Physics- The Nucleus
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
What is meant by nuclear fission?
Nuclear fission is the splitting of a large nucleus into two smaller nuclei, of roughly equal mass, with emission of neutrons and release of energy.
Describe the Bohr model of the atom.
Niels Bohr presented a model of atomic structure whereby the nucleus was made of tiny solid protons and neutrons packed together tightly in the nucleus. This was surrounded by electrons spinning in fixed orbits around the nucleus. The radius of the orbit corresponded to an energy level, with the smallest radius being the lowest energy level, and the energy
Name an instrument used to detect radioactivity. What is the principle of operation of this instrument?
Geiger muller tube- ionisation of gas
solid state detector - formation of ion-pairs
Name the naturally occurring radioactive gas which seeps into buildings from underground rocks and which can cause lung cancer
Radon
What is the role of neutrons in a nuclear reactor?
to sustain nuclear fission reactions
Give one benefit of switching from fossil fuels to nuclear power for the generation of electricity. Explain your answer.
less carbon dioxide produced- fossil fuels produce CO2 when burned
production of useful radioisotopes- strontium-90 and Cesium-137 are used in industry as gauges that detect the flow rates and to measure the thickness of materials, and medically to treat certain types of cancer
more energy per Kg- they produuce vast amounts of energy per gram of fuel used
Describe Rutherford’s model of the atom
Atoms are made up of negatively charged electrons orbiting a positively charged nucleus. They are mostly empty.
What is the function of the moderator in a fission reactor?
to slow down fast neutrons in order to facilitate fission, this ensures chain reaction continues.
In terms of how they interact with the neutrons in a fission reactor, distinguish between a moderator and a control rod.
The function of the moderator is to slow down fast neutrons in order to facilitate fission, this ensures chain reaction continues. The function of the control rods is to absorb neutrons and thus control the rate of the chain reaction, by having the rods shallower or deeper into the core.
Describe how the Bohr model of the atom explains emission line spectra.
The frequency of the emitted light depends on the change in energy levels (or states). When energy is supplied to the atom, electrons move to a higher (excited) energy level. This is unstable, so the electron falls to the lower level. The change in energy level equals the energy of the emitted photon, which is equal to plank’s constant multiplied by frequency; ℎ𝑓 =𝐸2−𝐸2.
Explain what is meant by a chain reaction in nuclear fission.
Chain reactions are reactions where at least one neutron released during each fission reaction. The neutrons released cause more fission when they are absorbed by another nucleus. It is a ‘self-sustaining’ reaction.
what is radioactivity
Radioactivity is the spontaneous emission of radiation due to the natural disintegration of an unstable nucleus.
give an application of (i) radioactivity (ii) fission
(i) smoke detectors, carbon dating
(ii) bombs, generating electrical energy
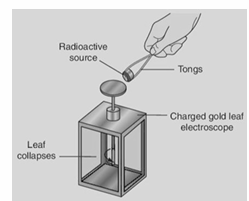
Describe an experiment to demonstrate the ionising effect of radioactivity.
Apparatus – Radioactive source, gold leaf electroscope Procedure – • Charge the electroscope (by induction). The leaf will diverge. • Bring radioactive source close to the cap. Observation– Leaf collapses much faster in the presence of the radioactive material than normal. Conclusion – As charge leaks away through ionised air the electroscope becomes neutralised. The radioactive source is ionising the air.
what is nuclear fussion
Nuclear fusion is the combining of two small nuclei to form a single larger nucleus, with the release of energy.
Why are large temperatures required for fusion to occur?
Nuclei are positively charged, so there is a strong repulsive force that must be overcome. This requires a large amount of energy.
Give one benefit of a terrestrial fusion reactor under each of the following headings: (i) fuel. (ii) energy; (iii) pollution.
(i) the raw fuel is cheap, plentiful and readily available
(ii) Vast amounts of energy released, more energy per gram than from other sources
(iii) little or no hazardous waste and no greenhouse gases
Describe how an emission line spectrum is produced.
When energy is given to the atoms of the gas, electrons to jump from their rest (lower) energy state to excited (higher) energy state. When the electrons return to the lower states they emit a fixed ‘quantum’ of energy (ℎ𝑓). This quantum of energy can be considered a photon of light or an electromagnetic wave of a specific frequency.
Explain, in terms of movement of electrons, why light is emitted when a metal is heated.
When a metal is heated to a sufficiently high temperature it emits visible light. This is because the electrons are gaining energy as heat is added to the metal. This causes the electrons to become excited and jump to higher energy states. When they return to the lower state they emit the ‘difference in energy’ as electromagnetic radiation, i.e. light. When the metal is sufficiently hot, such as with the tungsten in a filament bulb, all frequencies of light are present, and the metal becomes white hot. When heated to lower temperatures only the lower frequencies may be present. For example, the coil of a toaster emits red light.
What are the advantages of fusion over fission in terms of fuel sources and reaction products?
The main advantages of fission over fusion is that the hydrogen fuel is plentiful (in the sea) whereas uranium for fission is scarce. Also, there is no radioactive waste with fusion.
Define the becquerel.
1 becquerel is 1 radioactive disintegration per second.
compare alpha beta and gamma emissions using thhe following headings
(a) penetrating ability
(b) deflection in a magnetic field
(a) alpha-poor beta-medium gamma-good
(b) alpha-slight bea-considerable gamma-none
State two differences between the electromagnetic radiation emitted from a laser and the electromagnetic radiation emitted from a vapour lamp.
A laser emits light with a very narrow spectrum, i.e. they emit only one very specific colour, whereas vapour lamps usually emit a mix of colours.
