bio lecture 3 4/7
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
What are the 3 categories of the 9 functions of proteins?
Help move things along, help understand and build things, and behind the scenes actions
What are the functions of proteins that help move things along?
Enzymes, motor proteins, and transport proteins
What are the functions of proteins that help build and understand things?
Transcription proteins, receptor proteins, singal proteins.
What are the functions of the proteins involved in the behind the scenes action?
Structural proteins, storage proteins, and special purpose proteins
What are some examples of special purpose proteins?
Antibodies and they work to protect against viruses.
How are antibodies made up structurally?
Composed of four peptide chains (2 identical light chains and two identical heavy chains). They are all held together by disulfide bonds
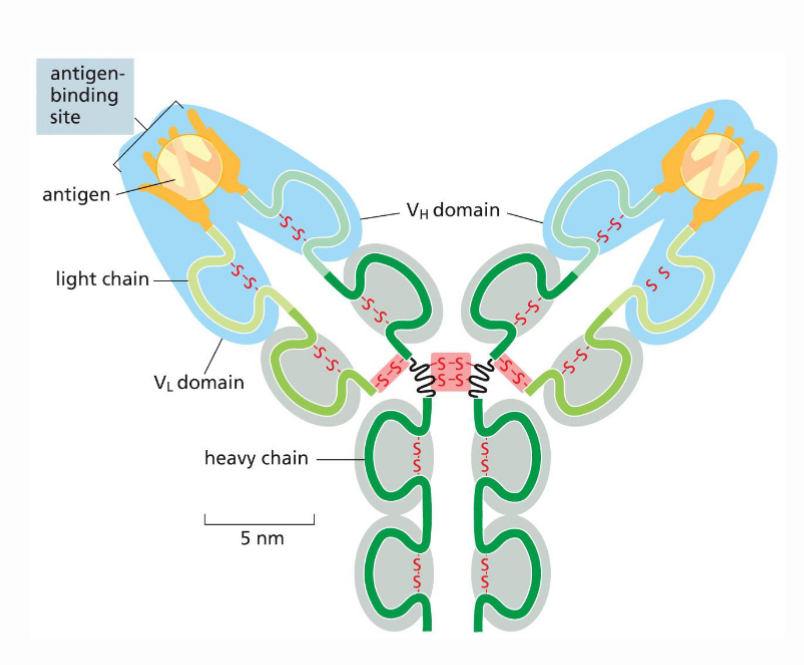
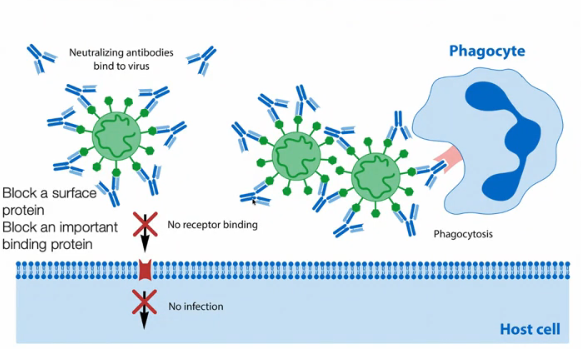
Describe the function of antibodies
Antibodies are a part of the immune system and work to protect against viral infections. They remain in the body for a while after the person is infected. There are two layers of protection that the antibody provides. First, the arms of the antibody will bind to the virus and prevents the virus from binding to our cells and us getting infected. The second mechanism of protection involves phagocytes which are another type of immune cell. The phagocytes will recognize the base of the antibody and eat it up along with the virus attached to it. Phagocytes contain enzymes that break down macromolecules, which allows them to perform this function.
What determines the 3D shape of the proteins?
The sequences of amino acids. Depending on the individual properties of each amino acid, it interacts and folds in unique ways, which all contribute to its final form.
What are the 4 main characteristics of the 20 amino acids?
Hydrophilic, Mildly Hydrophilic, Generally Hydrophobic, and Hydrophobic
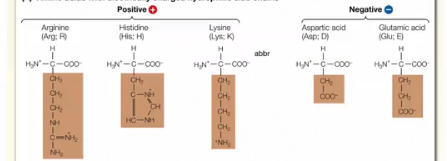
What are the characteristics of Strongly Hydrophilic Amino Acids?
