Chemistry CCEA AS Level 1.3 - 1.6 (Bonding, Intermolecular Forces, Structure, Shapes or molecules and ions)
1/61
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms
What is the octet rule?
When forming a compound, an atom tends to gain, lose or share electrons in its outer shell to achive 8 in its outer shell
What is ionic bonding?
Electrostatic attraction between oppositely charged ions formed by electron transfer
What does ionic bonding involve?
Transfer of one or more electrons from metal atoms to non-metal atoms
What does ionic bonding occur between?
A metal and a non metal
What happens to the metal atom in ionic bonding?
It loses electrons and becomes a positively charged cation
What happens to the non-metal atom in ionic bonding?
It gains electrons and becomes a negatively charged anion
What holds together a giant ionic lattice?
Strong electrostatic forces of attraction between cations and anions
What is a lattice?
Regular arrangement of particles.
Why do ionic compounds have high melting and boiling points?
Strong electrostatic bonds of attraction require large amounts of energy to break
Why do ionic compounds conduct electricity only when molten or in solution?
When in a solution/molten the ions are free to move and carry charge
What is a covalent bond?
The electrostatic attraction between a shared pair of electrons and the nuclei of bonded atoms
What is a lone pair?
An unshared pair of electrons in the outer shell of an atom
What is a coordinate covalent bond?
A shared pair of electrons between two atoms One atom provides both electrons.
Why do simple covalent molecules have low melting and boiling points?
Weak intermolecular forces between molecules which only require small amount of energy to break
Why don't simple covalent molecules conduct electricity?
They have no delocalised electrons which are free to move and carry charge
What compounds have giant covalent structures/lattices?
Diamond and Graphite
