C3.4 - Electrolysis
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
What is electrolysis?
The process of breaking down a compound using electricity. An electric current is passed through a molten or dissolved ionic compound (the electrolyte), causing it to decompose
What are the key terms in electrolysis?
Electrode: rod of metal or graphite through which current enters/leaves.
Anode: positive electrode.
Cathode: negative electrode.
Electrolyte: molten or dissolved ionic compound that conducts electricity.
Cation: positively charged ion.
Anion: negatively charged ion
Which way do ions move during electrolysis and why?
Positive ions (cations) move to the negative cathode. Negative ions (anions) move to the positive anode. Opposite charges attract
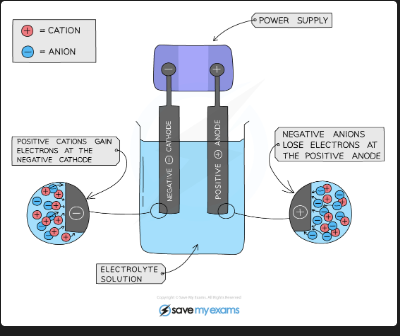
What happens at the cathode and anode during electrolysis?
Cathode (negative): positive ions gain electrons and are reduced. Anode (positive): negative ions lose electrons and are oxidised. Remember: OILRIG — Oxidation Is Loss, Reduction Is Gain
Describe the full process of electrolysis of a molten ionic compound
Set up electrolytic cell with two electrodes connected to a power supply, immersed in the molten/dissolved electrolyte.
Switch on power supply.
Ions in the electrolyte are free to move.
Cations migrate to the cathode and gain electrons (reduced) → form neutral atoms deposited at cathode.
Anions migrate to the anode and lose electrons (oxidised) → form neutral atoms or molecules released at anode.
Identify products by colour, gas produced, or mass change at electrodes
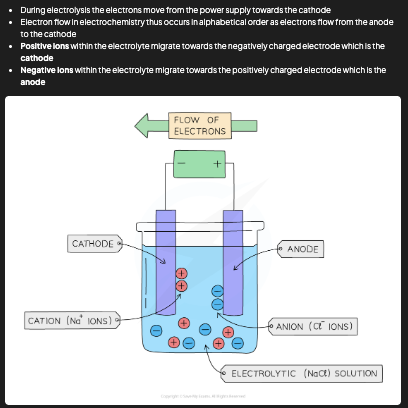
Why can ionic compounds only be electrolysed when molten or dissolved?
In a solid ionic compound, the ions are in fixed positions and cannot move. When melted or dissolved in water, the ions are free to move towards the electrodes and carry charge
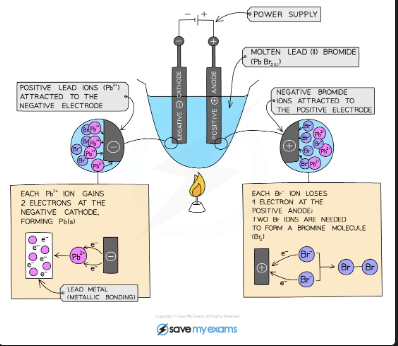
What are the products of electrolysis of molten lead(II) bromide?
Cathode: lead metal is deposited (Pb²⁺ ions reduced). Anode: orange-brown bromine gas is produced (Br⁻ ions oxidised). The molten compound provides only Pb²⁺ and Br⁻ ions so there is no competition
What are the products of electrolysis of molten aluminium oxide (Al₂O₃)?
Cathode: aluminium metal is deposited (Al³⁺ reduced). Anode: oxygen gas is produced (O²⁻ oxidised). This is how aluminium is extracted industrially
Why is electrolysis of aqueous solutions more complex than molten compounds?
Aqueous solutions always also contain water molecules which partially dissociate: H₂O ⇌ H⁺ + OH⁻. This means there are additional H⁺ and OH⁻ ions competing with the ions from the dissolved compound — so the product at each electrode depends on which ion is preferentially discharged
What is produced at the cathode in aqueous electrolysis and why?
If the metal is above hydrogen in the reactivity series → hydrogen gas is produced (H⁺ ions are discharged instead). If the metal is below hydrogen (e.g. Cu, Ag) → the metal is deposited at the cathode
What is produced at the anode in aqueous electrolysis and why?
If the anion is a halide (Cl⁻, Br⁻, I⁻) and it is concentrated → the halogen gas is produced. If the anion is a sulfate or nitrate (or dilute halide) → oxygen gas is produced from the discharge of OH⁻ ions from water
What are the products of electrolysis of concentrated sodium chloride solution (brine)?
