Unit 1: Matter, Chemical Trends, and Chemical Bonding
5.0(1)
Studied by 3 peopleCard Sorting
1/148
Earn XP
Description and Tags
Last updated 9:32 PM on 9/7/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
149 Terms
1
New cards
atomic radius
one-half the distance between the nuclei of identical atoms that are bonded together (size of the atom)
2
New cards
Democritus (400 BC)
Greek philosopher that said all matter is made of tiny particles called "atomos" (Greek for invisible) or atoms
3
New cards
Aristotle
rejected Democritus's theory and believed that everything was composed of four elements earth, air, fire, water
4
New cards
John Dalton (1804)
Theorized that elements are composed of small, indivisible particles called atoms and that each unique
element is composed of a single type of atom
○ Atoms of one element are different from the atoms
of all other elements
○ During chemical reactions, atoms are combined,
separated, or rearranged into different ratios
element is composed of a single type of atom
○ Atoms of one element are different from the atoms
of all other elements
○ During chemical reactions, atoms are combined,
separated, or rearranged into different ratios
5
New cards
J.J. Thomson (1897)
-discovered negative particles in atoms called electrons
-"plum pudding" model electrons (negatively charged subatomic particles) are scattered about the atom like plums in a plum pudding.
- the "pudding" part of the atom was composed of positively charged material
-"plum pudding" model electrons (negatively charged subatomic particles) are scattered about the atom like plums in a plum pudding.
- the "pudding" part of the atom was composed of positively charged material
6
New cards
Thomson's Model 1897
An atom is a positively charged mass with negatively
charged particles embedded throughout
○ Negatively charged particles = electrons
○ Model known as the plum pudding model
First model to confirm that atoms are made up of
smaller subatomic particles
● Knew that atoms were neutral, so a positively-charged particle must also exist
charged particles embedded throughout
○ Negatively charged particles = electrons
○ Model known as the plum pudding model
First model to confirm that atoms are made up of
smaller subatomic particles
● Knew that atoms were neutral, so a positively-charged particle must also exist
7
New cards
Ernest Rutherford (1909)
● Performed the gold foil experiment
○ Shot positively charged alpha particles (He2+)
through a thin sheet of gold
○ Based on Thomson's model, most of the particles
should have gone straight through
○ Shot positively charged alpha particles (He2+)
through a thin sheet of gold
○ Based on Thomson's model, most of the particles
should have gone straight through
8
New cards
What did Rutherford discover?
Nucleus and protons (gold foil experiment)
9
New cards
how did rutherford discover the proton?
Gold foil experiment (while most alpha particles went through the foil, some were deflected at large angles - Rutherford theorized that the alpha particles were hitting something dense and positively-charged)
10
New cards
Planetary Model
electrons move around the nucleus in fixed, circular orbits
11
New cards
issues with Rutherford's model
According to classical physics, electrons orbiting the nucleus should
-lose energy (and collapse into the nucleus)
-emit light
-lose energy (and collapse into the nucleus)
-emit light
12
New cards
Niels Bohr (1913)
Experimented with applying electricity and thermal energy to hydrogen gas. he observed that the hydrogen atoms emitted light of only certain colors they were "excited"
13
New cards
Bohr's Model
electrons move in fixed in circular orbits around nucleus of an atoms (in definite energy levels)
14
New cards
Three subatomic particles
protons (p+), neutrons (n0), electrons (e-)
15
New cards
atomic number
the number of protons in the nucleus of an atom
16
New cards
mass number
the sum of the number of neutrons and protons in an atomic nucleus
17
New cards
nuclear symbol
the superscript indicates the mass number and the subscript indicates the atomic number
18
New cards
notation of an element
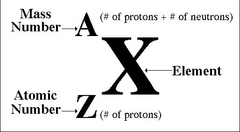
19
New cards
How to find the number of neutrons
mass number - atomic number
20
New cards
How to find the number of protons
same as atomic number
21
New cards
how to find the number of electrons
same as protons
22
New cards
different ways of writing the element
element name - mass # (ex. potassium-40) or element symbol - mass # (ex. K-40)
23
New cards
atomic mass unit (u)
a unit used to express the relative masses of atoms. One u is equal to 1/12 the mass of an atom of carbon-12
24
New cards
Isotopes
Atoms of the same element that have the same number of protons but different numbers of neutrons
25
New cards
difference between a isotope and a regular element?