What is the structure of diamond?
Each carbon is bonded to 4 others in a tetrahedral arrangement
What is the structure of graphite?
Each carbon atom forms three covalent bonds to create layers of hexagons. Each carbon atom also has one delocalised electron.
Why can diamond not conduct electricity?
It has no delocalised electrons
Why do diamond and graphite have high melting/boiling points?
The covalent bonds within their lattices require large amounts of energy to break
Why is diamond hard?
Each carbon forms four covalent bonds which are strong and require large amounts of energy to break
Why can graphite conduct electricity?
Once electron from each carbon is delocalised so can carry a charge throughout the structure
Why is graphite soft?
Graphite forms layers, which can slide over one another
What is metallic bonding?
Attraction between positive ions and delocalised electrons in a lattice
What is a delocalised electron?
An outer electron that does not have a fixed position but can move freely
Why do metals have high melting and boiling points?
Metallic bonds are strong and require large amounts of energy to break
Why do metals conduct electricity?
The bonding in metals creates delocalised electrons which are free to move and carry charge
Why are metals hard?
The metallic bonds within the lattice are strong and require large amounts of energy to break
What determines the shape of a molecule or ion?
Repulsion between the electron pairs surrounding a central atom
Why do lone pairs repel more than bonded pairs?
Lone pairs are more electron dense than bonded pairs
What is the order of the relative strengths of repulsion?
Lone pair/lone pair > bonded pair/lone pair > bonded pair/bonded pair
What shape is a molecule with 2 bonded pairs and no lone pairs?
Linear
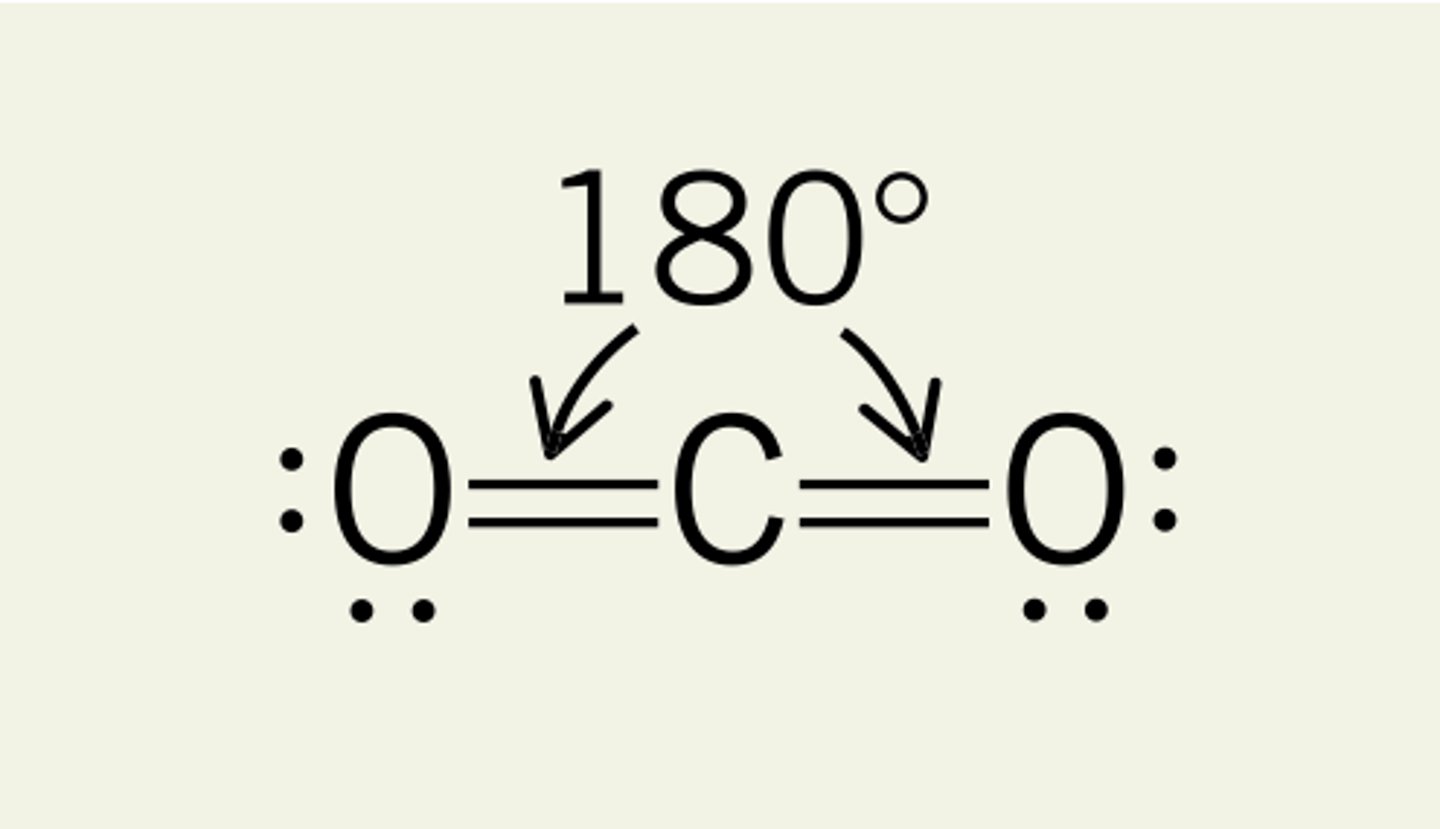
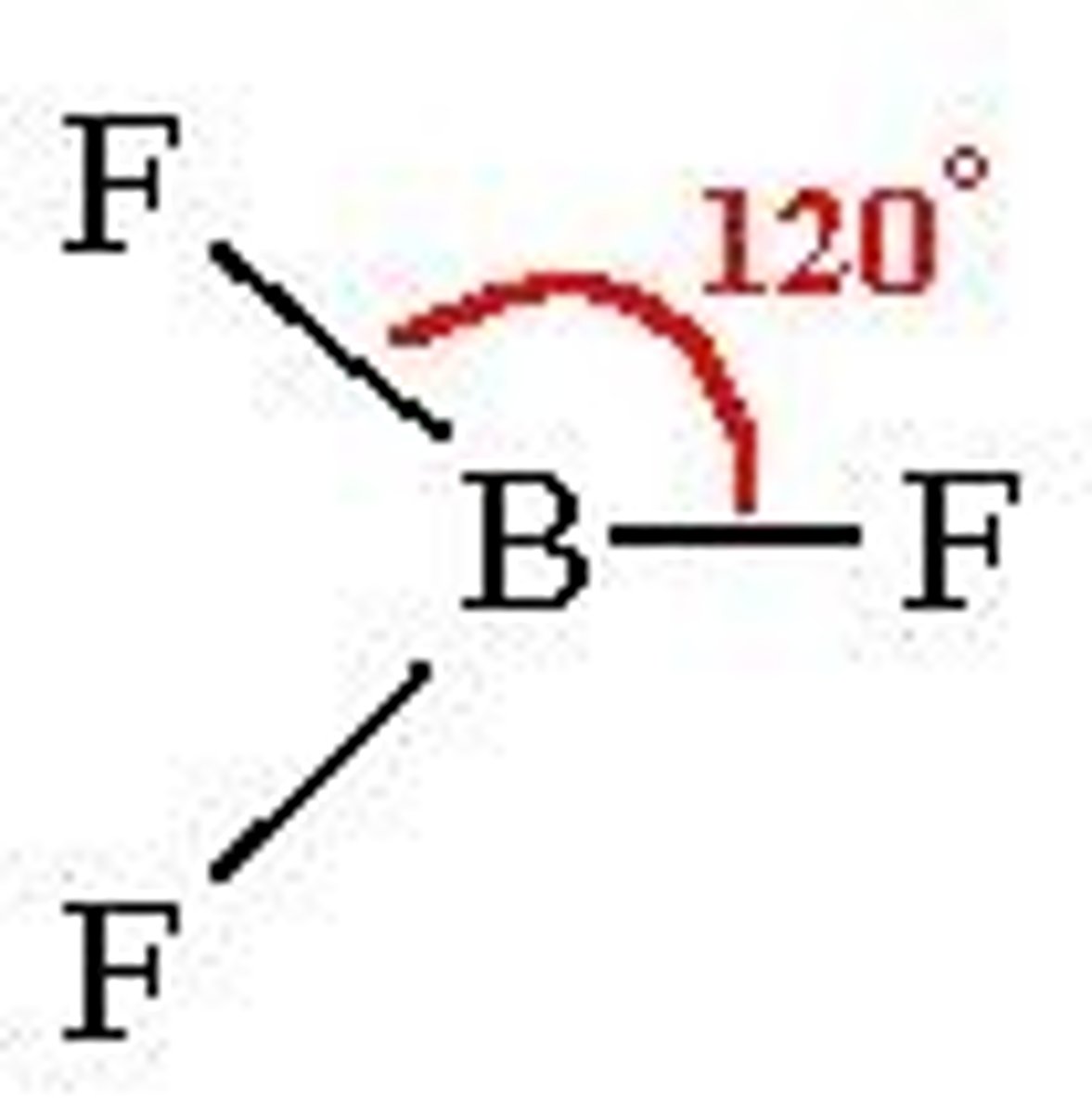
What shape is a molecule with 3 bonded pairs and no lone pairs?
