Bonding ✅
1/178
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
179 Terms
Why do chemical bonds form?
Electrons are transferred (ionic) or shared (covalent) so the atom can achieve a full outer shell of electrons as this is more energetically stable.
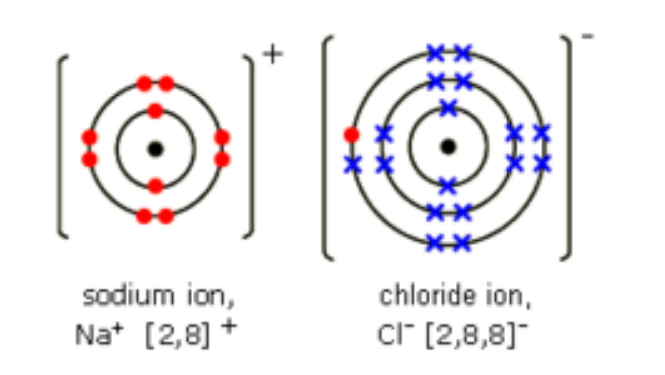
What is ionic bonding?
It is the electrostatic attraction between oppositely charged ions.
What type of elements form ionic compounds?
- When metals in react with non-metals.
- Mainly metals in groups 1 and 2 reacting with
non-metals in groups 6-7 but aluminium will form
ionic compounds
How does ionic bonding occur?
- The metal atom loses electrons (oxidation) to
become a positively charged ion.
- These are gained by the non-metal (reduction)
which becomes a negatively charged ion.
- The ions attract.
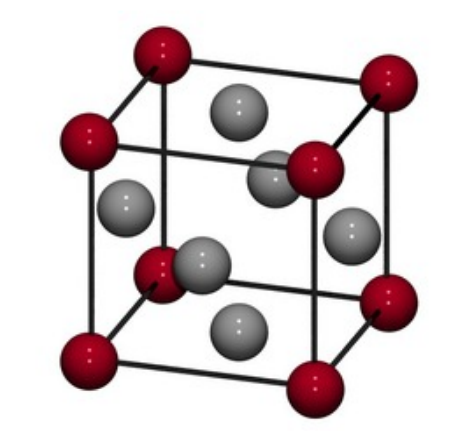
Describe the structure of ionic compounds.
- The ions formed in each formula unit attract each
other to form a giant ionic lattice, in a crystal structure.
- Giant because the same basic structure is repeated lots of times
- The attraction extends throughout the molecule,
all +ve ions attracted to all -ve ions
- Alternating +ve and -ve ions
Are ionic bonds strong or weak?
Strong
Explain the properties of ionic solids
- High melting points due to lots of strong electrostatic forces of attraction between oppositely
charged ions in a giant ionic lattice, require lots of
energy to break.
- Don't conduct electricity as solids, as the ions are
not free to move.
- As liquids or dissolved in water, they will conduct
electricity as the ions are free to move and carry
charge through the substance.
- Soluble (dissolve well)
Formations of ions in ionic bonding: Sodium chloride.
Draw the 3d structure of an ionic lattice
What 2 factors affect the strength of ionic bonding?
- Atomic radius
- Charge on ions
How does atomic radius affect the strength of an ionic bond?
- Smaller ions means stronger ionic bonding.
- Ions are packed more closely together in a
lattice.
- Shorter distance between oppositely charged
ions meaning electrostatic forces are stronger.
- Ions bigger down group, more shells, more
shielding down the group so weaker bonding
- Ions smaller across period as they have a
higher nuclear charge
How does the charge on ions affect the strength of an ionic bond?
- Higher charge, stronger bonding
- Ions exert a greater force into each other
What is the overall charge of an ionic compound?
0
Explain how caesium oxide forms and give the half equations
• 2 Caesium atoms each lose 1 electron to become Cs+ Cs → Cs+ + e⁻
• Oxygen gains 2 electrons to become O²⁻ O + 2e⁻ → O²⁻
Explain why ionic compounds are brittle
- Applying force disrupts regular lattice / arrangement of ions
- Results in ions with like charges next to each
other
- Ions with like charges repel
Explain why K⁺ ions are smaller than Cl⁻ ions.