These aa contain either a positive or negative charge. The positive charge is typically found on the N atom on the aa and the negative charge is typically found on the O atoms on the aa. The charged nature of the group attracts it strongly to water making it hydrophilic
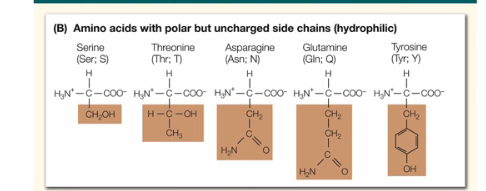
What are the characteristics of mildly hydrophilic aa?
These aa do not contains a charge, but the group is clearly polar (we know this because it contains CH and O atoms)
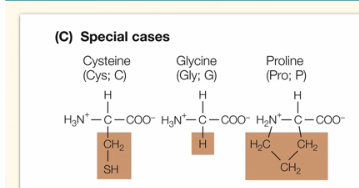
What are the characteristics of generally hydrophobic aa?
These 3 amino acids are very distinct. Cysteine contains S and bonds between two of these aa can be found in antibody structures. Glycine contains only an H atom for its R group. Proline is the only aa that interacts with its own amino group on the polypeptide backbone.
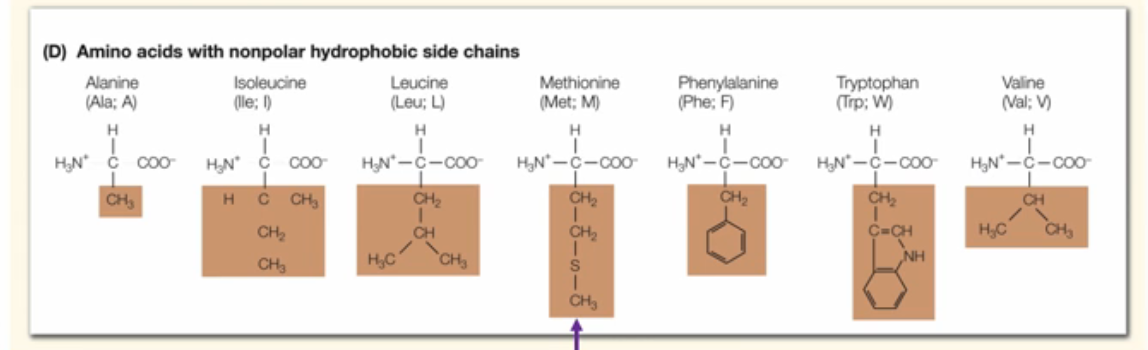
What are the characteristics of generally hydrophobic aa?
These animo acids are clearly non-polar typically containing chaining CH chains.
What are important hydrophobic Animo acids to take note of?
Note that some of the hydrophobic amino acids may appear to be polar because of the presence of a random S or N in the group. Do not confuse this with mildly hydrophilic aa. In these aa, the outlier atoms are ingrained in the chain of CH, whereas for moderately hydrophilic aa, the outlier atoms are always found on the outside. Also note that methionine is the first aa to start the chain.
What are the two components that contribute to the variety in proteins?