Trigonal Planar
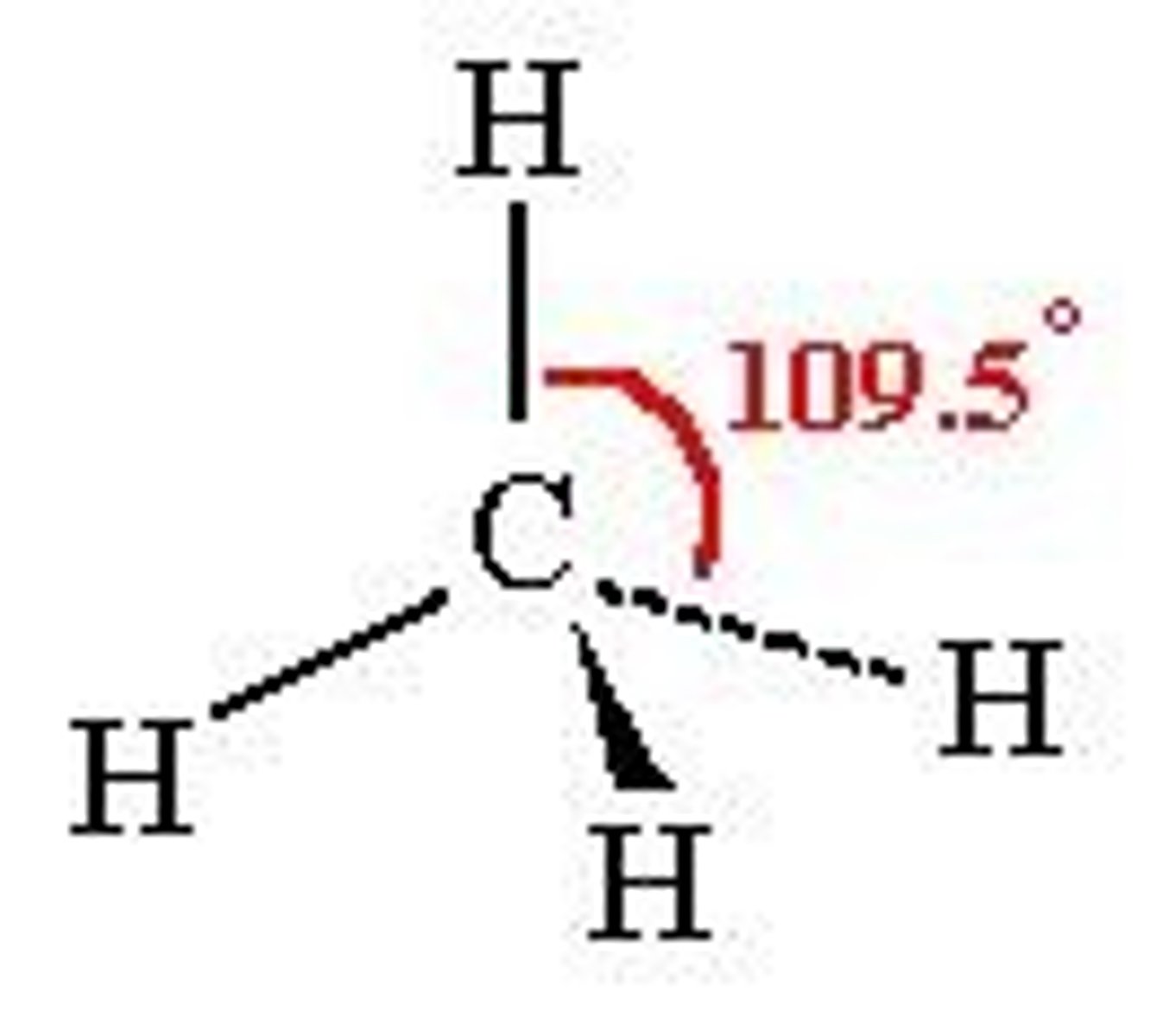
What shape is a molecule with 4 bonded pairs and no lone pairs?
