Chem Paper 2 Exam Qs
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
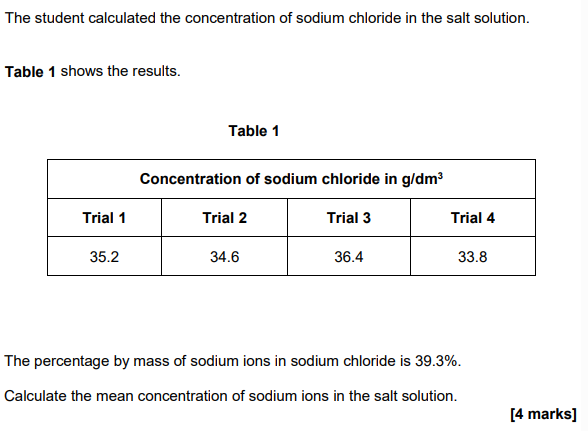
2024: (4)

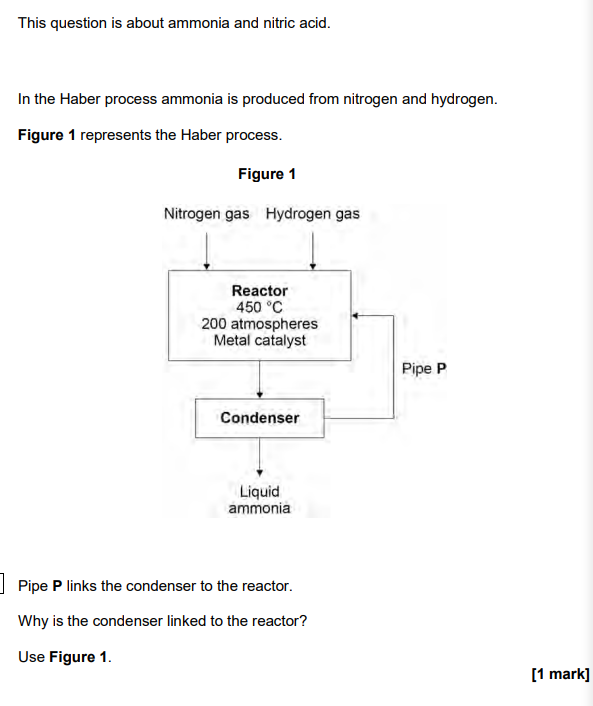
2024
to recycle remaining nitrogen and hydrogen
2024: Ammonia and nitric acid react to produce the salt, ammonium nitrate. Ammonium ions and nitrate ions both contain nitrogen.
Suggest one use of ammonium nitrate. (1)
fertilisers

2024: (2)
reversible, combustion
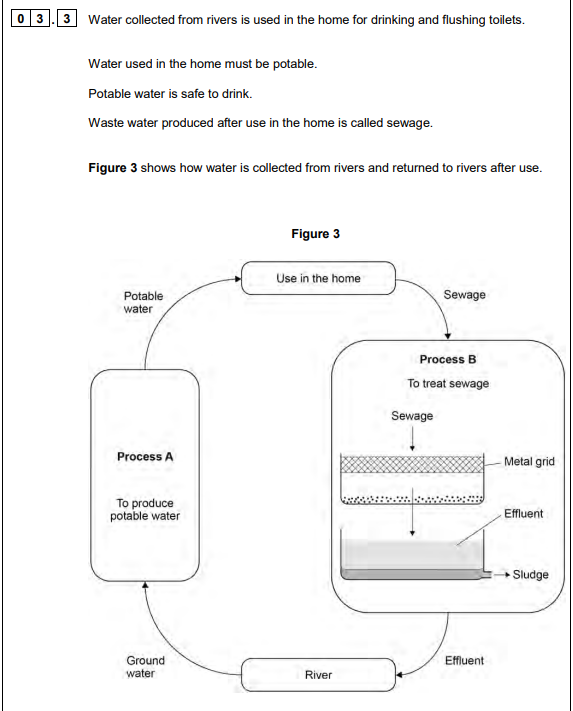
2024: (6)