The amino acid sequence (20^500 possible combinations), as well is how the protein folds upon itself
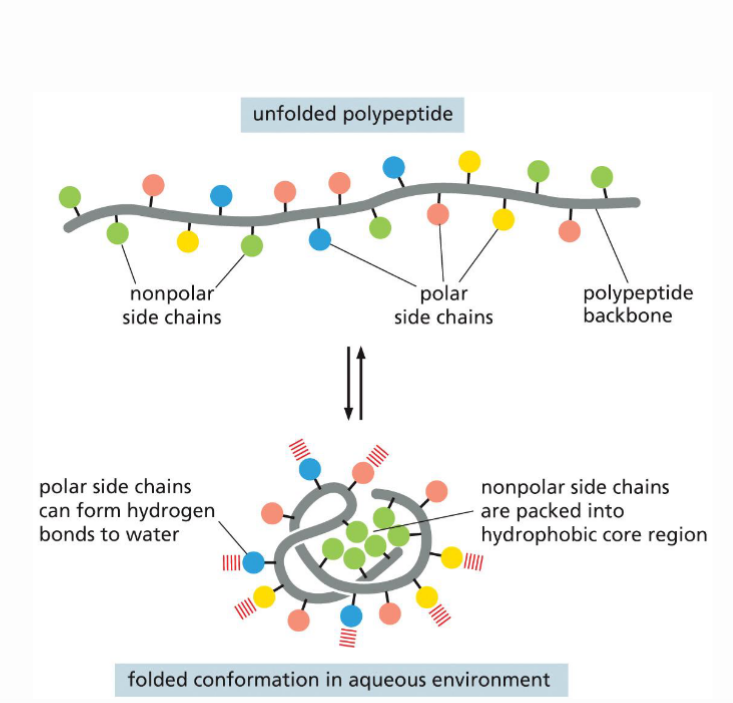
How does the aa sequence contribute to the protein’s shape?
The 4 groups of aa will interact with each other differently, typically the hydrophillic protein will form a shield around the hydrophobic aa, forming a hydrophobic core region and hydrophillic/polar outer region that can from hydrogen bonds with water.
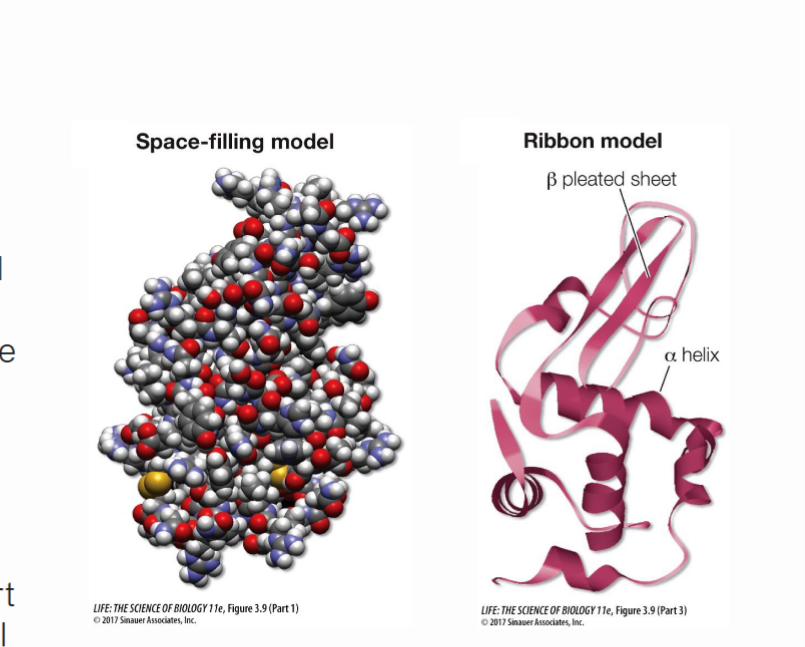
What is lysosome, how it is structured and what is its function?
The lysosome is composed of several secondary structures, which are built together in a final tertiary structure. Its 3D structure works to cleave peptidoglycan bonds, which are bacteria. It can be found in our tears and in some of our immune cells and helps protect against bacterial influence.
What are the four levels of protein structure?
Primary, secondary, tertiary, and quintenary
What are the bonds that stabilize the primary protein structure?
Covalent Peptide Bonds in between amino acids
What are the bonds that stabilize the secondary protein structure?
Stabilized by hydrogen bonds between the amino acids backbone.
Do R groups interact in the secondary structure?
No. Those interactions are only found in the tertiary structures.