Tetrahedral
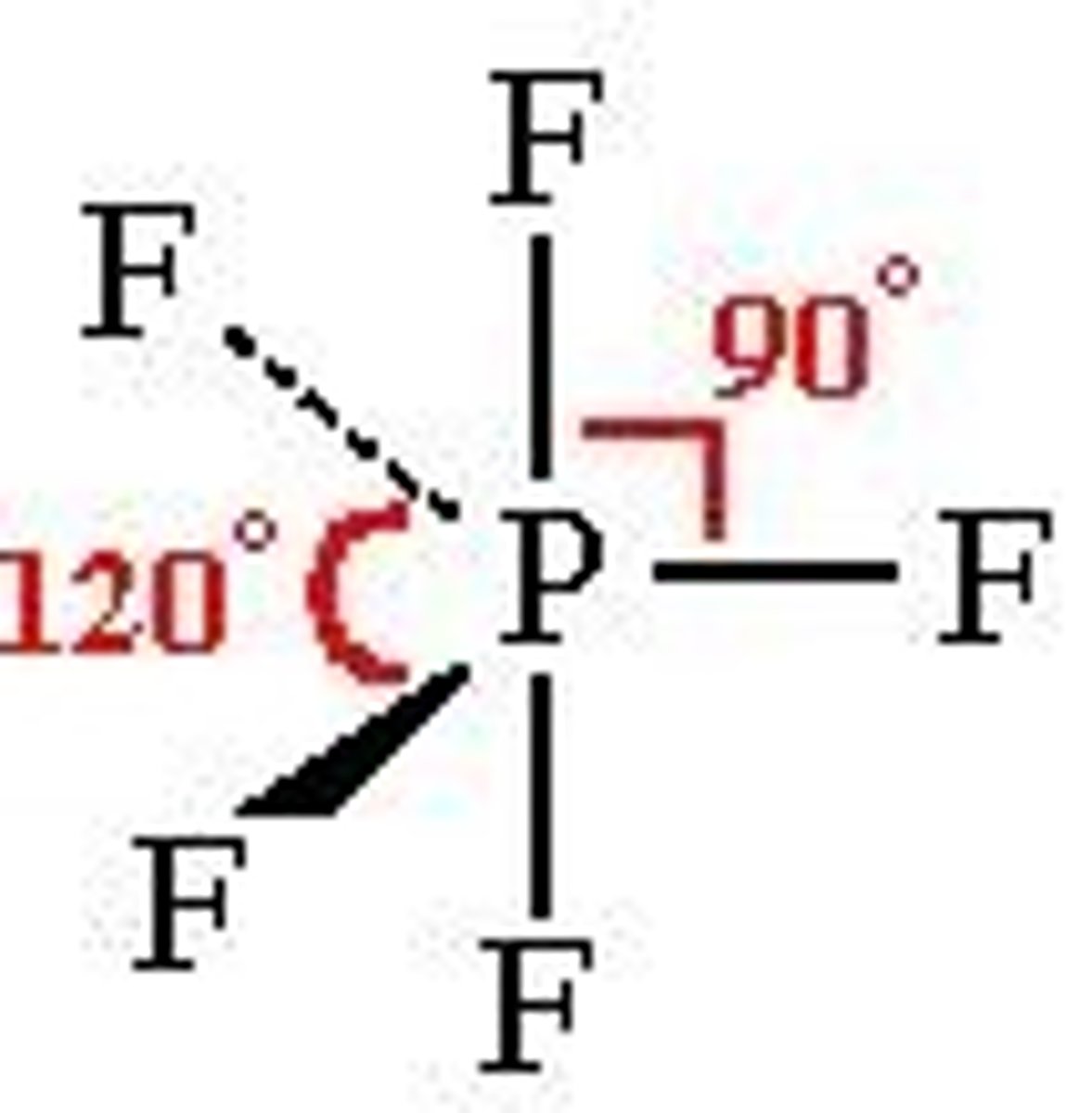
What shape is a molecule with 5 bonded pairs and no lone pairs?
Trigonal bipyramidal
What shape is a molecule with 6 bonded pairs and no lone pairs?
Octahedral
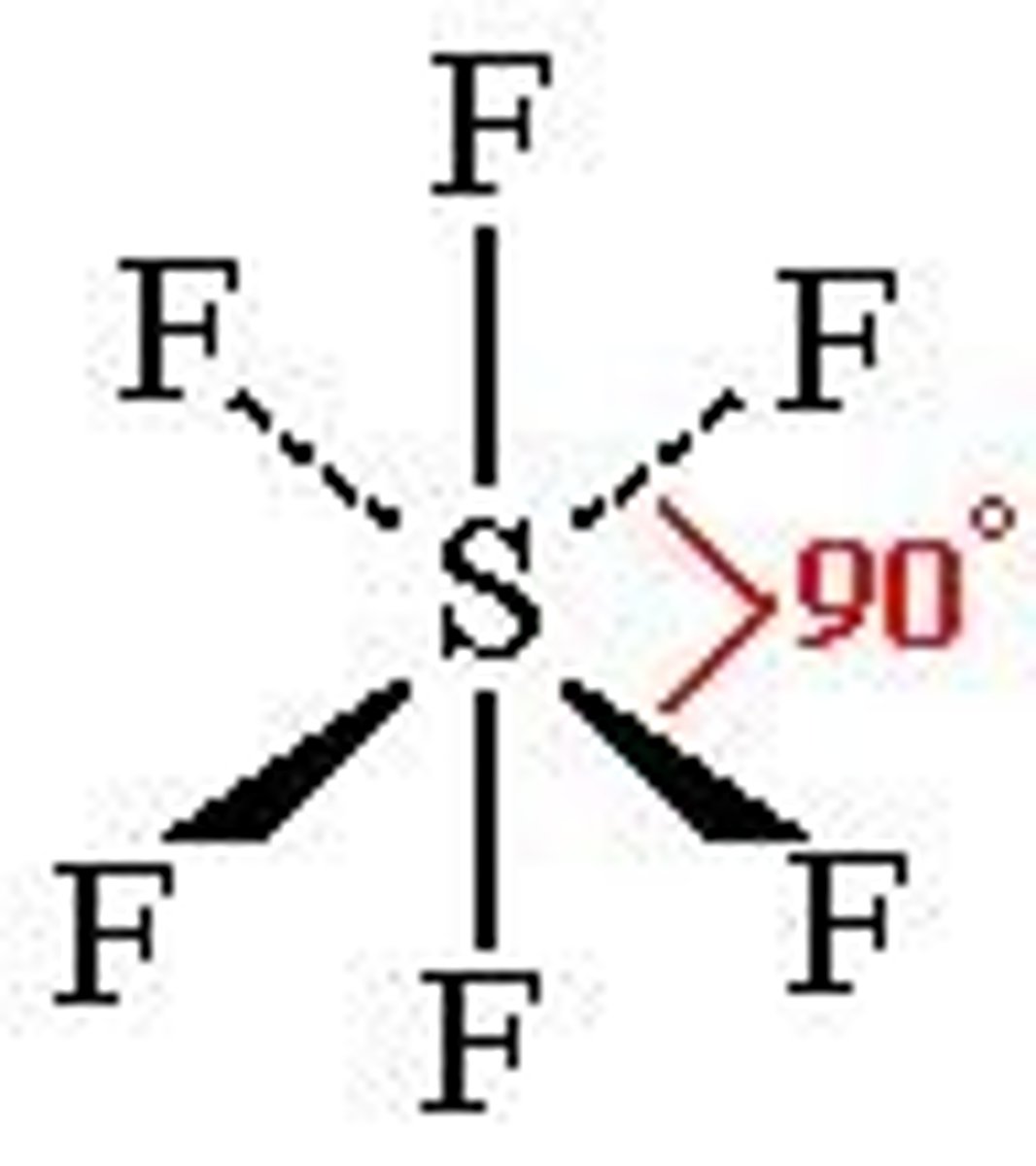
