Bio Transport Final Exam
1/92
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
93 Terms
What is diffusion?
the net movement of solute particles from a region of higher concentration to a region of lower concentraation due to random molecular motion and collision.
What is Convection?
It is the transport of solute via bbulk fluid flow, where the moving fluid carries the solute along with it.
[Concept Quiz 1] Fixed coordinates allow us to quantify solute transport due to:
Both convection and diffusion
![<p>[Quiz 1] What does the first term represent (left to right)? </p>](https://assets.knowt.com/user-attachments/20fd63ae-4155-4426-ab1c-c9713a52763f.png)
[Quiz 1] What does the first term represent (left to right)?
it is the accumulation term, it represents the net change in qunaitity over time
![<p>[Quiz 1] What does the second term represent (left to right)?</p>](https://assets.knowt.com/user-attachments/d0eb2746-40ea-4faa-a2ab-0fdf4695da23.png)
[Quiz 1] What does the second term represent (left to right)?
is is the convection term, it represents the transport of solute via bulk flow of fluid.
![<p>[Quiz 1] What does the third term represent (left to right)?</p>](https://assets.knowt.com/user-attachments/4d56b962-3205-4172-a52e-454d8a10e4d4.png)
[Quiz 1] What does the third term represent (left to right)?
it is the diffusion term, it represents the movement of molecules due to random motion.
[Quiz 1] What does the last term represent (left to right)?
it is the reaction term, it represents the rate of solute generation or consumption.
![<p>[Quiz 1] What assumptions do we need to make for the mass transport equation (equation 9) to aquire the equation in the picture?</p>](https://assets.knowt.com/user-attachments/f3209cb2-8534-4301-b023-7d31ef0181ca.png)
[Quiz 1] What assumptions do we need to make for the mass transport equation (equation 9) to aquire the equation in the picture?
Assume no reactions
Assume no convection
Assume 1 dimensional
![<p>[Quiz 1] What assumptions do we need to make for the mass transport equation (equation 9) to aquire the equation in the picture?</p>](https://assets.knowt.com/user-attachments/05c562f0-7c4f-4fab-84dd-d183aaa18608.png)
[Quiz 1] What assumptions do we need to make for the mass transport equation (equation 9) to aquire the equation in the picture?
Assume no reactions
Assume no convection
Assume 1 Dimensional
Assume Steady State
[Quiz 1] What are the three fundamental physical quantities goverend by conservation laws that are transported within the body?
Mass
Momentum
Energy
[Quiz 1] molecules with smaller diffusion will exhibit _______ diffusion than molecules with larger diffusion coefficients
slower
[Quiz 1] What is flux?
net movement of a quantitiy though a unit of area per time.
[Quiz 1] What is the name fore equation 7 represent?
This is the flux of molecules due to diffusion.
[Quiz 1] How does increasing moleuclar size affect a molecules diffusion coefficient?
it will increase drag and therefore decrease the diffusion coefficient
[Quiz 1] How does increasing tempereature affect a molecules diffusion coefficient in solution?
it provides more thermal energy, increasing the diffusion coefficient.
[Quiz 1] How does decreasing temperature affect a molecules diffusion coefficeint in solutions?
decreasing temperatre reduces molecualr motion and decreases the diffusion coefficient.
[Quiz 1] how does increasing solvent viscosity impact a molecules diffusioon coefficient?
increasing solvent viscosity shows molecular motion and decreases the diffusion coefficient.
[Quiz 1] For a fixed concentration gradient, how does increasing the diffusion coefficient D affect the diffusive flux according to Ficks law?
For a fixed concentration gradient, increasing D increases the magnitude of the diffusive flux.
[Quiz 1] In a very viscous fluid at low temperature, a solute will ____ than in a less viscous, warmer fluid.
diffuse more slowly[
[Quiz 1] How does solvent viscosity impact a molecules diffusion coefficeint?
Increasing viscosity will decrease the diffusion coefficient.
[Quiz 1] Whaat type of coordinatae system cna be used to observe diffsuion?
Fixed and Moving coordinates
[Quiz 1] What type of coordinate system can be used to observe convection
only fixed.
![<p>[Quiz 1] What is the dilute solution assumption?</p>](https://assets.knowt.com/user-attachments/a124b426-34c3-4e20-bba1-daa6cf4a46d1.png)
[Quiz 1] What is the dilute solution assumption?
it assumes that all solutes in a solute have concentrations much smaller than the solvent concentration, so the solutes are negligible compared with the solvent.
[Quiz 1] What is the biary diffusion coefficient model? (D)
it assumes that solute only interacts with the solvent, and interactions between different solutes are negligible.
[Quiz 1] Under the dilute solution assumption, how is mixture velocity impacted?