Anode: chlorine gas (Cl⁻ ions discharged — concentrated so halide wins). Cathode: hydrogen gas (Na is above H in reactivity series). Remaining solution: sodium hydroxide (NaOH)
What are the products of electrolysis of dilute sodium chloride (NaCl) solution?
Anode: oxygen gas (dilute — OH⁻ wins over Cl⁻). Cathode: hydrogen gas. Remaining solution: sodium hydroxide
What are the products of electrolysis of concentrated copper(II) sulfate solution (CuSO₄)?
Anode: oxygen gas (SO₄²⁻ not a halide so OH⁻ discharged). Cathode: copper metal deposited (Cu is below H in reactivity series). If copper electrodes are used — see electroplating/purification
What are inert electrodes and why are they used?
Inert electrodes (usually graphite or platinum) do not react with the electrolyte or products during electrolysis. They act as a surface for reactions to occur without affecting the outcome — used as the standard electrode for most electrolysis experiments
What are non-inert (reactive) electrodes and when are they used?
Sometimes the electrode material itself takes part in the reaction — e.g. copper electrodes in copper purification. The anode dissolves and the cathode gains mass as the same metal
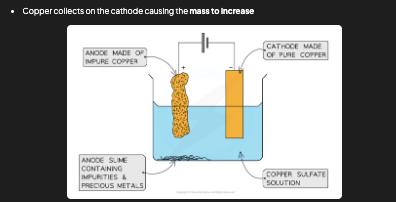
How is copper purified by electrolysis?
Anode = impure copper (dissolves). Cathode = pure copper (grows). Electrolyte = copper sulfate solution (CuSO₄). At anode: Cu → Cu²⁺ + 2e⁻ (oxidation). At cathode: Cu²⁺ + 2e⁻ → Cu (reduction). Impurities fall to the bottom. The mass of the anode decreases and the mass of the cathode increases
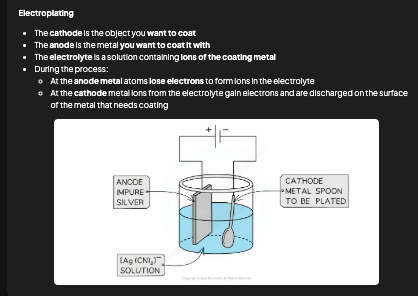
What is electroplating and how does it work?
Electroplating coats an object with a thin layer of metal. Cathode = object to be plated. Anode = metal you want to coat with. Electrolyte = solution containing ions of the plating metal. Metal ions are deposited from the solution onto the cathode surface
[HIGHER] What is a half equation and what must it show?
A half equation shows the oxidation or reduction reaction at one electrode, including electrons. The number of electrons must balance the charges on each side. Electrons on the left = reduction (cathode). Electrons on the right = oxidation (anode)
[HIGHER] Write the half equations for electrolysis of molten lead(II) bromide
Cathode: Pb²⁺ + 2e⁻ → Pb (reduction). Anode: 2Br⁻ → Br₂ + 2e⁻ (oxidation). OR: 2Br⁻ - 2e⁻ → Br₂
[HIGHER] Write the half equations for electrolysis of molten aluminium oxide
Cathode: Al³⁺ + 3e⁻ → Al (reduction). Anode: 2O²⁻ → O₂ + 4e⁻ (oxidation). OR: 4O²⁻ - 4e⁻ → 2O₂
[HIGHER] Write the half equations for concentrated NaCl solution (brine)
Cathode: 2H⁺ + 2e⁻ → H₂ (reduction). Anode: 2Cl⁻ → Cl₂ + 2e⁻ (oxidation)
[HIGHER] Write the half equations when oxygen is produced at the anode
4OH⁻ → O₂ + 2H₂O + 4e⁻ (oxidation of hydroxide ions from water). OR: 2H₂O → O₂ + 4H⁺ + 4e⁻
[HIGHER] Write the half equations for electrolysis of concentrated copper(II) sulfate with inert electrodes
Cathode: Cu²⁺ + 2e⁻ → Cu (reduction). Anode: 4OH⁻ → O₂ + 2H₂O + 4e⁻ (oxidation — sulfate not discharged)
[HIGHER] Write the half equations for copper purification with copper electrodes
Anode: Cu → Cu²⁺ + 2e⁻ (oxidation — anode dissolves).
Cathode: Cu²⁺ + 2e⁻ → Cu (reduction — pure copper deposited)