What shape is a molecule with 3 bonded pairs and 1 lone pair?
Pyramidal
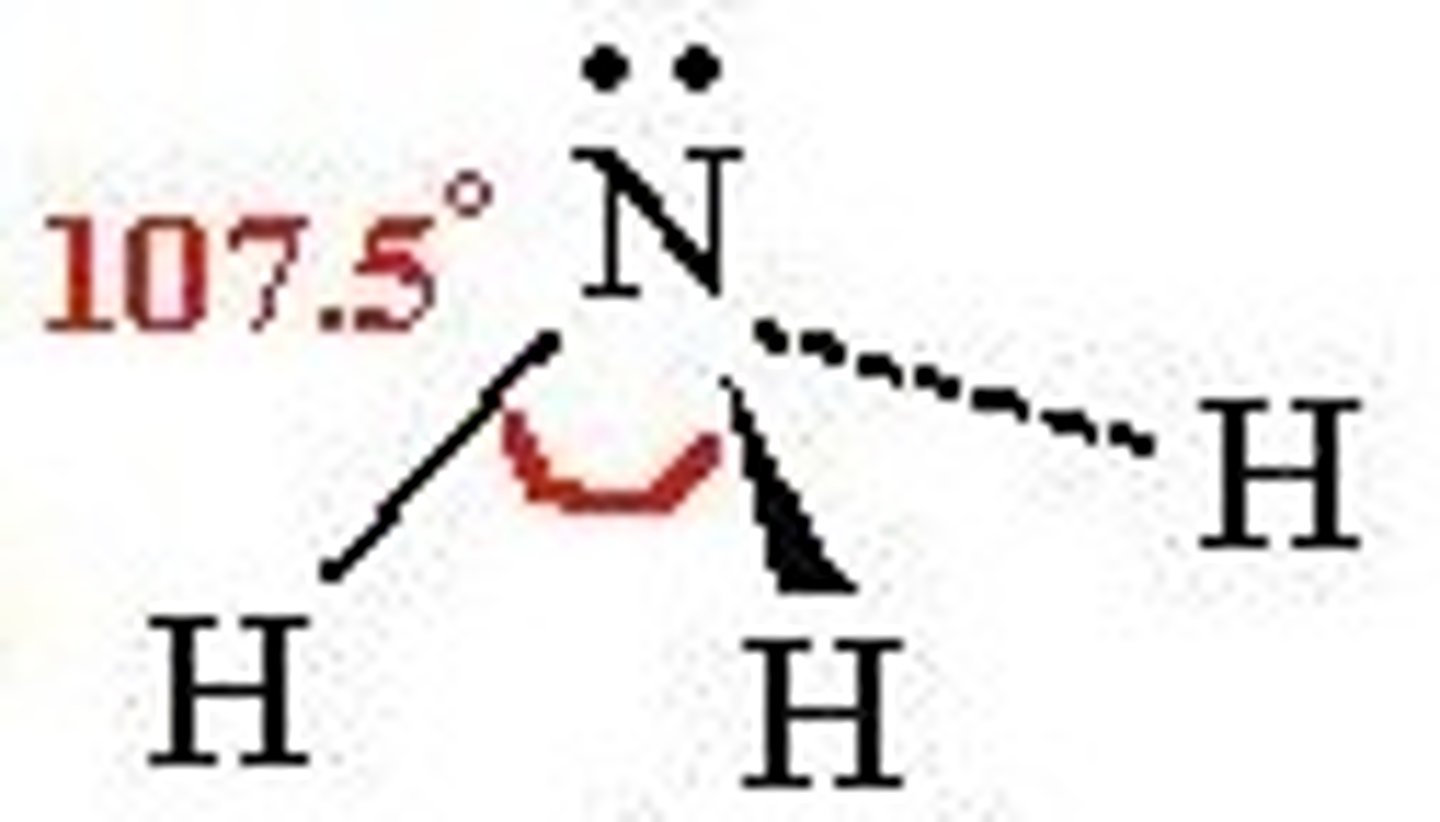
What shape is a molecule with 2 bonded pairs and 2 lone pairs?
bent
What shape is a molecule with 3 bonded pairs and 2 lone pairs?