2024: Why is the Rf value of a dye not affected by how far the solvent front is allowed to travel? (1)
the ratio/proportion of spot distance to solvent distance is fixed/constant
2024: Advantages of instrumental methods (2)
more sensitive
more accurate
faster
2024: Explain how oxides of nitrogen are produced when burning fuels (2)
nitrogen and oxygen (from air) react
at high temperatures
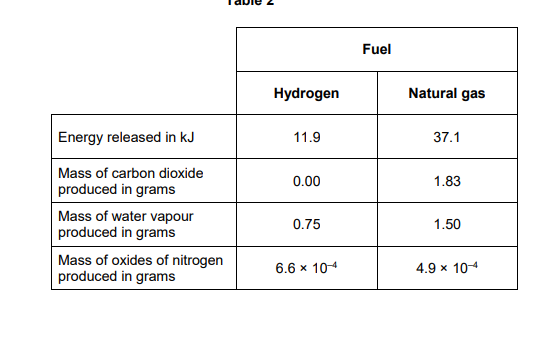
2024: Explain one positive impact on the environment of burning hydrogen rather than natural gas as a fuel (2)
Use table 2
less climate change/global warming
because no carbon dioxide produced
Effects of oxides of nitrogen
acid rain
respiratory problems

2024:
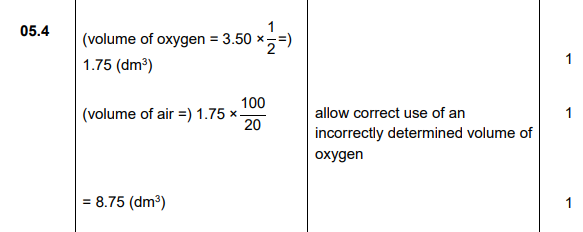

2024 (3)
Are metals finite or renewable resources?
finite
2024: Explain why aluminium alloy bicycle frames do not need protection from corrosion. (2)
aluminium has an oxide coating
so contact between aluminium and water/air/oxygen is prevented
2024: Suggest how bicycle chains made from an alloy of iron can be protected from rusting without using paint (1)
any one from:
coating with grease
coating with oil
galvanise
2024: To produce sulfur trioxide, a reversible reaction is used
To favour the products, a higher pressure should be used
In industry, the reaction is done at 450 °C and atmospheric pressure.
Under these conditions the yield of sulfur trioxide is 86%.
Suggest two reasons why a higher pressure is not used. (2)


2024 (1)
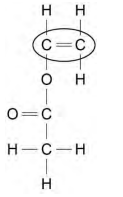
2024: Describe what will be seen when an alkene is shaken with bromine water (2)
orange bromine water
turns colourless
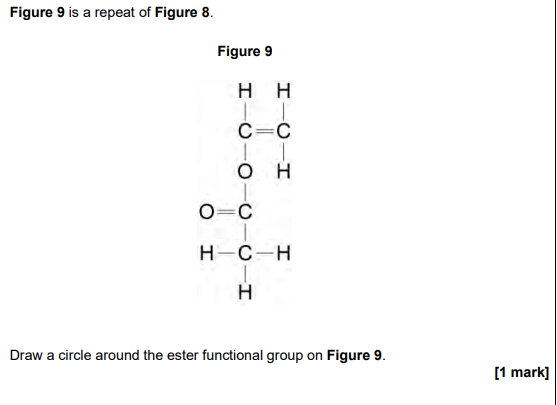
2024
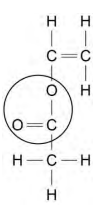
2024: Explain why some polymers do not melt when heated. (2)
(thermosetting polymers)
cross-links between polymer chains
so too much energy needed to overcome the cross-links
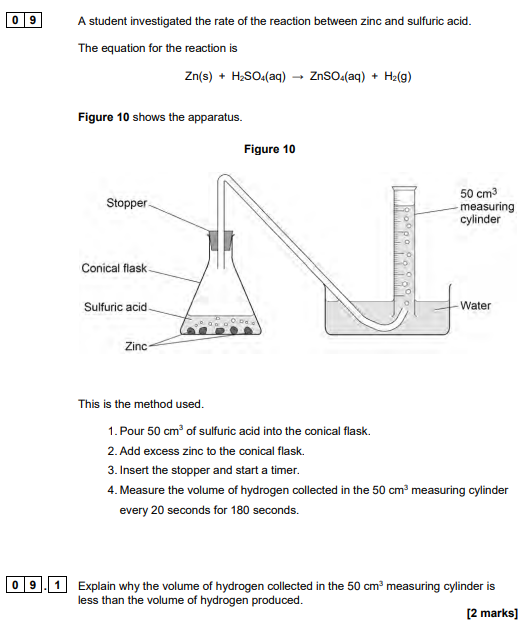
2024
some hydrogen/gas escapes
before the stopper is put in
2023: What is limewater?
an aqueous solution of calcium hydroxide
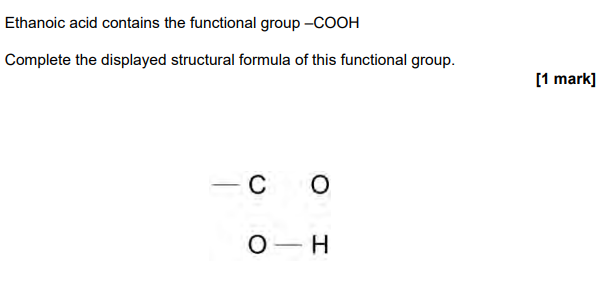
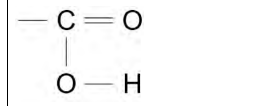
2023