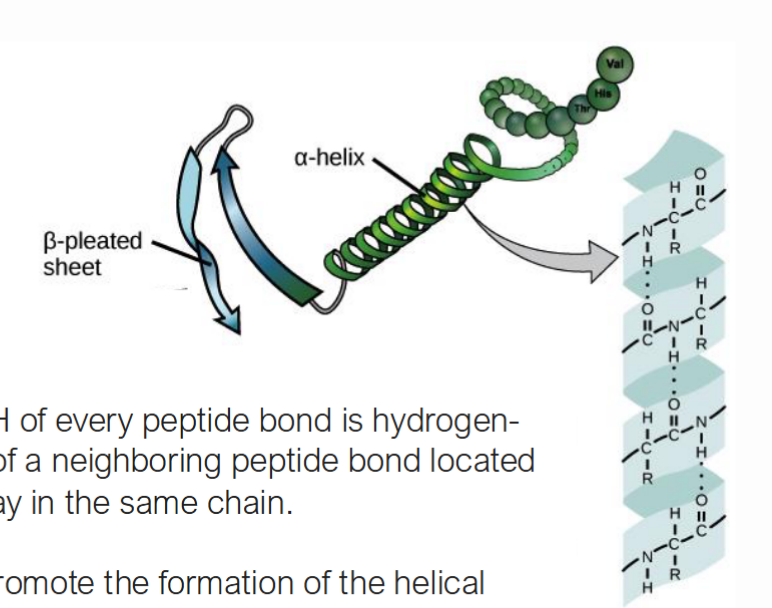
What are the alpha helices and how do they interact with each other?
A helices are ribbon shaped structures found in the secondary proteins structures. The interaction that form this shape is between the N-H of one amino acids to the C=O of another amino acid. The other amino acid is must be four away to allow for correct oritentation
Are the interactions of H groups in a helices and b sheets covalent or non covalent and why?
These interactions are non-covalent. Covalent means that the bond is created through the sharing of electrons. However in this case, there is no electron sharing, rather an attraction between weaker partial charges. This interaction is weak and allows bonds to break and reform easily.
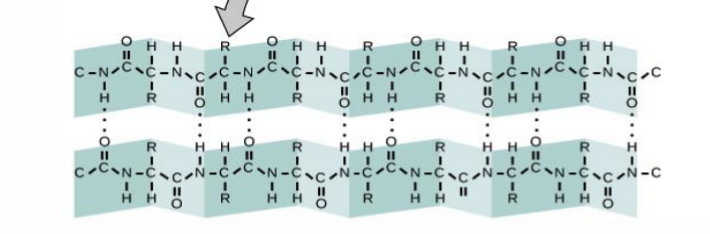
What are the Beta sheets and how do they interact with each other?
In a beta sheets, there is again multiple links of amino acids that fold upon itself. The N-H and C-O interactions occur between neighboring strands.
Where do the R groups project in the B sheets?
The project outside the ribbon because there isn’t enough room for them in the strand.
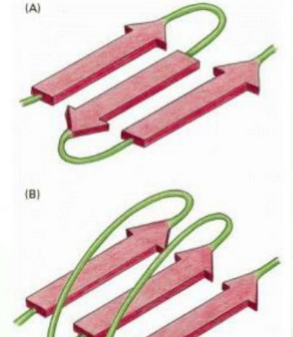
What are the two types of “folding” of the B sheets, and how do you differentiate between them
Parallel: all the strands go in the same direction (N to C), the unstructured region is long, the H bonds are angles/slanted across, making them slightly less stable.
Antiparallel: Strands go in opposite directions. The unstructured regions connecting the strands are short, and the H bonds are straight across, making them more stable
What are the bonds that stabilize the tertiary structure?
Hydrogen bonds, disulfide interactions, hydrophobic interactions, and ionic bonds.
Do interactions occurs between R groups in tertiary structures?
Yes. These interactions play an important role in the folding and resulting shape of the proteins
What is different about the interaction in the secondary and tertiary structures?
Secondary structures ONLY have interactions between the a helices and the b sheets. Tertiary structures have interaction between the a helices/b sheets and other unstructured regions of the aa chain.
