Structure and Bonding (3)
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
Why do elements in groups 1, 2, 6 and 7
readily form ions?
So they become more stable and
achieve the electron structure of the
noble gases (group 8).
What type of ions do elements in group 1
and 2 form?
Cations (positive)
- Group 1 metals will form 1+ ions
- Group 2 metals will form 2+ ions
What type of ions do elements in groups
6 and 7 form?
They are non-metals so form anions
(negative)
- Group 6 will form 2- ions
- Group 7 will form 1- ions
What does it mean if an ionic compound
ends in -ide?
The compound contains 2 elements.
What does it mean if an ionic compound
ends in -ate?
The compound contains at least three
elements, one of which is oxygen.
Describe the structure of an ionic
compound
(LIR)
Lattice structure.
Ions held together by strong electrostatic forces between ions with opposite charges.
Regular arrangement of ions.
What is the chemical formula of sodium
oxide, formed from Na+ and O2-?
Na2O
Charges must balance so 2 sodium ions
are required.
What is the chemical formula of
magnesium hydroxide, formed from Mg2+ and OH-?
Mg(OH)2
Charges must balance so 2 hydroxide
ions are required.
What is a covalent bond?
A bond formed when a pair of electrons is
shared between two atoms.
What forms as a result of covalent
bonding?
A molecule
Draw a dot and cross diagram for the
formation of methane (CH4
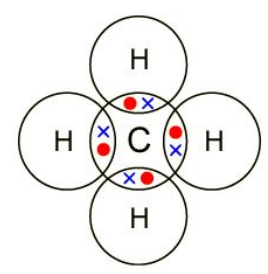
True or false?
‘Covalent bonds are weak’
FALSE
Covalent bonds are strong
Which is smaller, an atom or a molecule?
An atom.
Simple molecules consist of atoms
joined by strong covalent bonds within
the molecule.
Why do ionic compounds have high
melting points?
Strong electrostatic attraction between the positive and negative ions requires alot of energy to overcome.
When do ionic compounds conduct electricity and why?
When molten or aqueous (dissolved in water) because the ions are charged and
free to move.
When solid, the ions are fixed in an ionic lattice so they can’t move.
Why do simple molecular compounds
have low melting and boiling points?
They have weak intermolecular forces (forces between molecules) which only
require a little energy to overcome.
Do simple molecular compounds
conduct electricity? Why / why not?
No because there are no charged
particles.
Do giant covalent structures have a high
melting point? Explain your answer.
Yes because they have lots of strong
covalent bonds which require a lot of
energy to break
How do metals conduct electricity and
heat?
The positive ions are fixed in a sea of delocalised electrons.
These electrons are free to move and carry charge/energy.
True or false?
‘Metals are insoluble in water’
TRUE
Name two giant covalent structures
formed from carbon atoms
Graphite
Diamond
Describe the structure of graphite
Each carbon atom bonded to 3 other carbon atoms.
Layers of hexagonal rings of carbon atoms.
Weak intermolecular forces between layers.
One delocalised electron per carbon atom.
Describe and explain the properties of
graphite
Graphite is soft / slippery because there are only weak intermolecular forces between layers which allow the layers to slide over one another.
Graphite conducts electricity because there is one delocalised electron per carbon atom. The delocalised electrons are mobile charges.
Describe the structure of diamond
All carbon atom are covalently bonded to four other carbon atoms.
No delocalised electrons.
Describe the properties of diamond
Very hard.
Very high melting point.
Doesn’t conduct electricity as there are
no charged particles.
What are the uses of graphite? Why?
Electrodes because graphite conducts
electricity and has a high melting point.
Lubricant because it’s slippery (the layers
in graphite can slide over each other).
Why is diamond used in cutting tools?
It’s very hard.
What is a fullerene?
A molecule made of carbon shaped like a closed tube or hollow ball
Name two fullerenes
Graphene
C60 (buckminsterfullerene)
What are the properties of the fullerene
C60?
- Slippery due to weak intermolecular forces.
- Low melting point.
- Spherical.
- Strong covalent bonds between carbon atoms in a molecule.
- Large surface area.
What are the properties of graphene?
-High melting point due to covalent
bonding between carbon atoms.
- Conducts electricity because it has
delocalised electrons.
Why is graphene useful in electronics?
It is extremely strong and has delocalised
electrons which are free to move and carry
charge.
It is only one atom thick as it is a single layer of
graphite.
What is a polymer? Name a polymer
Long chain molecules formed from many
monomers.
E.g. poly(ethene)
Draw the structure of poly(ethene)
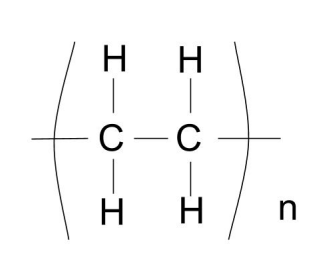
What bond is formed between carbon
atoms in polymer molecules?
Covalent bonds
What are the properties of metals?
- High melting point.
- Good conductors of electricity.
- High density.
- Malleable and ductile.
- Generally shiny.
Explain why metals are malleable
The atoms are arranged in uniform rows
which can slide over one another.
Explain why metals can conduct
electricity
The electrons in the metal are charges
that can move.
What are the properties of non-metals?
-Low boiling points.
- Brittle when solid.
- Poor conductors of electricity.
List the limitations of the following
models when representing ionic
compounds: dot and cross, 2D diagrams
and 3D diagrams
● Dot and cross - no lattice structure or ionic bonds.
● 2D diagrams - only shows one layer, doesn’t show
formation of ions.
● 3D diagrams - shows spaces between the ions,
doesn’t show charges.
List the limitations of the following
models when representing covalent
molecules: dot and cross and ball and
stick
Dot and cross - doesn’t show relative sizes of
atoms or intermolecular forces.
● Ball and stick - bonds shown as sticks rather
than forces, doesn’t show how covalent bonds
form.