2023 (2)
Ethanol = ethyl ethanoate
Sodium carbonate = carbon dioxide

2023 (6)

2023: Advantages of flame emission spectroscopy
more accurate
more sensitive
faster
can determine concentration of ions present
2023: In the past 50 years, there has been an increase in:
• the world population
• the concentration of carbon dioxide in the atmosphere
• the concentration of methane in the atmosphere
• the mean temperature of the atmosphere at the Earth’s surface.
Most scientists think this information can be used to explain climate change.
Explain why the increase in world population may have caused the increase in the concentration of carbon dioxide in the atmosphere (2)

2023: Explain why the increase in world population may have caused the increase in the concentration of methane in the atmosphere (2)

2023: The mean temperature of the atmosphere at the Earth’s surface has increased.
Most scientists think that this has been caused by an increase in the concentration of greenhouse gases in the atmosphere.
Give one reason why some scientists do not accept this theory (1)

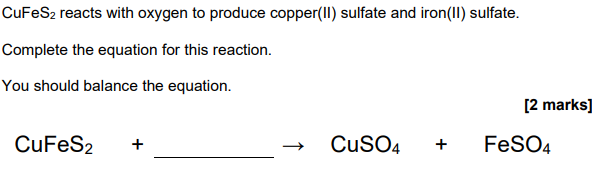
4O2
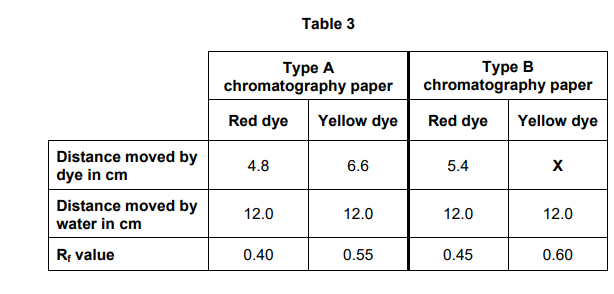
2023: Explain why the Rf values for the red dye are different using the two types of chromatography paper. (3)

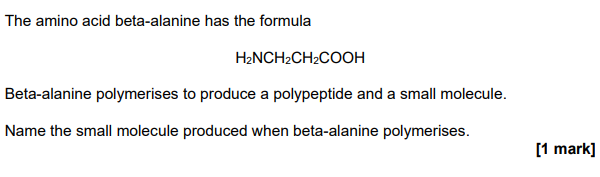
2023
water
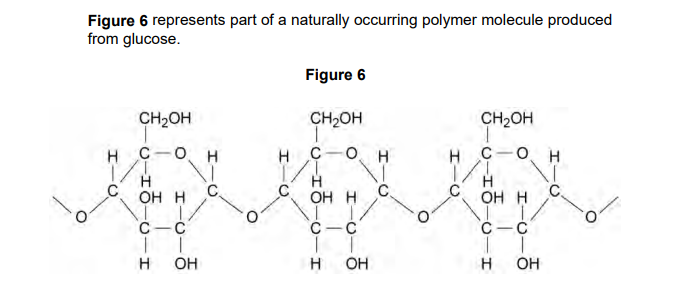
2023: Suggest the identity of this polymer (1)
any one from:
starch
cellulose
glycogen
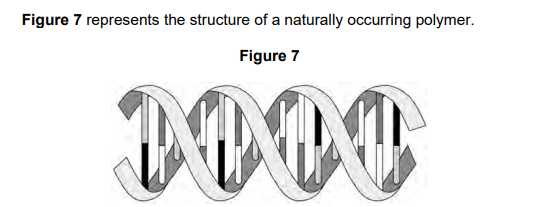
2023: Give the general name for the four different monomers which make up the structure shown (1)
nucleotides