In depth, describe the four the interactions that occur in the tertiary structures
1) Ionic interactions between two hydrophilic amino acids (side chain to side chain) 2) small hydrophobic interactions that cause protein to fold over on itself (side chain to side chain), 3)H-bond interactions between the amino acid backbone, BUT these interactions aren’t between helices or β-sheets, they are in between unstructured regions. (backbone to backbone). There are also H bond interaction that can occur between the aa r group and the amino acid backbone (side chain to backbone)
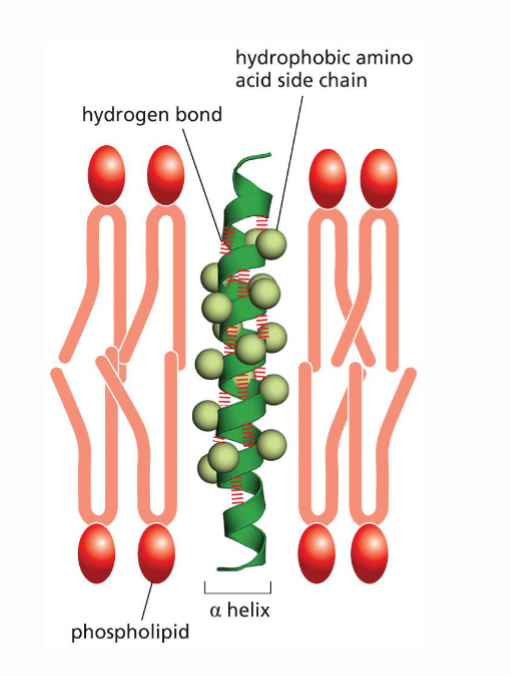
What is the difference in how hydrophobic groups interact withing the cell v.s in the PS Bilayer? Explain the difference in mechanisms
Within the cell, hydrophobic R groups are hidden inside of the cell, and phydrophillic regions provide a protective outer core. Within the PL bilayer, the hydrophobic R groups are exposed on the outside of the protein and interact with the hydrophobic tails of the PL bilayer. Proteins can flip their behavior based on the environment they are in.
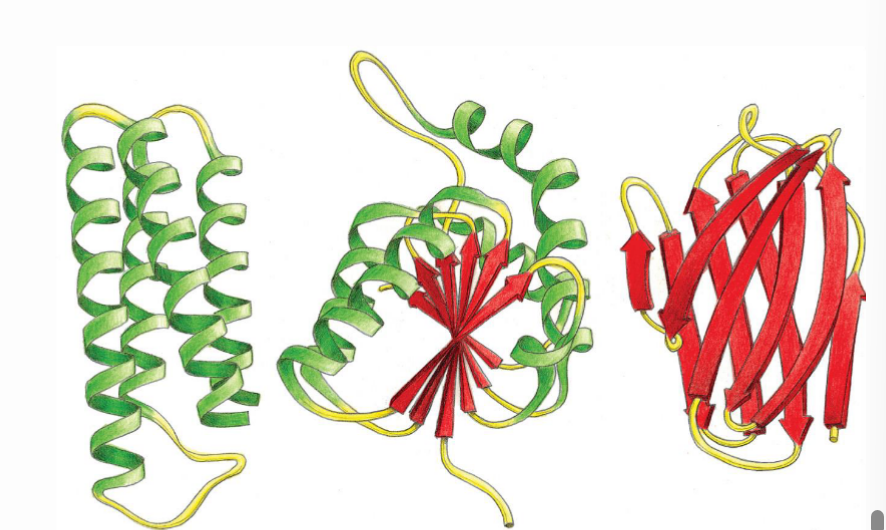
How can tertiary structures vary and what is the reasoning behind this?
Structures can be made up of only a helices, only b sheets, or a mixture of both, all depends of the sequence of the amino acids.