Since all solutes have concentrations smaller than the solvent concentration, it barely contributes to the total momentum of the mixture. Therefore mixture veloicty = solvents motion
[Quiz 1] At what scale is diffusion good at?
at the nano to micro scale
[Quiz 1] At what scale is convection good at?
at larger scales
[Quiz 1] What is the peclet number?
it tells you if convection or diffusion is more important in a transport problem.
![<p>[Quiz 1] what is equation 6 on the final exam equation sheet?</p>](https://assets.knowt.com/user-attachments/990e67ae-10ba-4c7d-a956-2f1921b8b09b.png)
[Quiz 1] what is equation 6 on the final exam equation sheet?
it is the peclet number
[Quiz 1] What does a Pe > 1 mean?
it means convection dominates transport
[Quiz 1] what does a Pe < 1 mean?
it means diffusion dominates transport
[Quiz 1] what does Pe about equal to 1 mean?
both convection and diffusion are important
[Quiz 1] what are then units for the peclet number?
the peclet number is unitless.
[Quiz 1] Is mass a scalar, vector, or tensor?:
it is a scalar
[Quiz 1] is Density a scalar, vector, or tensor?
it is a scalar
[Quiz 1] is temperature a scalar, vector, or tensor?
it is a scalar
[Quiz 1] Is pressure (in a fluid) a scalar, vector, or tensor?
is a scalar
is displacement of a point a scalar, vector or tensor?
it is a vector
is velocity a scalar, vector, or tensor?
it is a vector
is force a scalar, vector, or tesnor?
it is a vector
is momentum a scalar, vector, or tensor?
it is a vector
is area of a surface a scalar, vector, or tensor
is it a scalar.
is strain a scalar, vector, or tensor?
it is a tensor
is stress a scalar, vector, or tensor?
it is a tensori
is the velocity gradient a scalar, vector, or tensor?
it is a tensor?
is the concentration gradient a scalar, vector, or tensor?
it is a tensor
[Quiz 1] what does σij notation mean?
i = face
j = direction
[quiz 1] what is flux?
it is the measure of how much stuff moves through a cross sectional area over time.
[Quiz 2] what is the schmidt number?
it compares how faast momentum spreads ina fluid to how fast mass spreads.
[Quiz 2] Does schmidt number have any units?
it is a unitless number
[Quiz 2] what does a Scmidt number > 1 mean?
momentum diffuses faster than mass
[Quiz 2] what does a schmidt number < 1 mean?
mass diffuses faster than momentum
[Quiz 2] What does a schmidt number = 1 mean?
mass and monetum spread at the same rate.
[Quiz 1] In which coordinate system is the diffusive flux most naturally defined?
it it defined in the moving coordinate frame
[Quiz 1]what does a fixed coordinate system represent in transport problems?
it is a frame where you watch fluid flow past you
[Quiz 1]what does a moving coordinat4e system represent in trasnport problems?
it represents a frame where you move with the fluid .
[Quiz 1] In moving coordinates, the total flux of solutes can be written as the sum of which contributions?
contributions from diffusion, electircla field, pressure gradients, and thermal gradients.
[Quiz 2] What is elecroneutrality?
it states that a solution must be electically neutraal, therefore the sum of charges must be zero.
![<p>[Quiz 2] what does equation 11 represent in the final exam equation sheet?</p>](https://assets.knowt.com/user-attachments/2bbc9f7c-2147-4c42-8904-5f5d17b79ee7.png)
[Quiz 2] what does equation 11 represent in the final exam equation sheet?
The stokes einstein equation
[Quiz 2] what is stokes einstein equation?
it states that a particles diffusion coefficient is determined bby its shape, thermal energy, and how viscous the fluid is.
[Quiz 2] What is equation 12 on the final exam equation sheet?
it is the mean frictional drag coefficient
[Quiz 2] what units does temperature need to be for the stokes einstiens equation?
it needs to be in kelvin
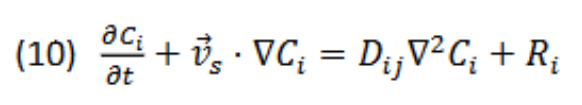
What are the units for each term term (left to right)?
Mol/(m³ * s)
![<p>[Quiz 2 - Lecture 9] what is the flux due to an electic field dependent on?</p>](https://assets.knowt.com/user-attachments/6294107a-1eec-4ad4-9b05-485e69215ed6.png)
[Quiz 2 - Lecture 9] what is the flux due to an electic field dependent on?
concentration of ions and the velocity of ions.
[quiz 2 - Lecture 9] What is the velocity of ion migration due to electrical feild dependent on?
Forces exerted per mole due to electical field
avagadros number
the shape / drag of the molecule.
[Quiz 2 - Lecture 9] What is flux due to an electical field for ions?
it is when an electical potential gradient pushing charged solutes through the fluid.
[Quiz 2 - Lecture 9] How does ion charage sign affect the direction of electical flux?
positive ions move opposite to the electical potential
negative ions move in the same direction as the electical potential.