2023: Nitric acid is needed to produce ammonium nitrate. Name a compound needed to produce nitric acid (1)
ammonia
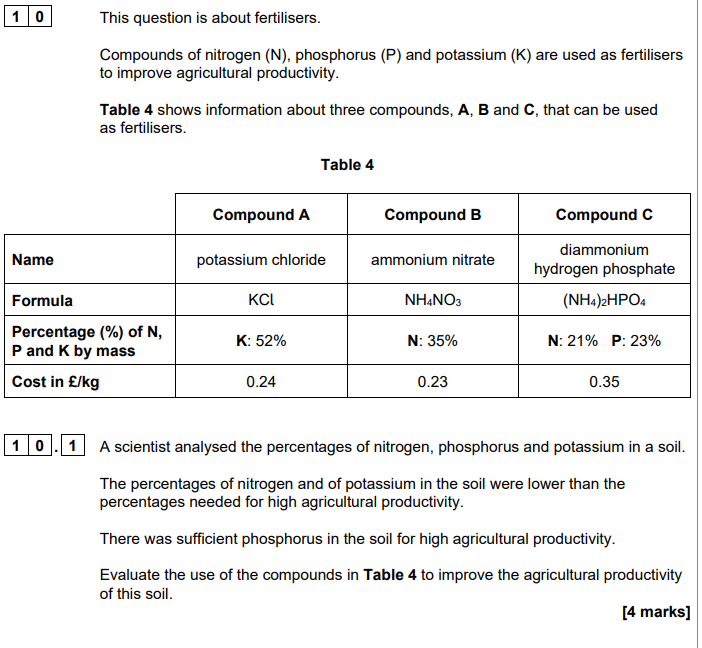
2023: Phosphate rock contains phosphorus compounds. Plants absorb phosphorus from compounds dissolved in rainwater.
Suggest why phosphate rock cannot be used directly as a fertiliser (1)
insoluble in water

2023

2022: Suggest two reasons why recycling scrap copper is more sustainable than extracting copper from copper ores (2)
any two from:
(recycling copper ores)
uses less energy
conserves copper (ore)
(produces) less waste
specified environmental impact
less landfill required
2022: A solution can be added to copper sulfate solution to show the presence of copper(II) ions.
Name the solution added. Give the result of the test. (2)
sodium hydroxide
blue precipitate
2022: Describe one test to show the presence of sulfate ions in copper sulfate solution. Give the result of the test. (2)


2022: Explain why the mass of the test tube and contents decreased (2)

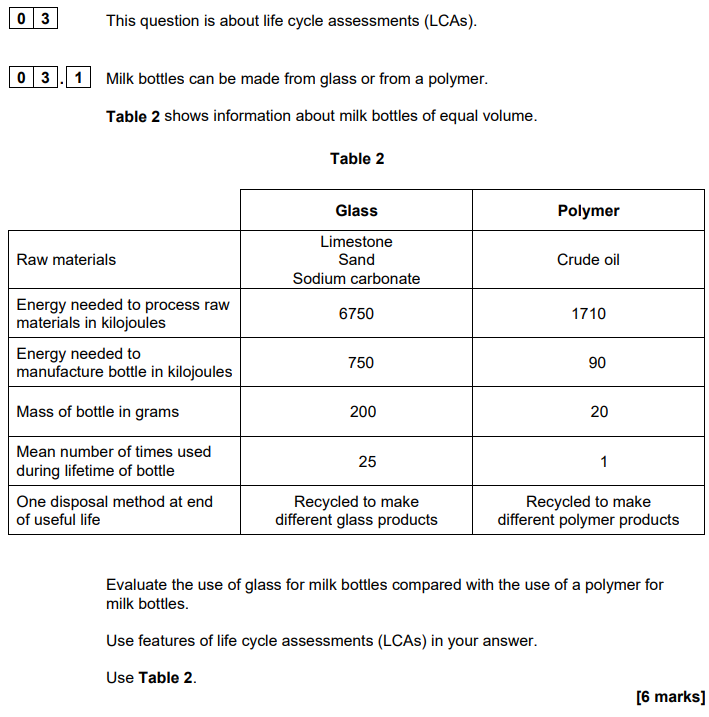

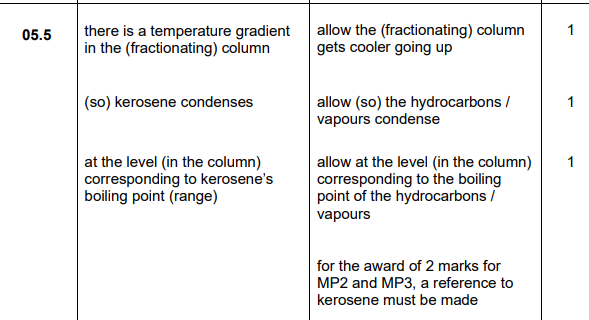
2022: Explain how fractions are obtained from crude oil by fractional distillation (4)


