
function MRI (fMRI)
radiactive tracer - uses increases in metabolic activity or blood flow to infer which areas of the body are associated with a function
PET scan
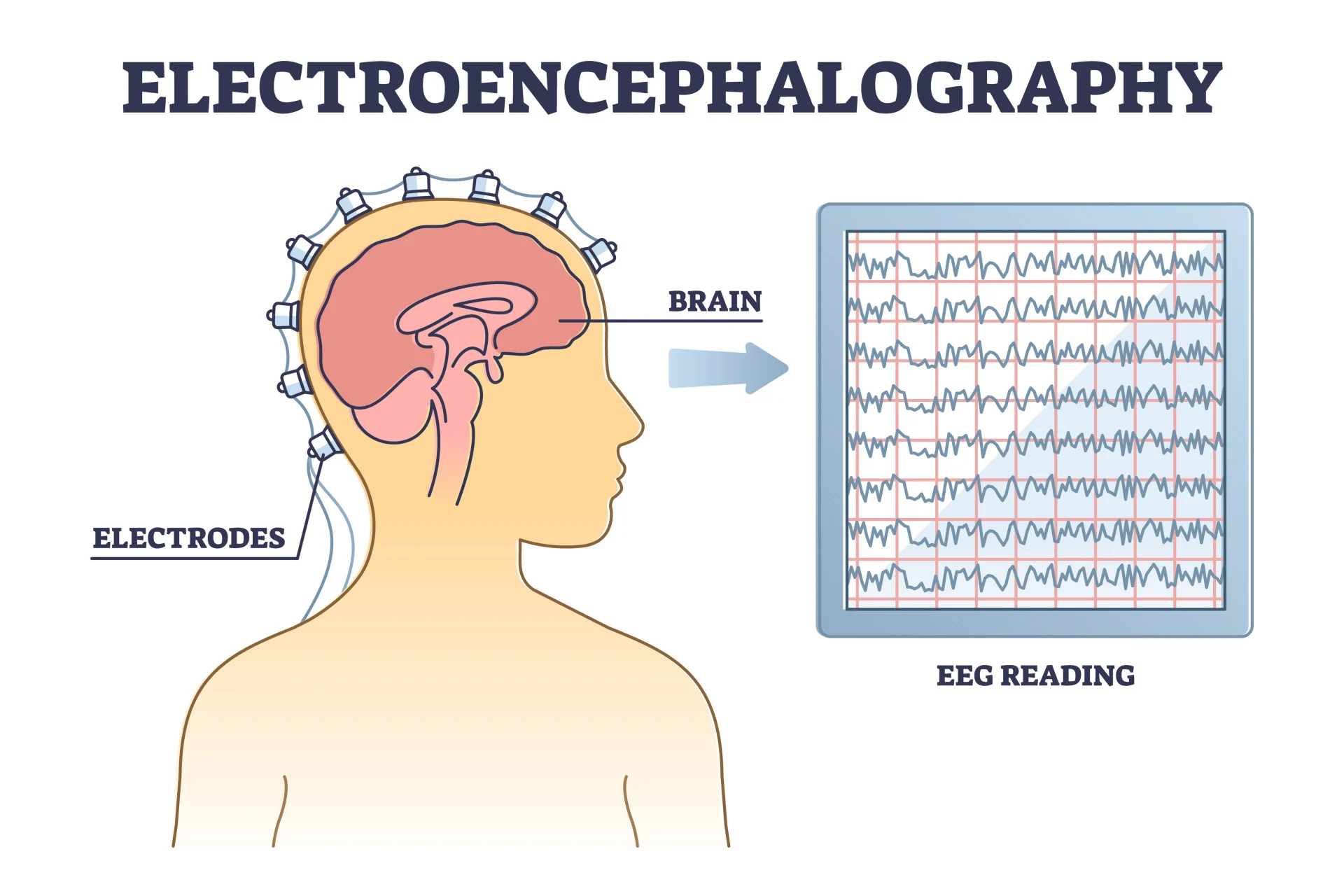
electroencephalogram =
brain activity
Observing the superficial appearance of the body
Inspection
Tapping the body and listening to the sounds produced in response to the tapping
Percussion
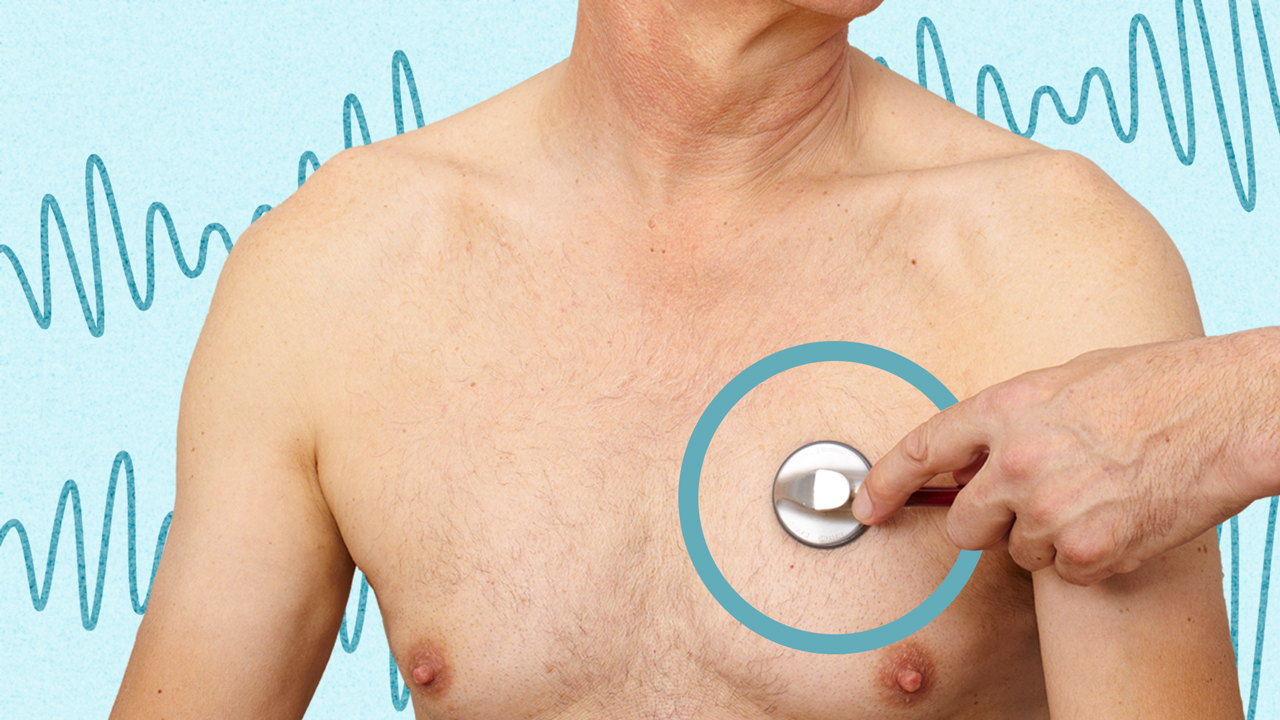
Using a stethoscope to listen to sounds inside the body (without tapping)
Auscultation
Making a small incision in the body to remove tissue for evaluation
Biopsy
Uses sound waves to produce images. Used to assess fetal development during pregnancy.
Sonography
the ability to maintain relatively stable internal conditions
homeostasis
what feedback loop opposes change + promotes a stable environment found inside our bodies?
negative feedback loop
When our body temperature rises, blood vessels in the skin dilate, Heat moves from high temperature towards the lower temperature (feedback loop)
Negative feedback loop
Can be used to maintain a homeostatic blood pressure
(feedback loop)
negative feedback loop
what does negative feedback loop help maintain?
homeostatic blood pressure, temperature
With enough change, an effector is triggered, By opposing the change, the effector pushes the factor back towards the set point
shivering when you have a fever
Negative feedback loop
The change that occurs results in more of the trigger that produced the change in the first place
→ rapid change in same direction
positive feedback loop
examples of positive feedback loop
childbirth, blood clotting, protein digestion, fever, and generation of nerve signals
Diffusion
Movement of molecules from an area of higher concentration to an area of lower concentration.
