chapter 8: peri-implant health and diseases
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
components of an implant system
implant, abutment, crown
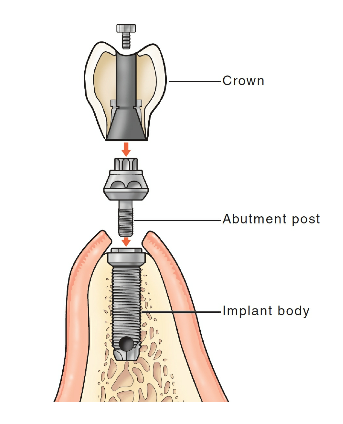
implant body
portion that is surgically placed into the living, vital alveolar bone
varying shapes and roughness provide greater surface area → engage peri-implant bone so there’s better stress distribution and lowering micromotion
why is titanium commonly used for conventional dental implants
titanium is a lightweight metal with high strength and durability
biocompatible, doesn’t readily corrode
poor conductor of heat and electricity
disadvantages of titanium for implants
softer and scratched easily by instruments → bacterial adherence
some report adverse reactions to titanium particles from corrosion/wear
grayish dark color that could shine through thin gingiva
zirconia for dental implants
ceramic material with similar/superior biocompatibility can characteristics to titanium
better esthetics (whitish color, high degree of translucency) with thin gingival phenotypes
implant abutment
metal post that connects the implant prosthesis to the implant body and secures the prosthesis in place
either protrudes partially or completely through the gingival tissue
also made from either titanium or zirconia
tissues surrounding a dental implant
junctional epithelium- attaches to the implant or abutment surface (biologic seal)
connective tissue (supragingival fiber bundles)- run parallel or obliquely to the implant/abutment or encircles it entirely
no pdl or cementum
alveolar bone is in direct contact with the implant surface (osteointegration)
implications of an implant having no pdl fibers
periodontal pathogens can create inflammation and destroy bone much more rapidly along a dental implant (no protective barrier)
osseointegration
the direct contact of the living bone with the surface of the implant body
success: no clinical mobility of the implant, no discomfort or pain when the implant is in function, no increased bone loss or radiolucency on rads, less than 0.2 mm of bone loss annually after the first year
peri-implant health
absence of erythema, BOP, swelling, and suppuration
no visual difference compared to healthy periodontal tissues
probing depths may still be deeper due to less resistance
peri-implant mucositis
plaque biofilm-induced inflammation of the soft tissues with no loss of supporting bone
reversible if etiologic factors are removed
includes red inflammation
peri-implantitis
periodontitis affecting the soft and hard tissues surrounding a functioning osseointegrated dental implant
plaque-biofilm inflammation with progressive loss of supporting alveolar bone
can be a nonlinear accelerated pattern of progression
implant does not become mobile until the final stages (loss of osseointegration)
factors contributing to hard tissue deficiencies
loss of periodontal support caused by periodontitis, endodontic infections, thin buccal bone plates, traumatic injury
primary etiology of peri-implant mucositis and peri-implantitis
dental biofilm
is there any single microorganism that has ever been implicated to be the causative agent of peri-implantitis
no, it is a polymicrobial infection
risk factors of peri-implantitis
history of previous periodontal disease
poor plaque biofilm control
smoking
residual cement may induce inflammation
biomechanical overload (forces and duration of them on the tooth) (can also lead to implant failure even w/o plaque)
clinical signs of a failing implant
soft tissue indicators: BOP, suppuration, pain usually not present
implant mobility (loss of osseointegration)
radiographic signs of a failing implant
vertical destruction of the crestal bone around the implant- assumes shape of a saucer
treatment modalities for failing implants
NSPT, anti-septics, local/systemic antibodies, access flap surgery
when should probing of the implant occur
once the final restoration has been installed
use a weak hand
probing depths alone < changes in probing depths over time