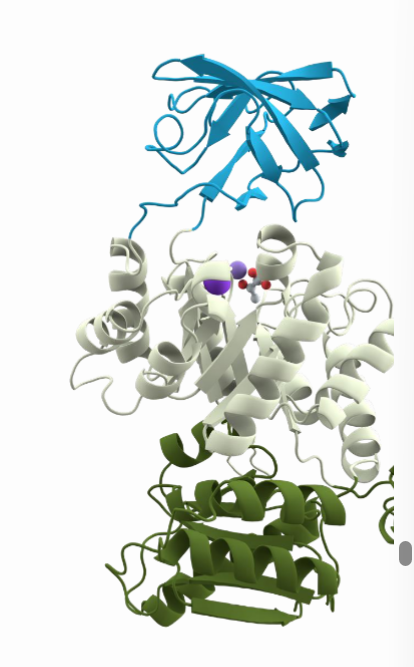
What are proteins domain and what are the ways they differ from each other?
Protein domains are regions within one specific protein that act independently from one another. They differ in function and differ in formation.
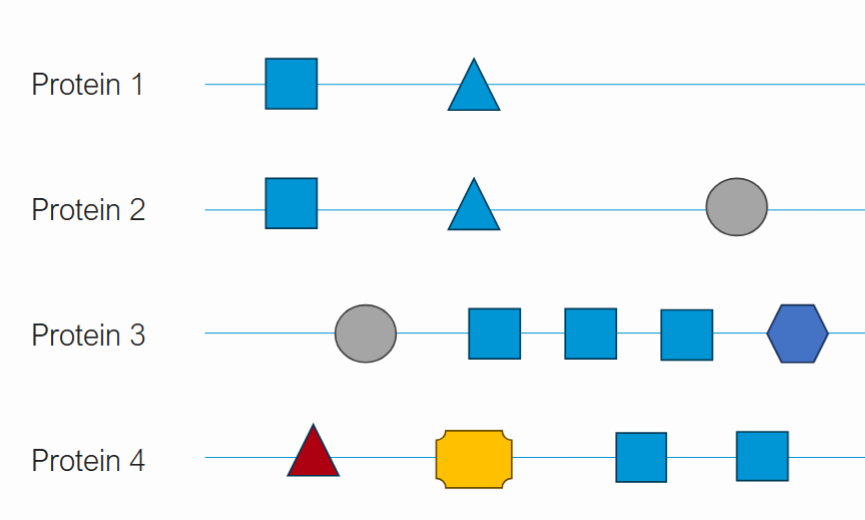
How can proteins have related but distinct functions?
Proteins may have some of the same protein domains. For example, protein 1 and 2 may have a similar protein domain that helps with binding and another that helps with catalyzing a reaction. However, protein 2 may have an additional protein domain that makes it different. The more differences in protein domains, the more specific and diverse functions a protein can have.
What are the bond interactions that stabilize quaternary structures?
Noncovalent hydrogen bonds, disulfide bonds, ionic interactions, hydrophobic interactions
What are some characteristics of quantenary structures?
Do not occur for every protein. Composition of multiple subunits (tertiary proteins structures). Each subunit is an individual polypeptide chain.
What is the quaternary structure of hemoglobin?
Two subunits are a globin, and two subunits are b globin. Each of these subunits carries a heme molecule where oxygen can reversibly bind.
What type of protein is hemoglobin?
transport proteins
What are the causes and effects of protein misfolding?
Improper folding is caused by a mutation in the amino acid sequence. When mechanisms that control misfolding do not work properly, misfolding will occur. This leads to many neurodegenerative diseases, including Alzheimers, Parkinsons, huntingtons, and ALS
What are molecules that aid protein folding in cell and how do they work to prevent misfolding?
Chaperones are molecules that aid proteins folding in cell. Typically, when a protein folds, it favors folding together hx regions together despite is that folding is correct. this may lead to improper folding. Chaperones work by shielding the hx regions without the proteins having to improperly fold.
How do cells deal with protein misfolding? Explain the mechanism of destroying misfolded proteins
Through proteosome mediated proteins destruction. First, the misfolded protein will be tagged with ubiquitin, a protein that marks the bad protein for destruction. Then the ubiquitin signals the protein to move to a proteosome to be destroyed. Within the proteosome, the target protein will bind to the polyubiquitin site. Once attached, the ubiquitin will be removed and recycled, and the target protein will stretch out to the active site of the proteosome. The active site contains a protease, which cleaves amino acids. As the protein passes through the protease, it will degrade into amino acids.