difference between 2 points or compartments, Difference in concentration of a molecule (chemical gradient), Others: temperature, pressure, charge differences (+/-)
Gradient
A downhill gradient, favors spontaneous movement, Movement of particles, down a gradient is called diffusion, In the heart, blood flows out, “down” a positive pressure gradient
positive gradient
An uphill gradient, the resting membrane potential of a cell, particularly a neuron or muscle cell. This is the condition where the interior of the cell has a negative electrical charge relative to the extracellular fluid surrounding it.
negative gradient
Eating carbs results in high glucose concentration in the lumen of the small intestine, Glucose diffuses out of lumen into cells
concentration gradient
With this type of gradient, movement will require additional energy.
Negative gradient (uphill)
Diffusion can only occur with this type of Gradient
Positive gradient
Cells keep Na+ concentration high on the outside & low on the inside, Inward flow of Na+ is driven by a concentration and a charge gradient
Electrochemical Gradient
Electrochemical gradient
Cells keep _____ concentration ____ on the outside & ___ on the inside, Inward flow of Na+ is driven by a ______ and a _____ gradient
Na+, high, low, concentration, charge
difference in pressure between two points
pressure gradient
Diffusion & Gradient:🧂💪🏼 → 🍌🧠
move _____ ions into a muscle cell and _____ ions out of a nerve cell, redirect blood by _______
Na+, K+, vasomotion
Protons (location, charge, mass)
Nucleus, +1 charge, 1 Dalton (amu)
Electrons (location, charge, mass)
around the nucleus, -1 charge, negligible
Neutrons (location, charge, mass)
Nucleus, 0 charge, 1 Dalton (amu)
6 most common elements in living things
(CHONy baby p.s)
Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorus, and Sulfur
Elements with a variant number of neutrons
More or fewer neutrons change the mass
Isotope
(Isotopes) more or fewer neutrons changes the ______
mass
Extra neutrons make isotopes of hydrogen _________
heavier
isotopes that are not stable
radioisotopes
charged particles with unequal numbers of protons and electrons
Ion
Ions have an unequal amount of what?
protons and electrons
salts in our body that dissociate to release ions
They can conduct an electric current & have important functions
Electrolytes
Imbalances of electrolytes can cause . . .
problems ranging from muscle cramps to heart failure
ions are important to ______, ______, and ______ physiology
bone (Calcium), nerve (sodium + potassium) and muscle (Magnesium and others)
opposites attract!
bond forms between oppositely charged ions, the force that holds them together is the electrostatic attraction
Ionic bond
form between atoms with partially filled shells by sharing electrons, shared electrons are the “force” that holds them together
Covalent bonds
electrons shared equally, no change
ex.) C-C, C-H, F-F
Non-polar covalent bonds
unequal sharing of electrons, charge is distributed unequally
ex.) C-O, C-N, H-O, P-O, Oxygen is more electronegative than C or H
Polar covalent bonds
weak attraction between a slightly positive region of a molecule and and slightly negative region of a molecule
ex.) protein, protein folding, H2O
hydrogen bond
molecules change shape or they “stick” to other molecules, does NOT form new molecules
hydrogen bond
resist changes in pH (maintain homeostasis), often weak acids
buffer
acidic pH
< pH7
Basic (alkaline) pH
> pH 7
substances that donate protons (H+) or accept electron pairs, tasting sour and having a pH below 7
acids
accept protons or donate electron pairs, often tasting bitter, feeling slippery, and having a pH above 7
bases
All of the chemical reactions that occur within an organism
metabolism
Metabolic pathways that break down molecules, releasing energy
Catabolic
Metabolic pathways that construct molecules, requiring energy.