- K⁺ and Cl⁻ are isoelectronic (same number of electrons)
- K⁺ has greater nuclear charge / more protons / electrons more strongly attracted to nucleus
Give the charges of: Silver, zinc, iron, copper, lead (cations)
Silver: Ag⁺
Zinc: Zn²⁺
Iron: Fe²⁺ / Fe³⁺
Copper: Cu⁺ / Cu²⁺
Lead: Pb²⁺ / Pb⁴⁺
How do we name anions? What charges do they have?
- Take prefix and add 'ide' e.g. oxide, phosphide,
selenide
- Groups 5,6,7: 3- , 2- , 1- as each gain(s) electrons
What state are anions they in?
Gaseous
Give the half equation for the formation of a hydrogen ion.
H - e⁻ → H+
What is a polyatomic ion?
Ion made of more than 1 atom
Name the following polyatomic ions:
HCO³⁻
SO₄²⁻
PO₄³
MnO₄⁻
OH⁻
HCO³⁻ hydrogencarbonate
SO₄²⁻ sulfate
PO₄³⁻ phosphate
MnO₄⁻ manganate
OH⁻ hydroxide
Give the formula for an ammonium ion
NH₄⁺
What are isoelectronic ions?
Ions of different atoms with the same number of electrons
Predict whether lithium chloride or potassium iodide has a higher melting point.
Lithium chloride has a higher melting point
- Lithium's cation is smaller than potassium's
cation
- Chloride is smaller than iodide
- Lithium chloride has stronger ionic bonding than
potassium iodide
The electrical conductivity of NH₄Cl (aq) is much greater than the electrical conductivity of NH₃ (aq).
Explain the difference in electrical conductivity of these solutions
- NH₄Cl (aq) contains (NH₄⁺ and Cl⁻) ions
- Only ions can carry charge
Suggest why it is difficult to predict whether sodium iodide has a greater melting point than potassium bromide
- Na+ and K+ have the same charge.
- I− and Br− also have the same charge.
- Therefore, we must consider the sizes of the
ions.
Na+ ions are smaller than K+ ions, which suggests
that NaI has a higher melting point.
- However, Br− ions are smaller than I− ions, which
suggests that KBr has the higher melting point
What do we call the ionic compound containing Pb⁴⁺ ions and Cl⁻ ions ?
We call it lead (IV) chloride (Lead four chloride)
What ratio of caesium to oxygen atoms is there in caesium oxide?
2:1 2 caesium to 1 oxygen, as charges must cancel out.
What is the empirical formula of sodium sulphate?
- Sodium ions have a 1+ charge
- Sulphate ions have a 2− charge.
- So, to make a neutral ionic compound, these ions
must combine in a 2 : 1 ratio
What is covalent bonding?
- 2 non-metals share electrons
- To achieve a full outer shell
What are the bonds for molecules which form covalent bonds?
- Can have a double/triple covalent bond where 2/3 electrons are shared
Which diagrams show covalent bonds?
- Stick diagrams
- Dot cross diagrams
How do we draw a covalent bond?
- For a single covalent bond, we do a dot and a
cross to show that the electrons have come from
different atoms.
- When a double covalent bond forms there are 2 shared pairs of electrons. This has to be drawn dot,
cross, dot, cross to show that each pair of electrons
has one electron from each atom.
- A triple covalent bond has to be drawn dot, cross,
dot, cross, dot, cross for the same reason.
