Fractional distillation of crude oil
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
What is the world’s main source of organic chemicals?
Crude oil
What is crude oil mostly a mixture of?
Alkanes - unbranched and branched
Why is crude oil as a mixture not useful?
It contains various impurities and different hydrocarbon compounds with different boiling points. This makes it difficult to use directly as a fuel or for other purposes.
How is the crude oil mixture separated?
Into fractions based on boiling points
What is the process of separating crude oil called?
Fractional distillation
What type of process is fractional distillation?
Physical, not chemical
Where does fractional distillation take place?
In a fractioning tower
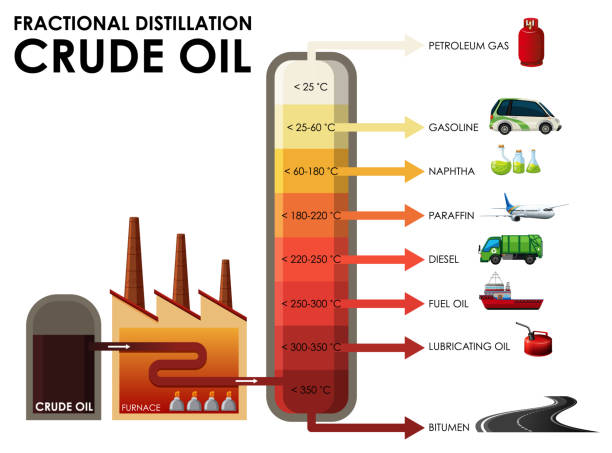
What are the temperatures of a fractionating column like?
Very hot at the bottom and cool at the top
When the crude oil is heated, what happens to the vapours of hydrocarbons with very high boiling points?
They will immediately condense into liquid at the higher temperatures lower down the column and are tapped off at the bottom.
What happens to the vapours of hydrocarbons with low boiling points?
They will rise up the column and condense at the top to be tapped off
Where are the fractions containing small and big hydrocarbons collected from on the fractionating column?
The fractions containing smaller hydrocarbons are collected at the top of the fractionating column as gases.
The fractions containing bigger hydrocarbons are collected at the lower sections of the fractionating column
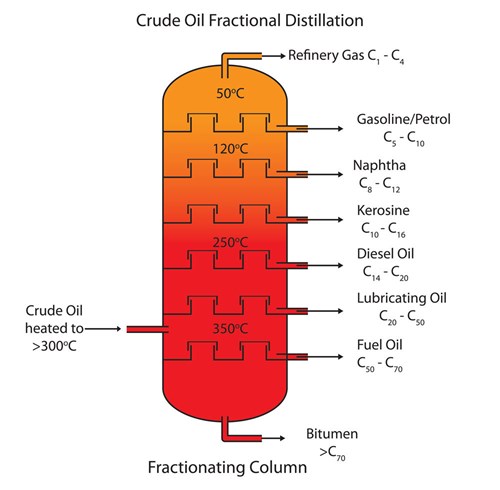
Does crude oil just contain hydrocarbons?
No it contains small amounts of other compounds
What about alkanes are important for predicting their properties?
Their chain length
The fractions that get tapped off at different heights are likely to have…
similar properties
Properties of shorter-chain alkanes compared to longer-chain alkanes
Lower boiling points
More volatile
More flammable
Less viscous (more runny)
Less coloured - bitumen is black compared to gasoline which is a pale yellow
What does volatile mean?
A compound that easily evaporates at normal temperatures - changes from a liquid or solid state to a gas quickly.