[Quiz 9 - Lecture 9] Why is there a negative sign in the electical flux term?
to ensure that ions move downhill in electical potential
[Quiz 9 - Lecture 9] What is a cation?
a positively chared molecule
[Quiz 2 - Lecture 9] what is an anion?
a negatively charged molecule.
![<p>[Quiz 2 - Lecture 9] how does a large diffusion coefficient affect flux due to an electic field? if temperature was constant. </p>](https://assets.knowt.com/user-attachments/4a5d6de1-0387-417c-8f68-aef137a53a5a.png)
[Quiz 2 - Lecture 9] how does a large diffusion coefficient affect flux due to an electic field? if temperature was constant.
it will increase the electical feild flux
![<p>[Quiz 2 - Lecture 9] how does a small diffusion coefficient affect flux due to an electric field? if temperatue was constant</p>](https://assets.knowt.com/user-attachments/dc01f3fc-0737-414a-b051-fa0a18afe790.png)
[Quiz 2 - Lecture 9] how does a small diffusion coefficient affect flux due to an electric field? if temperatue was constant
it will increase the electical feild flux
![<p>[Quiz 2 - Lecture 9] How is flux due to an electical field affected by higher temperatures? assume diffusion is constant.</p>](https://assets.knowt.com/user-attachments/593d7746-45c8-4187-9b91-a6f4ad73fef8.png)
[Quiz 2 - Lecture 9] How is flux due to an electical field affected by higher temperatures? assume diffusion is constant.
it will decrease the electical feild flux
[Quiz 2 - Lecture 9] How is flux due to an electical field affected by lower temperatures? assume diffusion is constant.
it will increase the electical feild flux[
[Quiz 2 - Lecture 9] what is the definition of current density?
it is the sum of ion fluxes. [
[Quiz 2 - Lecture 9] what are the units for current density?
A/m²
[Quiz 2 - Lecture 9] what is equation 13 on the final exam equation sheet?
it is the flux due to an electical field
[Quiz 2 - Lecture 9 & 10] Can a solution be electroneutral and still have nonzero current density?
Yes because ions can move in oppositve direction and create flowing charge.
[Quiz 2 - Lecture 9 & 10] for an uncharged membrane with no applied voltage, what assumption must we make about the membrane?
it has no net current across the membrane.
[Quiz 2 - Lecture 9 & 10] What drives ions transport across an uncharged membrane?
Diffusion due to concentration gradients and electical potential gradient.
[Quiz 2 - Lecture 9 & 10] What assumptions must we make for a uncharged membrane?
electoneutrality
no net current
[Quiz 2 - Lecture 9 & 10] If a positive ion diffuses faster than a negative ion, what happens?
an electical potential gradient forms to equaliz the fluxes
[Quiz 2 - Lecture 9 & 10] If a negative ion diffuses faster than a positive ion, what happens?
an electical potential gradient forms to equaliz the fluxes
[Quiz 2 - Lecture 9 & 10] If a positive ion diffuses faster than a negative ion, what is the sign for the potential gradient?
it will be positive
[Quiz 2 - Lecture 9 & 10] If a negative ion diffuses faster than a positive ion, what sign is the potential gradient?
it will be negative.
[Quiz 2 - Lecture 10] What would happen to elecical potnetial gradient if temperatures were to increase?
the gradient will increase
[Quiz 2 - Lecture 10] What would happen to elecical potnetial gradient if temperatures were to decrease?
the gradient will decrease.
[Quiz 2] Consider transport of a binary electrolyte though an uncharged potential arises across the membrane primarly due to:
Differences in the ion concentration on either side of the membrane.
[Quiz 2] for electolyte transport across an uncharged membrane, if the cation (+) has a larger diffusion coefficient thatn the anion (-), which direction will the electical potential gradient point?
opposite the direction of the concentration gradient
[Quiz 2] For electrolyte trasnport across an unchargd membrane where the cation (+) has a larger diffusion coefficient than the anion (-), what is true about the effects of the electical potetial?
The electical potential acts to decrease the cation flux and increase the anion flux
[Quiz 2 - Lecture 9 & 10] what is the definition of Nernst Plank equation?
it says the flux of an ion is the sum of diffusion, electical migration, and convection.
![<p>[Quiz 2 - Lectur 9] How do we derive the Nernst plank equation from the final exam equation sheet/</p>](https://assets.knowt.com/user-attachments/b550a761-9308-4670-b05a-b3c83ddf4e4a.png)
[Quiz 2 - Lectur 9] How do we derive the Nernst plank equation from the final exam equation sheet/
substitute equtiaon 7 into 8 and add the combined equation into 13.
[Quiz 2 - Lecture 9] What terms are in the Nerrnst Planck equation?
diffusion gradient
electromigration
convection