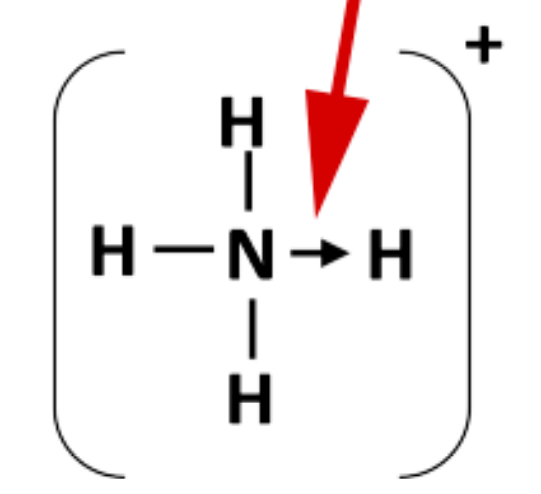
What is a co-ordinate/dative bond?
- A covalent bond formed when one atom shares 2 of its electrons with another atom that is electron deficient. - It is often drawn as an arrow, or a pair of either dots/crosses
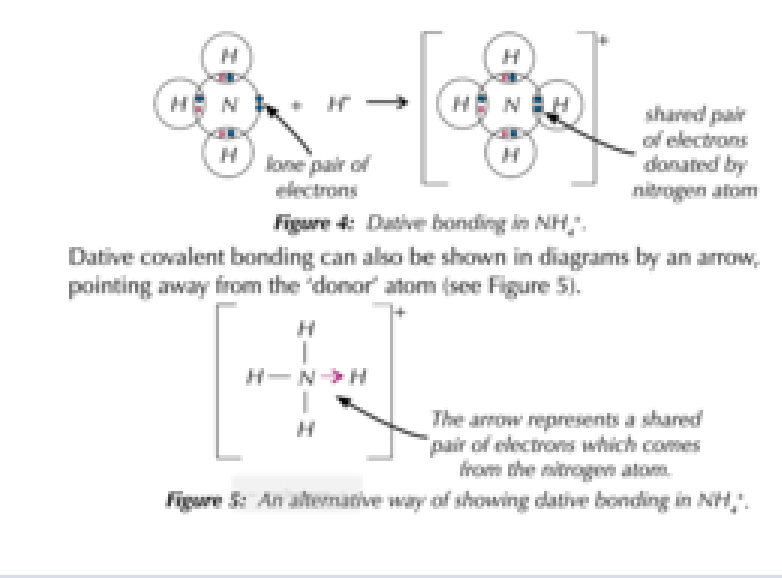
In what types of substances can covalent bonding exist?
- Covalent bonding occurs in small and long chain molecules such as water, methane, carbon dioxide and polymers. - It can also occur in giant lattices e.g. graphite, diamond and silicon dioxide.
What are small molecules?
- 2 or more uncharged atoms bonded together
e.g. Cl₂, H₂O, CH₂OH.
- Molecules have covalent bonds between atoms
to hold them together
- Between molecules are intermolecular forces
which are the bonds that need breaking to
melt/boil the compound.
Do substances with metallic or ionic bonding contain molecules?
NO
What are the four main types of crystal structures?
Ionic
Metallic
Macromolecular (giant covalent)
Molecular (ice, iodine)
Suggest why magnesium is a liquid over a much greater temperature range compared to bromine.
Mg has a much greater liquid range because forces of attraction in liquid / molten metal are strong(er) OR converse argument for Br2
How do we represent molecules with: a) ionic/metallic bonding b) covalent
a) Empirical formula, as the behaviour of the substance ISN'T affected by the exact number of ions.
b) Molecular formula, as the behaviour of the substance IS affected by the exact number of ions. e.g.
O₂ and O₃ are different
How do we name molecules made of 1 element?
Reference the number of atoms in the molecule using a prefix. e.g. F₂ is difluorine, O₃ is trioxygen, S₈ is octasulfur.
How do we name molecules made of 2 elements?
1. Name the 1st element in the molecular formula, followed by the 2nd element 2. Change the ending of the 2nd element to 'ide'. 3. Use prefixes to represent how many atoms of each element are present in the molecule. e.g. P₂I₄ ’ diphosphorus tetraiodide
Are these naming systems always used?
No. e.g. O₃ is mostly called "ozone" rather than "trioxygen" and NH₃ is mostly called "ammonia" rather than "nitrogen trihydride"
Which type of covalent bond is the strongest?