isotopes have different masses, they have difference physical properties (some are radioactive due to the nuclei breaking down spontaneously)
26
New cards
how are isotopes written?
with their mass # shown (ex. Cl-37)
27
New cards
isotopic abundance
the percentage of a given isotope in a sample of an element
28
New cards
isotopic abundance formula
Atomic mass = (% abundance of isotope 1)(mass of isotope 1) + (% abundance of isotope 2)(mass of isotope 2)+...
29
New cards
radioactive decay
A spontaneous process in which unstable nuclei lose energy by emitting radiation
30
New cards
when do isotopes emit nuclear radiation?
when they are decaying
31
New cards
alpha particle
A cluster of 2 protons and 2 neutrons emitted from a nucleus in one type of radioactivity
32
New cards
beta particle
a negatively charged particle that is emitted from the nucleus of a radioactive atom
33
New cards
gamma ray
a form of high-energy electromagnetic radiation
34
New cards
Radioisotopes
Isotopes that have unstable nuclei and undergo radioactive decay.
35
New cards
valence electrons
Electrons on the outermost energy level of an atom
36
New cards
core electrons
The electrons in the inner shells of an atom; these electrons are not involved in forming bonds.
37
New cards
Cation
A positively charged ion
38
New cards
Anion
A negatively charged ion
39
New cards
Ion
An atom or group of atoms that has a positive or negative charge.
40
New cards
What electrons are involved in bonding?
valence electrons
41
New cards
where can you find valence electrons on the periodic table?
the last digit of the group number
42
New cards
what is the atomic radius of diatomic molecules?
the distance between the two nuclei divided by two
43
New cards
what is atomic radius measured in?
picometers (pm) (1 pm = 1 x 10^-12m)
44
New cards
what are the periodic trends for atomic radius?
atomic radius decreases left to right, it increases going down a group

45
New cards
what increases atomic radius?
more electrons means the electrons from the outermost shells will be further away
46
New cards
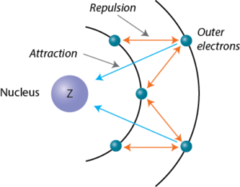
shielding or screening
a reduction in nuclear charge felt by an electron due to repulsions from other electrons in the same shell or in a smaller n-shell
47
New cards
effective nuclear charge (Zeff)
the net positive charge experienced by electrons in the valence shell and forms the foundation for all periodic trends
48
New cards
how do we calculate Zeff?
Zeff = Z - S (Zeff = effective nuclear charge, Z = atomic number, S = core electrons or the shielding constant)
49
New cards
where can we find the shielding constant?
by counting the number of core electrons (usually even, electrons without the valence electrons)
50
New cards
what are the smallest atoms in the respective periods?
noble gases
51
New cards
Why does atomic radius increase down a group?
increasing number of energy levels and the core electrons increases
52
New cards
why does atomic radius decrease left to right?
more effective nuclear charge is pulling e-, screening by core electrons is the same within the same energy level
53
New cards
ionic radius
Distance from the center of an ion's nucleus to its outermost electron
54
New cards
what is ionic radius measured in?
picometers
55
New cards
is a cation smaller or larger than their original atom?
smaller (the number of valence electrons decreases, this causes the # of electron orbits to decrease, the remaining electrons have a stronger attractive to the nucleus)
56
New cards
are anions smaller or larger than their original atom?
larger (when anions form, there is a higher electron to electron repulsion, making them spread out more)
57
New cards
ionization energy
The amount of energy required to REMOVE an electron from an atom (in kJ/mol)
58
New cards
the first ionization energy is given by (IE1)
X(g) -> X+(g) + e- (the g and the first plus sign is smaller)
59
New cards
the second ionization energy is given by (IE2)
X+(g) → X2+(g) + e-
60
New cards
the (number) ionization energy is given by (IEn)
X(n-1)+(g) → Xn+(g) + e- (every other number from 3)
61
New cards
do metals have a low ionization energy? how about non-metals?