T shaped
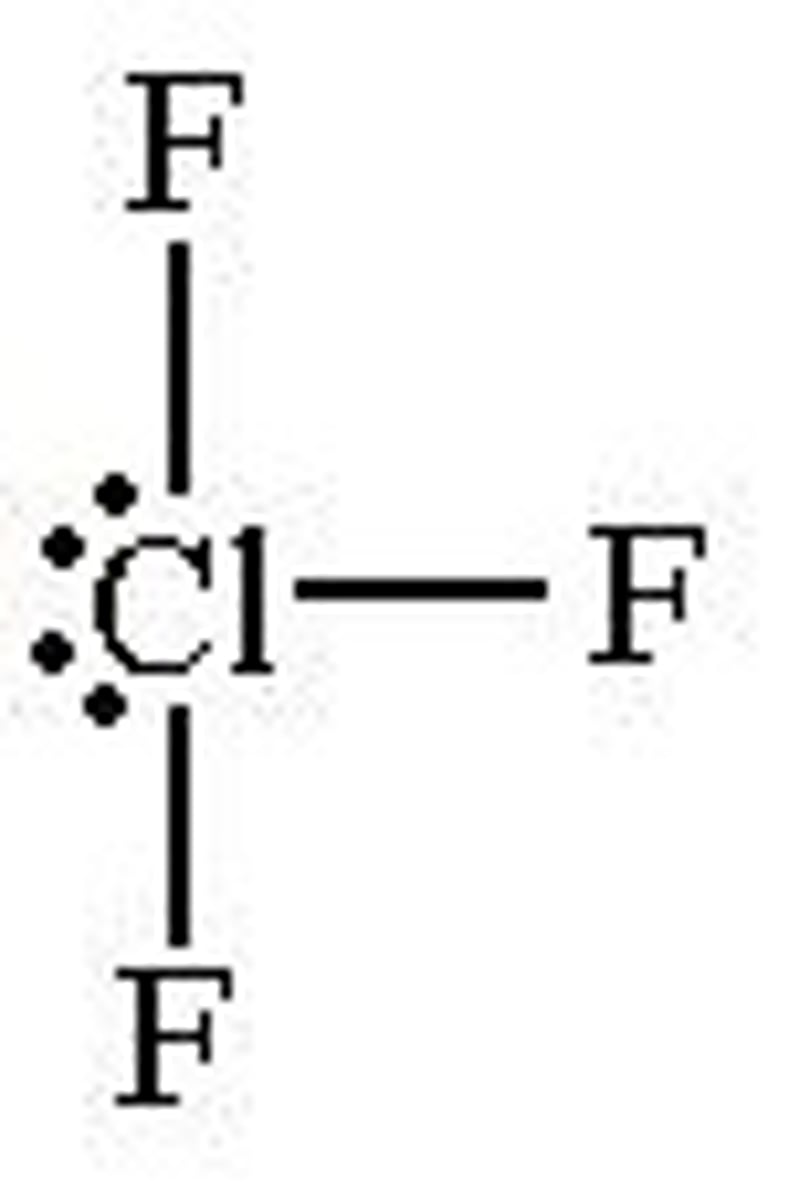
What shape is a molecule with 4 bonded pairs and 2 lone pairs?