2022
detergents, solvents
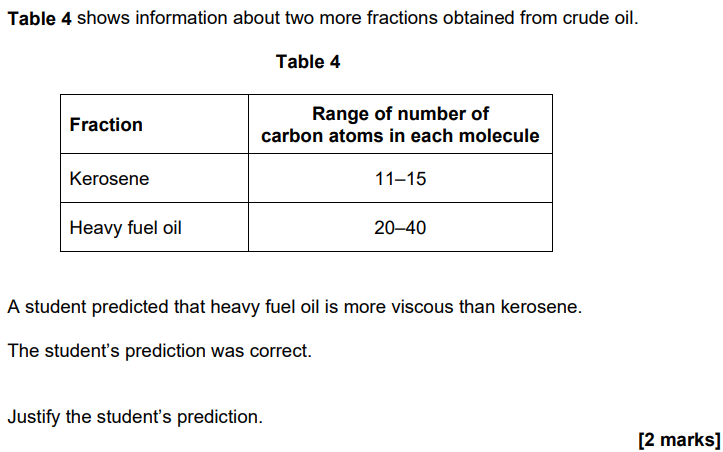
2022

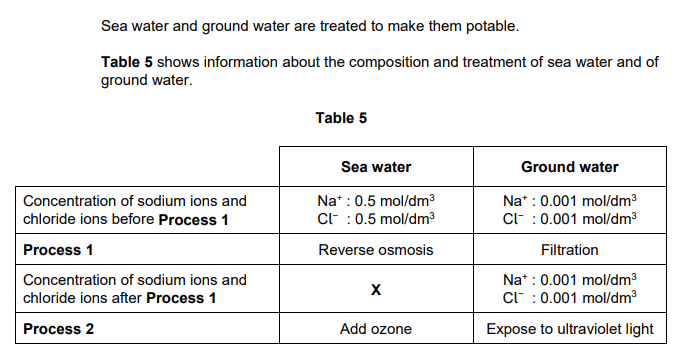
2022: Explain why the concentrations of sodium ions and of chloride ions in the ground water in Table 5 are unchanged by Process 1 (2)

2022: Explain why water needs to be sterilised before it is safe to drink (2)
contains harmful microbes
sterilising destroys the microbes

2022


2022


2022: How does the equation for the reaction show the atom economy for forward reaction is 100%
Only one product
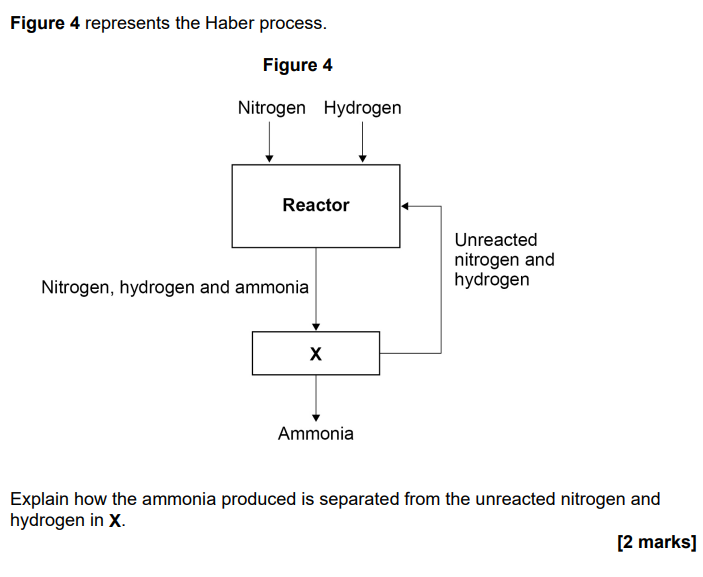
2022
(mixture is) cooled
only ammonia liquefies

2022