Anabolic
List the 4 major types of organic molecules (polymers)
Carbohydrates(monosaccharides), Lipids, Proteins, Nucleic Acids
monomers for
Carbohydrates
Lipids
Proteins
Nucleic acid
monosaccharide ring
fatty acids + glycerol
amino acids
nucleotid
ribose(RNA) function
gene expression
glucose (carbohydrate monomer) function
provide quick energy
Lactose (Carbohydrate disaccharide) function
sugar in milk ( glucose + galactose )
Energy that is stable enough to transport
Glycogen (Carbohydrate polysaccharide) function
Long-term storage
Protein monomer function
monomer = amino acid
Some are neurotransmitters (nerve cell communication)
Polypeptide (Protein Polymer) Function
neurotransmitters and hormones
Long polypeptides (proteins) are the “workers of the cell”
the hydrophobic macromolecules of life
lipids
lipids are the group into which all the _________ ____________ of life are placed
hydrophobic macromolecules
amino acids are the _________ of proteins and there are _#_ different amino acids in living things
monomers, 20
extreme conformational change that destroys a protein’s shape & function
Denaturation
unique, 3-D shape of proteins, Often crucial to protein function
Conformation
a long, continuous chain of amino acids linked together by peptide bonds
polypeptide
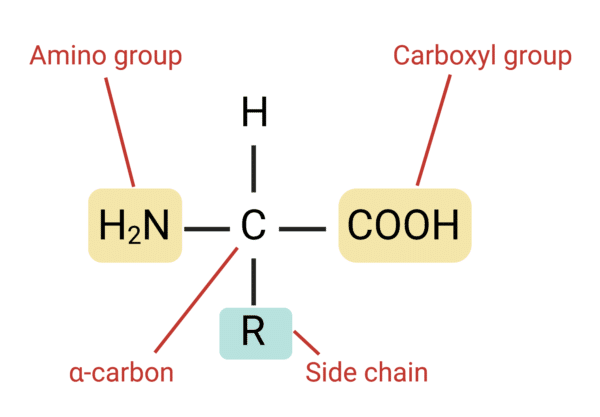
have a central carbon bonded to four components: an amino group, a carboxyl group, a hydrogen atom, and a variable side chain group
general structure of amino acid
7 Functions of protein
“Sal Can Make Real Movement Called Edge”
Structure - Keratin in skin, hair & nails; Collagen in bones, etc.
Communication - Receptors
Membrane transport - Channels and Carriers
Recognition and protection - Recognition of self versus foreign; antibodies, etc.
Movement - cilia & flagella (protein); motor proteins; contractile proteins
Cell adhesion - Bind cells to form tissue ; Immune docking
Enzymes - Protein catalyst (catalysts speed up chemical reactions)
speeds up chemical reactions in our cells, lowers activation energy
enzyme
is a protein catalyst
enzyme
the (kinetic) energy it takes to bring two molecules together in the correct orientation and weaken old bonds
activation energy
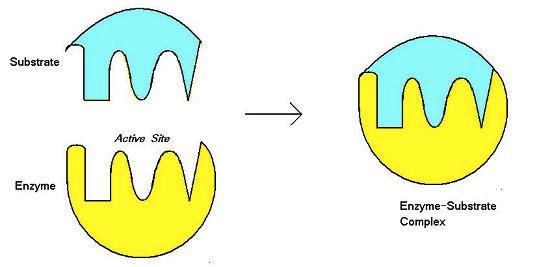
3 dimensional “pocket” on an enzyme, where the substrate fits
active site
The molecules that are brought together are called the ______ of the enzyme
substrate
monomers of nucleic acids
nucleotides (RNA and DNA)
ATP, DNA, and RNA are examples of _______
nucleic acids
the “energy currency” of the cell
ATP monomer
important nucleotide polymers
DNA and mRNA, tRNA & rRNA
nucleic acid that stores our genetic information
DNA
3 types of RNA names + function
Messenger RNA (mRNA), Transfer RNA (tRNA), and Ribosomal RNA (rRNA
Gene Expression: Using the information in a DNA gene, to make proteins at the right time and in the right amounts
A fluid oily film (mostly lipid) – Lipids, proteins and carbohydrates
plasma membrane
Solid substances secreted by cells, found in Extracellular fluid
Extracellular matrix
Extensions of the plasma membrane (NOT microtubules), Increase a cell’s surface area 15-40X, In cells specialized for absorption, Core is actin microfilaments
Microvilli
jelly-like, semi-fluid material enclosed within the cell membrane that fills cells and suspends organelles
cytoplasm
Phospholipids - polar head (phosphate group, glycerol)
hydrophilic