Square planar
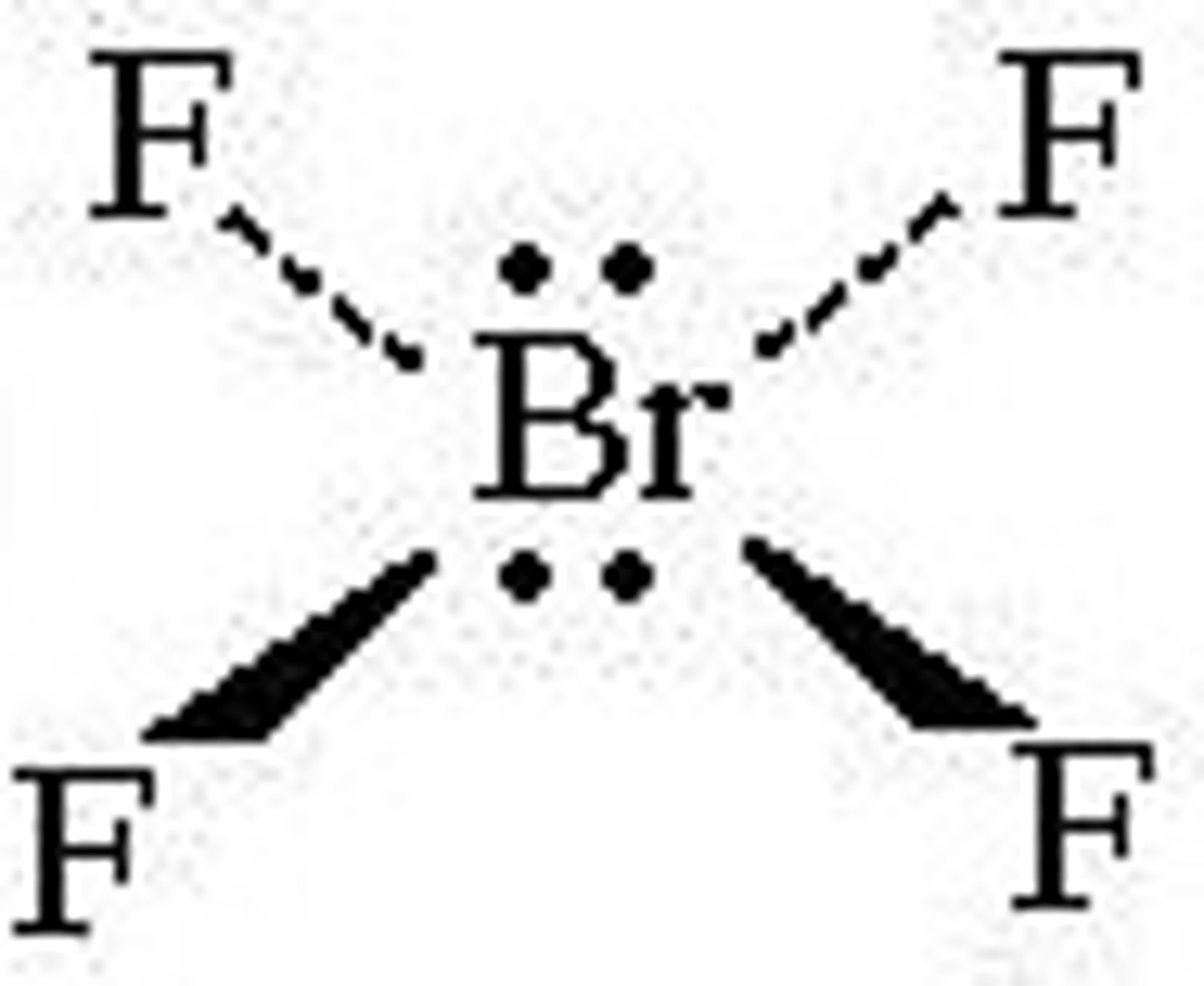
What shape is a molecule with 2 bonded pairs and 3 lone pairs?
Linear
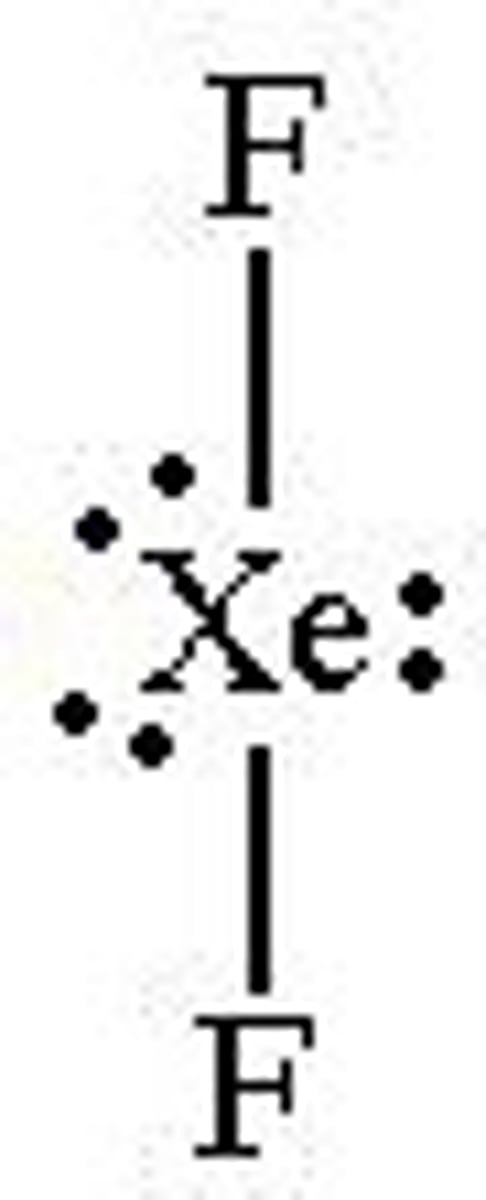
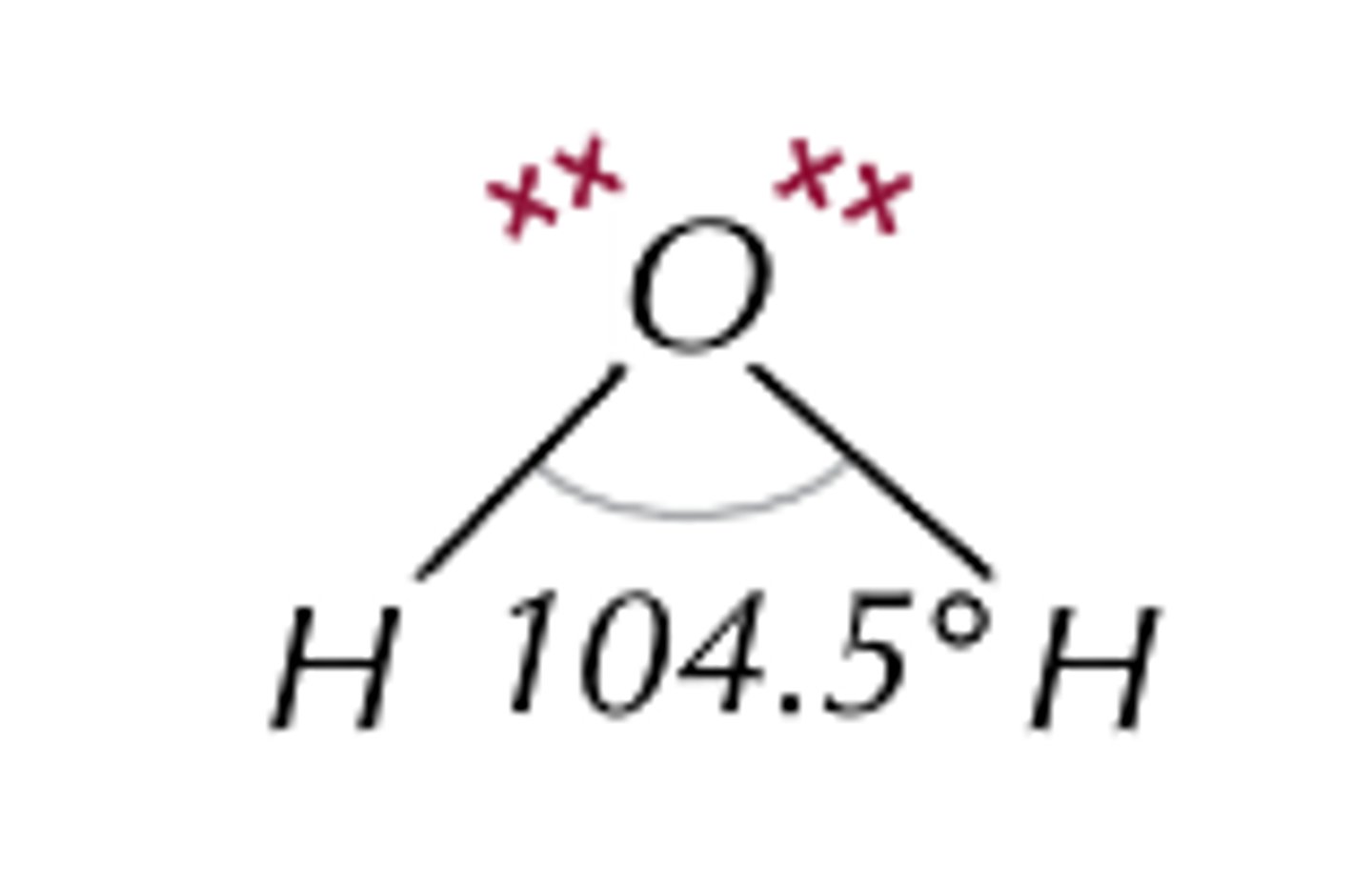
What is the bond angle of a non-linear molecule?
104.5°
What is the bond angle of a trigonal planar molecule?
120°
What is the bond angle of a linear molecule?
180°
What is the bond angle of a tetrahedral molecule?
109.5°
What is the bond angle of a trigonal bipyramidal molecule?
90° and 120°
What is the bond angle of a octahedral molecule?
90°
What is the bond angle of a pyramidal molecule?
107°
What is the bond angle of a T shaped molecule?
86° (<90°)
What is the bond angle of a square planar molecule?
90°
What is electronegativity?
The extent to which an atom attracts the bonding pair of electrons in a covalent bond to itself
What causes bond polarity?
Covalently bonded atoms with different electronegativities
What is a polar bond?
A covalent bond where there is unequal sharing of bonding electrons
What are the 3 intermolecular forces?
- Hydrogen bonds
- Permanent dipole-permanent dipole attraction
- Van der Waals forces/Instantaneous dipole attraction
What is a hydrogen bond?
Attraction between a lone pair of electrons on a very electronegative atom and a hydrogen on a neighbouring molecule
Why is ice less dense than water?
Ice crystallizes with an open structure, and the gaps that form between the water molecules in ice increase its volume.
Why does water have a higher melting/boiling point than other group 6 hydrides?
The hydrogen bonds in water are stronger than the permanent dipole-permanent dipole attraction in the other molecules and thus require more energy to break
What is permanent dipole-permanent dipole attraction?
Attraction between the positive end of the permanent dipole on one molecule and the negative end of the permanent dipole on another
What are Van der Waals forces?
Attraction between instantaneous and reduced dipoles on neighbouring molecules
Why do van der Waals forces increase down a group?
Increased RMM and larger electron cloud meaning more points of contact between separate molecules
Why does melting point decrease with branching?
The electron cloud becomes more spherical, meaning there are fewer points of contact between molecules and therefore weaker van der Waals forces
What determines ionic or covalent character?
The greater the difference in electronegativity of the atoms involved, the more ionic the bond and the smaller the difference in electronegativity of the atoms involved the more covalent the bond