The shortest - so the triple bond
What are the properties of small molecules?
- Low melting and boiling points as there are weak
intermolecular forces between the molecules
- that doesn't take a lot of energy to overcome them. (The molecules stay intact when they
melt/boil).
What type(s) of bonding does NH₄Cl (s) contain?
- Ionic bonding between NH₄⁺ and Cl⁻ ions - In NH₄⁺ there are 3 covalent N-H bonds and 1 dative covalent bond between the nitrogen's lone pair and a H⁺ ion
What type(s) of bonding does KOH(s) contain?
There is ionic bonding between K⁺ and OH⁻ ions In OH⁻ there is a covalent O-H bond.
What group is hydrogen in?
Group 1
How do we draw a dot cross diagram for a polyatomic ion?
- Start by drawing all the atoms out, showing their
outer shell electrons
- Next, add electrons the atoms to reflect the
charge of the polyatomic ion:
- If the ion has a -ve charge then add electron(s)
to/from the most electronegative atom(s).
- If the ion has a +ve charge then remove electron(s) to/from the least electronegative atom(s). - Pair up resulting atoms and ions until they all have full outer shells. - Label with dots and crosses.
What are giant covalent structures?
- Huge network of covalently bonded atoms. - Carbon forms these structures as it can form 4 strong, covalent bonds. - Contain more than 1000 atoms
Macromolecules
A giant molecule consisting of thousands of atoms which are held together by covalent bonds.
What are chain macromolecules, sheet macromolecules, and 3D macromolecules held together by?
- chain macromolecules = intermolecular forces
- sheet macromolecules = intermolecular forces
- 3D macromolecules = covalent bonds
. What is an allotrope?
Structures where the atoms of the same element bond in different ways.
How do we represent macromolecules and simple molecules?
macromolecules = empirical formula simple molecules = molecular formula, as number of atoms affects it's properties
Describe the melting/boiling point of diamond.
High melting point —> lots of strong covalent bonds between carbon atoms in a giant lattice, require a lot of energy to break.
Describe the structure of diamond.
- Tetrahedral shape as each carbon atom is covalently bonded to 4 other carbon atoms
- Bond angle 109.5°
- All 4 carbon atoms used in bonding
Describe the conductivity of diamond.
Will not conduct electricity as there are no delocalised electrons to flow throughout structure to carry the charge.
Describe the physical properties of diamond.
- Hard, due to the closeness and rigidity of the
covalent bonds.
- Good thermal conductor, as vibrations easily
travel through the stiff lattice.
- Insoluble, as the covalent bonds are too strong
to break.
Describe the structure of graphite.
- Carbon atoms are arranged in sheets of hexagonal layers
- Each carbon bonding to 3 others. The 4th outer
electron of each carbon is delocalised
- Sheets of hexagons are connected with weak van
der waals.
Describe the melting/boiling point of graphite.
High melting point —> lots of strong covalent bonds in a giant lattice, as well as weak IMF, require a lot of energy to break.
Describe the conductivity of graphite
Contains delocalised electrons, one from each atom, that can move and carry charge throughout the structure.
Describe the physical properties of graphite.
- Slippery as the weak bonds between layers are
easily broken so the layers can slide.
- Low density as the layers are far apart compared to the length of the covalent bonds.
- Insoluble as covalent bonds are too strong to
break.
Used in pencils, are dry lubricants and strong, lightweight sports equipment.
Is silicon (Si) a macromolecular lattice?
Yes
Explain these structures of diamond and graphite.
- The black circles are carbon atoms
- The sticks between the carbon atoms are covalent
bonds.
- In graphite, there are dotted lines between the
layers. These are intermolecular forces.
What shape and bond angle is in diamond and graphite?
Diamond - tetrahedral 109.5° Graphite - triangular planar 120°
What is a metallic bond?
- In a metallic structure, the outer electrons are lost
from each ion and become delocalised throughout
the structure.
- The atoms therefore become positively charged
ions.