The light from lasers is also focussed into a very narrow beam, which stays focussed over a very large distance. With vapour lamps, the light tends to spread out in all directions.
A substance called a moderator is mixed with the fuel in a nuclear reactor. Control rods are used to control the rate of the reaction. Give an example of a moderator.
Graphite or heavy water can be used as a moderator in a fission reactor.
Explain (i) why a moderator is needed in a nuclear reactor and (ii) how the control rods affect the rate of the reaction.
(i) The function of the moderator is to slow down fast neutrons in order to facilitate fission, this ensures chain reaction continues.
(ii) The function of the control rods is to absorb neutrons and thus control the rate of the chain reaction, by having the rods shallower or deeper into the core.
A heat exchanger is used in a nuclear reactor. Explain how the heat exchanger operates. Why is it necessary to use a heat exchanger?
Its function is to transfers heat (energy) from the core to the turbines used to generate electricity. It heats water, turning it to steam, which drives the turbine. They are necessary because the material in a reactor is radioactive and the heat exchanger allows the core to reach a higher temperature.
How many of the neutrons emitted in a fission reaction must, on average, cause a further fission so that the reaction is self-sustaining and safe?
For a controlled, safe self-sustaining chain reaction, you need on average one neutron to cause further fission. If the average is greater than one, the reaction is uncontrolled, if it’s less than 1, the reaction will end.
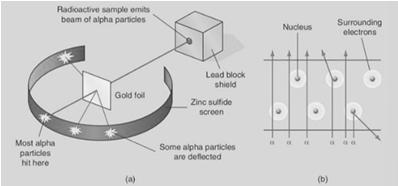
Describe Rutherford’s experiment to investigate the structure of the atom.
Apparatus – Radioactive material (alpha emitter), lead box containing a small opening, thin sheet of gold foil, vacuum, zinc sulphide screen. Procedure – • An alpha emitter is placed in the lead box containing a small opening. • Set up apparatus within a vacuum as shown in diagram. • Allow time for alpha particles to travel to gold foil. • Take note of the positions where the alpha particles hit the zinc sulphide screen, as they cause scintillations (flashes of light) on the screen. Observation– The majority of the alpha particle pass through the gold foil without deviation, others are deflected through varying angles, from small angles up to 180°.
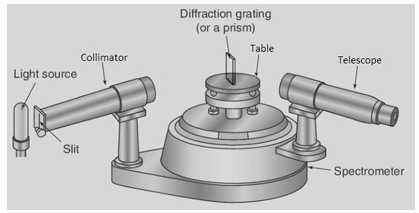
Draw a labelled diagram of a spectrometer and describe how a spectrometer and diffraction grating can be used to observe (i) a line spectrum and (ii) a continuous spectrum.
Apparatus – Spectrometer, mercury vapour lamp, filament bulb, diffraction grating. Procedure – • Shine the mercury vapour lamp through grating. • View the light through the telescope, rotate telescope. (18) • Repeat the process with the filament bulb. Observation– When viewing the light from the mercury vapour lamp, there is a series of bright lines of different colours, a line spectrum. • When viewing the light from the filament bulb, there is a continuous spectrum visible Conclusion – Mercury vapour emits only specific wavelength (colours) of light, producing a line spectrum, while the filament bulb produces a continuous spectrum.
Give one other advantage that a fusion reactor would have over a fission reactor.
Huge amounts of energy are needed to reach the temperature required overcome the repulsive electrostatic force between the nuclei to start the reaction. The main advantages of fission over fusion is that the hydrogen fuel is plentiful (in the sea) whereas uranium for fission is scarce. Also, there is no radioactive waste with fusion.
How can the build‐up of radon in a building be reduced?
One can guard against the build-up of radon in the home by installing a radon membrane under the building, sealing cracks and gaps under the floor or having good ventilation.
A G‐M tube and a solid‐state detector have the same function. What is this function?
Their purpose is to detect (ionising) radiation
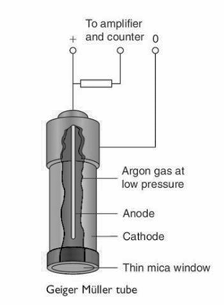
Describe, with the aid of a labelled diagram, the principle of operation of a Geiger muller tube
The G-M tube is based on the principle that radiation ionises a gas contained in a tube and the number of ionisations can be counted as current flows (via ions in the gas) from (central wire) anode to (surrounding) cathode.
what is meant by ionisation
the charging of a neutral atom when it loses or gains one or more electrons
Why are alpha particles not considered a health hazard?
Alpha emitters are not very penetrating, resulting in a short range.
Why was it necessary to carry out the gold foil experiment in a vacuum?
The experiment required a vacuum to prevent alpha particles colliding with other particles and being scattered, and because the range of alpha particles in air is very short.
explain the term half life
the time taken for half of the nuclei in a given sample to decay
In a nuclear fission reactor, neutrons are slowed down after being emitted. Why are the neutrons slowed down?
The speed at which the neutrons strike the large nucleus affects the likelihood of each outcome. Slow neutron may cause fission whereas fast neutrons undergo radiative capture.
give one positive and negative environmental impact of fission reactors
positive- less carbon dioxide produced, no greenhouse gases, less dependance on fossil fuels
negative- radioactive waste , potentiol for majjor accidents
How are the alpha particles produced?
Americium is radioactive and disintegrates, undergoing α-decay.
Explain why americium-241 does not exist naturally
Americium-241 has half life is short (with respect to the age of universe). It is not a member of a decay series.