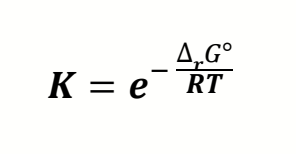solution equilibria
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
what is the equilibrium constant a ratio of?
ratio between products and reactants at constant temperature
when is equilibrium reached in an irreversible reaction?
when all reactants used up
what is the equation to convert between Kc and Kp?
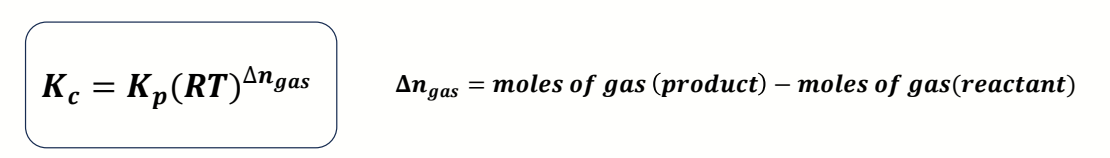
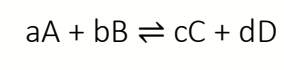
write Keq with activities and explain why dimensionless
is it concentration?
activity is dimensionless
effective concentrations = chemical potential of a species in a real solution or gas
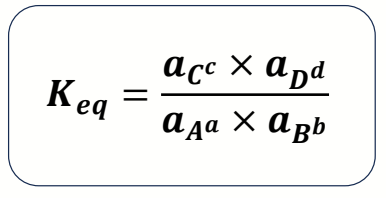
what does effective concentration depend on? 3
temperature
pressure
composition - e.g. conc or ionic strength of compounds
what is the equation for activity including activity coefficient?
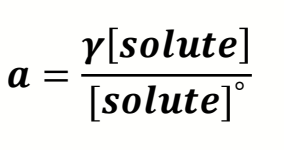
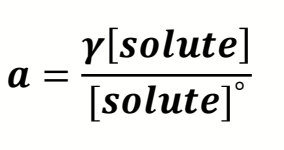
how does γ change when close/far from ideal species?
γ varies between 0 and 1
far away from ideal = close to 0
close to ideal = nearer 1
what is chemical potential μi? how does reaction proceed based on it?
partial molar Gibbs free energy of a species i
driving potential of transfer of matter between species
spontaneous reaction proceeds from high chemical potential to low chemical potential
what is equation for chemical potential of a species using activity?
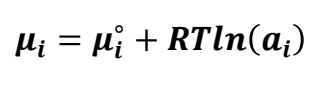
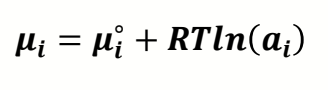
how does this become Gibbs?
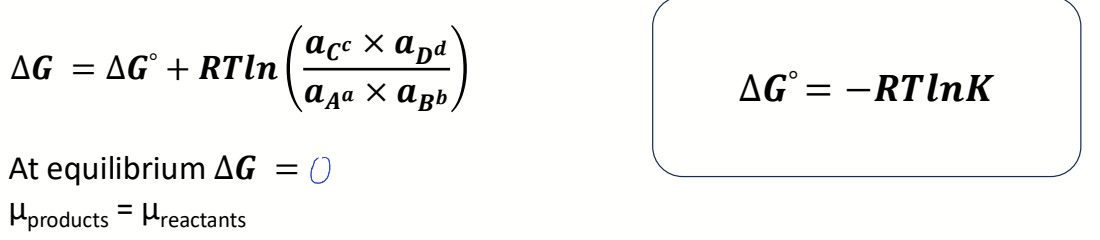
![<p>what is [solute]<sup>0</sup> approximated to?</p>](https://knowt-user-attachments.s3.amazonaws.com/da77ad91-5def-4e75-935d-7f26ed8ca092.png)
what is [solute]0 approximated to?
1 mol dm-3
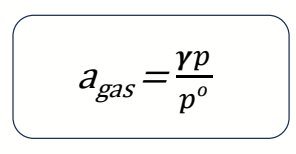
what is standard state for gas pressure?
what is activity of a gas?
p0 = 1 bar
fugacity
what is K’?
equilibrium constant for reverse reaction
inverse of K
what is le Chatelier’s principle?
if an equilibrium is disturbed the system will proceed in the direction to oppose the change
which way does reaction go if Q<K? if Q>K?
Q<K proceeds to right,
Q>K proceeds to left
how to find K from Gibbs?