- The metallic bond is the electrostatic attraction
of the positively charged ions to the sea of delocalised electrons.
Give the structure of metals.
- Metals exist as giant lattice structures.
- The outermost electron on a metal atom is delocalised as is free to move.
- This leaves positive metal ions.
- The positive metal ions are attracted to the delocalised negative electrons.
- The positive ions are packed closely together in a
sea of delocalised electrons.
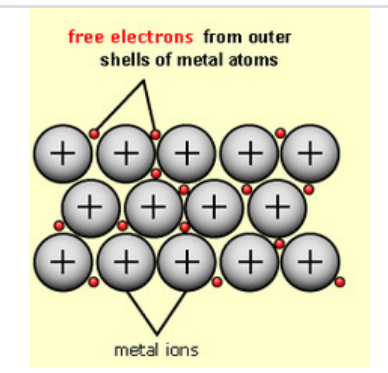
Explain the properties of metals
- Good conductors of electricity as the delocalised electrons can move and carry charge
through the structure.
- Good conductors of heat as the electrons can
carry energy as they move through the structure.
The metals ions increasingly vibrate as they are
heated so this transfers heat energy from one
ion to the next as they are close together.
- High melting points as there are lots of strong
metallic bonds in a giant lattice structure that
require a lot of energy to break
Why does the strength of metals vary?
1. The greater the charge on the metal ion, the
greater the number of delocalised electrons and
so the greater is the attraction between the ion
and the sea of delocalised electrons and so the
stronger the metal. (As you go from group 1 to
group 3 there is an increasing charge on the ion
and more delocalised electrons)
2. The smaller the ion, the closer the electrons
are to the positive nucleus and the stronger is the
metallic bond.
What are the trends of metals down the groups?
- Down a group the melting point of metals decrease,
- As the size of the cation increases,
- So the distance between cations (nucleus) and delocalised electrons increases, - So the strength of metallic bonding decreases
Why do metals have high melting points?
- The greater the charge on the ion, the greater the
attraction between the ion and the sea of electrons
and more energy is needed to break the electrostatic bond. Stronger metallic bonding.
- As there are more delocalised electrons and the
charge on the ion is greater as you go from groups
1-3, the melting points get higher.
Explain why aluminium has a higher melting point than magnesium.
- Al³⁺ ions are more highly charged than Mg²⁺
ions
- Aluminium delocalises more electrons per ion
than magnesium
- Al³⁺ Is smaller than the Mg²⁺, so there is a
stronger attraction between the aluminium cations
and the delocalised electrons, so the metallic
bonding in aluminium is stronger than that in
magnesium, so more energy is required to melt
aluminium.
Why are metals malleable and ductile?
- Metals are malleable (can be hammered into
shape)
- ductile (pulled into wires) as the atoms can slide
over each other retaining the electrostatic attraction.
For every Mg²⁺ cation in a lump of magnesium, there must also be how many delocalised electrons?
2.
- The lump of magnesium has no charge.
- This means the positive charge of the Mg²⁺ cations must be balanced by the negative charges
of the delocalised electrons (e−)
- This means there must be 2 delocalised electrons
per Mg²⁺ ion in the metal.
Metals in the liquid state...
Conduct electricity
Have metallic bonding
Metals in the gaseous state...
Have metallic atoms.
DO NOT conduct, no lattice of cations, no delocalised electrons.
What is a molecular lattice?
- Molecular lattices are made up of molecules with
less than 1000 atoms
- Held together by intermolecular forces (+ covalent bonding)
- Variable solubility but usually low
What is a macromolecular lattice?
- Macromolecular lattices are made up of molecules with more than 1000 atoms.
- In general, macromolecular lattices are held together by covalent bonds.
4 types of crystal structure
Ionic: NaCl
Metallic: Mg, Na
Molecular: I₂, Ice,
Macromolecular (giant covalent): diamond,
graphite
See slide to see the structure of each.
Describe particles in a solid.
- Particles close together giving it a high density
and incompressible.