metals have a low ionization energy, while non-metals have high ionization energies
62
New cards
do successive ionization energies increase or decrease, why?
increases, since you are removing electrons from already positively charged ions
63
New cards
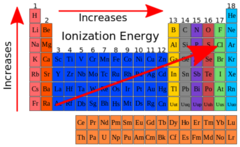
trends in ionization energies
increases from left to right a group, increases going down a group (opposite of atomic radius)
64
New cards
why does ionization energy increase from left to right?
the effective nuclear charge increases so atomic radius decreases, valence electrons are held more closely to the nucleus (these factors make it harder to remove a valence electron from the atom/ion)
65
New cards
why does ionization energy decrease going down a group?
the atomic radius increases due to increased shielding, the attraction between valence electrons and the nucleus decreases (these factors make it easier to remove a valence electron from the atom/ion)
66
New cards
electron affinity (Eea)
the energy change that occurs when an electron is ADDED to a neutral atom or ion in the gaseous state
67
New cards
general electron affinity equation
X(g) + e- → X- (g) + energy
68
New cards
what does it mean if the electron affinity value is negative?
this indicates that energy is released when the electron is added to the atom/ion (if attraction > repulsion -> energy < 0, if attraction < repulsion -> energy > 0)
69
New cards
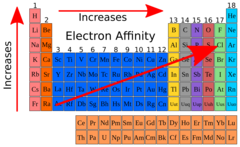
Trends in Electron Affinity
increases from left to right (number becomes more negative), decreases going down (number becomes more positive)
70
New cards
electron affinity increases from left to right because
effective nuclear charge increases so atomic radius decreases, the force of attraction between the nucleus and valence electrons increases (these factors mean that more energy is released when a new electron is acquired)
71
New cards
electron affinity decreases down a group because
atomic radius increases due to increased shielding, attraction between valence electrons and the nucleus decreases
72
New cards
Electronegativity
the ability of an atom to attract electrons in a covalent bond (A chemical bond formed when two atoms share electrons)
73
New cards
electronegativity increases left to right because
atomic radius decreases and effective nuclear charge increases so there is a stronger attraction for electrons
74
New cards
electronegativity decreases down a group because
atomic radius increases so atoms have a weaker attraction for electrons
75
New cards
periodic trends of electronegativity
it increases from left to right across a period and decreases from top to bottom in a group
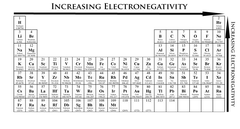
76
New cards
summary of all periodic trends
atomic radius (increases, left and down) ionization energy (increases, right and up), electron affinity (increases, right and up), electronegativity (increases right and down)
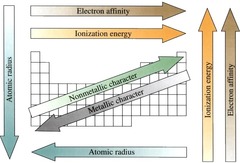
77
New cards
ionic compounds
composed of a metallic element combined with one or more non-metallic elements
78
New cards
ionic bond
an electrostatic force of attraction between a positive ion and a negative ion, DONATES its electrons (electrons are transferred from the metal atom(s) to non-metal atom(s), NON-METALS + METALS)
79
New cards
When do cations form?
When a neutral atom LOSES one or more electrons to form a positively charged ion (brackets around the element with a plus on the side + e-)
80
New cards
when do anions form?
when a neutral atom GAINS one or more electrons to form a negatively charged ion (brackets around the element with a minus sign on the side)
81
New cards
are anions smaller than original atoms or larger?
larger, there is more electrostatic repulsion between the negatively charged electrons

82
New cards
are cations smaller or larger than their original atoms?