- Particles vibrate in a fixed position
Describe particles in a liquid.
- Similar density to a solid so cannot be compressed
- Particles move freely and randomly (Brownian motion) meaning a liquid can flow.
Describe particles in a gas.
- Particles have a lot more kinetic energy so move
around more.
- Low density so can be compressed.
- It flows as particles move freely and randomly
(Brownian motion) with little attraction.
- Diffuse to fill a container
What do you call a change of state from a solid to a liquid?
Melting/fusion
What do you call a change of state from a liquid to a gas?
Boiling/vaporisation
What are crystals? Give examples.
They are solids with regular structures. E.g. ionic solids, giant covalent lattices, metals.
What is electronegativity?
The power of an atom to attract the electron density in a covalent bond toward itself
What is meant by electron density?
- Electron density is when we consider the electrons as charge clouds.
- A charge cloud is a region of space where there
is a pair electrons.
What is the Pauling scale?
- Used as a measure of electronegativity.
- Numbers range from 0-4 depending on how
electronegative elements are. Bigger number =
more electronegative.
What does electronegativity depend on?
Atomic radius
Shielding
Nuclear charg
Explain what affects electronegativity
. ATOMIC RADIUS
• Smaller atomic radius = higher electronegativity.
• Stronger attraction between electrons + nucleus.
SHIELDING
• More shielding = lower electronegativity.
• Less shells better as will be a stronger attraction
between electrons + nucleus.
NUCLEAR CHARGE
• Higher nuclear charge = higher electronegativity.
• More protons nucleus, creates stronger attraction with the electrons.
• Also decreases atomic radius.
Why does nuclear charge affect electronegativity?
• The higher the nuclear charge, the higher the
electronegativity.
• More protons in the nucleus create a stronger
attraction with the electrons.
• A higher nuclear charge also decreases atomic
radius.
What are the trends in electronegativity?
INCREASES ACROSS A PERIOD
- Bigger nuclear charge as there are more protons
in the nucleus but same amount shells, so atomic
radius decreases.
DECREASES DOWN A GROUP
- Atomic radius increases, distance between electrons and nucleus increases.
Shielding increases.
- Less attraction.
What are the most electronegative elements?
- Fluorine, Oxygen, Nitrogen, Chlorine, Bromine
(FONClBr or NOFClBr)
- Can be S and P too, see this in molecules inside
living things
What bonding is present in a compound containing elements of similar electronegativity ?
Small electronegativity difference
So purely covalent
What bonding is present in a compound containing elements of very different electronegativity ?
Very large electronegativity difference ionic
What are the bonding trends in electronegative compounds (ionic or covalent)?
- Compounds whose elements have difference in
electronegativity bigger than or equal to 2.5 tend
to bond ionically.
- Smaller than 2.5 bonds covalently.
What are the bonding trends in electronegative compounds (metallic or covalent)?
- Elements with electronegativities more than or
equal to 2.1 tend to bond covalently
- Elements with electronegativities less than or
equal to 1.5 tend to bond metallically
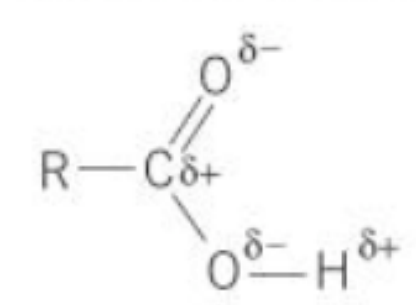
What is polarity?
- Polarity is the unequal sharing of electrons in
a covalent bond
- It is a property of the bond and not the molecule.
- This is a covalent molecule but we say it has a
bit of ionic character.
Explain when polarity occurs and in what type of atoms
- Occurs in covalent bond between 2 atoms with
different electronegativities, so unequal sharing of
electrons
- Electrons are more attracted to more electronegative atom.
- The less electronegative element becomes δ+ The
more becomes δ-
- Bigger difference in electronegativity = more polar
- Bonds must be on one side rather than symmetrical
- E.g. H δ₊ -- Cl δ