smaller, the removal of electrons causes the electrons to come closer together since there is less electrons for electrostatic repulsion

83
New cards
Lewis symbols (dot structures)
the chemical symbol for an atom surrounded by one or more dots representing the valence electrons
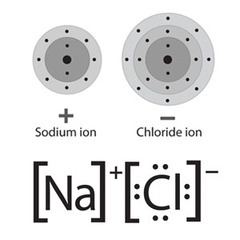
84
New cards
structure of ionic compounds
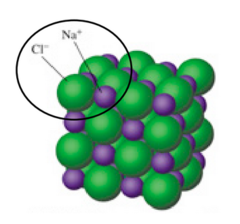
consists of alternating units of positive and negative ions in a rigid arrangement called a crystal lattice structure (no single unit of an ionic compound, they exist as a giant, endlessly repeating lattice of ions)
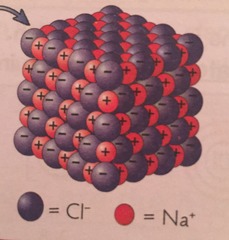
85
New cards
formula unit
the lowest whole-number ratio of ions in an ionic compound (tiny section of the repeating pattern)
86
New cards
properties of ionic compounds
- high melting and boiling points due to many strong bonds between the ions, since the attraction between ions of opposite charges
- many (but not all) are soluble in water (water molecules are able to pull apart the ions and separate them)
- conduct electricity as molten liquids (but not as solids) or when dissolved in water
- hard, but brittle
- many (but not all) are soluble in water (water molecules are able to pull apart the ions and separate them)
- conduct electricity as molten liquids (but not as solids) or when dissolved in water
- hard, but brittle
87
New cards
Why do ionic compounds conduct electricity only when molten or in solution?
in liquid form, ions are free-flowing and are able to carry electric charges
88
New cards
Electrolyte
An ionic compound whose aqueous solution conducts an electric current
89
New cards
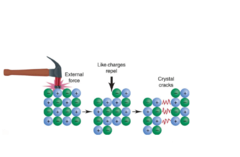
Why are ionic compounds brittle?
They are brittle because when the ions are forced out of position, like charges line up and cause repulsion and cracking of the ionic compound.
90
New cards
molecular elements
elements whose particles are multi-atom molecules
91
New cards
molecular compound
a pure substance composed of molecules made up of two or more non-metallic elements
92
New cards
properties of molecular substances
- can exist as gases, liquids, and solids at room temp. (solids can be soft, waxy, flexible, or hard)
- generally have low boiling and melting points
- poor conductors of electricity
- molecular elements are typically gases at room temp. due to their low boiling points, typically do no dissolve readily in water
- generally have low boiling and melting points
- poor conductors of electricity
- molecular elements are typically gases at room temp. due to their low boiling points, typically do no dissolve readily in water
93
New cards
covalent bond
a covalent bond forms when a pair(s) of electrons are SHARED by two atoms (usually occur between non-metals)
- The shared electrons are attracted to both nuclei, but simultaneously repelled by the other electron
- Each atom achieves a full valence shell and has a stable electron arrangement (like a noble gas)
- The shared electrons are attracted to both nuclei, but simultaneously repelled by the other electron
- Each atom achieves a full valence shell and has a stable electron arrangement (like a noble gas)
94
New cards
each atom in a covalent bond has a
bonding capacity (the number of covalent bonds that it can form)
95
New cards
bonding electron
an electron, in the valence shell of an atom, that is available to form a covalent bond with another atom
96
New cards
Three types of covalent bonds
single - 2e- (share one PAIR of electrons, double 4e-(share two PAIRS of electrons, triple 6e-(share three pairs of electrons)
97
New cards
lone pairs
pairs of valence electrons remaining that are not involved in covalent bond formation
98
New cards
octet rule
States that atoms lose, gain or share electrons in order to acquire a full set of 8 valence electrons
99
New cards
how can we represent molecules?
- lewis structures, lines represent a pair of bonding electrons, pairs of individual dots are used to represent lone pairs
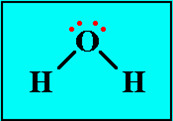
100
New cards
exceptions to the octet rule
atoms in period 3 and higher can have an expanded octet (valence shell with more than 8e-), beryllium and boron can have an incomplete octet (valence shell with less than 8e-)