topic 18 organic chem 3
1/101
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
102 Terms
define benzene and give its formula
- simplest aromatic cyclic hydrocarbon
- C6H6
give 3 properties of benzene in terms of its physical and chemical properties
- planar ring
state the two models that can be used to represent benzene
a) Kekulé model
b) delocalied model
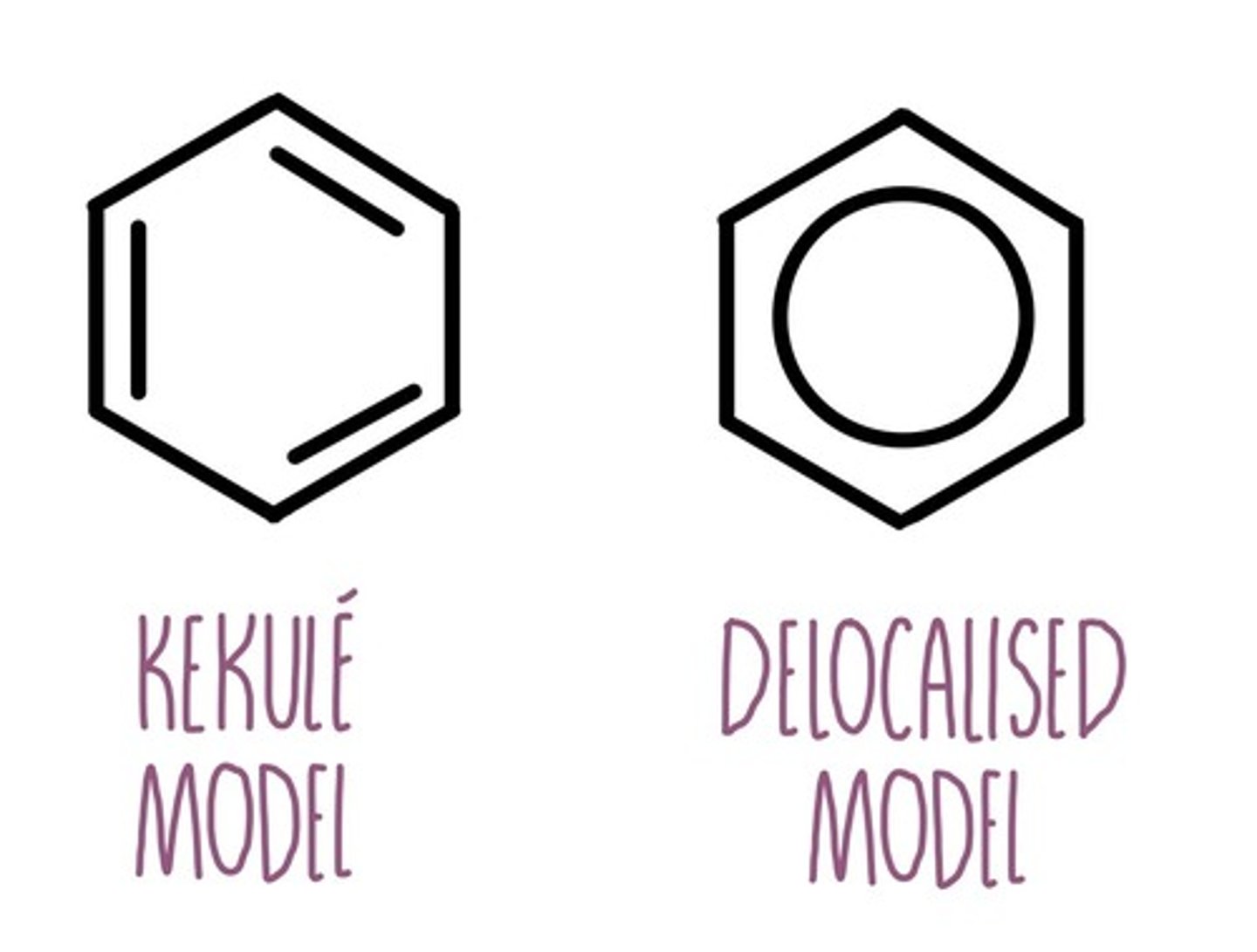
Kekule model for benzene
planar ring with C atoms with alternating single and double bonds
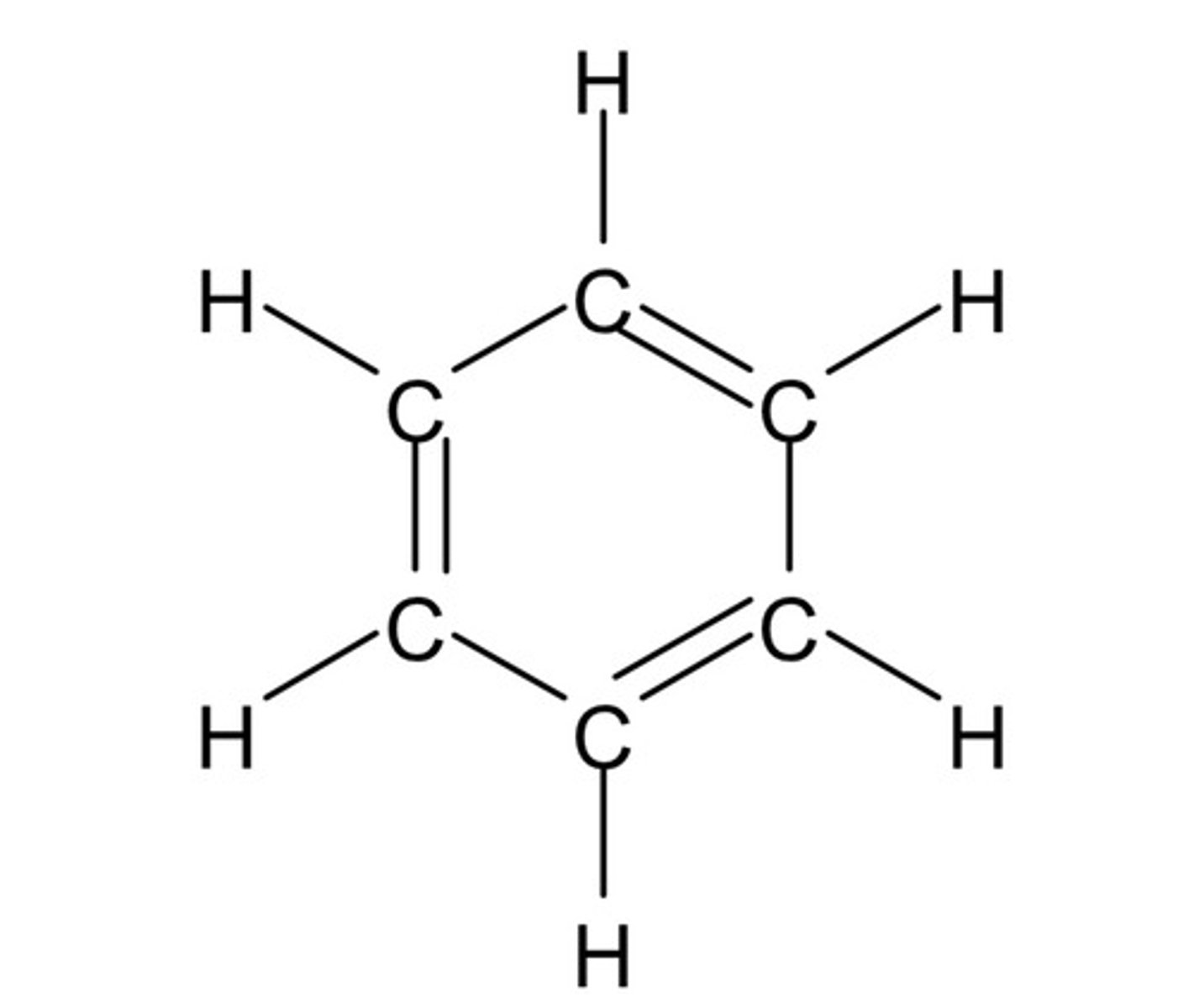
limitations of Kekule's model: benzene is a perfectly regular hexagon
- due to the difference length of C-C and C=C, this would mean the hexagon is irregular if it had the Kekulé structure
- however benzene is actually a perfectly regular hexagon
limitations of Kekule's model: enthalpy data
- theoretically if there were 3 C=C then the enthalpy value should be 3x (-360 kJ mol-1) as much as 1 C=C (-120 kJ mol-1)
- however the real amount of energy for benzene is only -208 kJ mol-1, suggesting that the 6 pi electrons are delocalised
limitations of Kekule's model: infrared data using spectroscopy
- all bond lengths between carbon atoms in benzene are the same
- Kekule model suggest that there would be 3 bonds that are double and 3 that are single carbon donds
- however there was no peak at C=C for benzene
limitations of Kekule's model: benzene only has 3 isomers
- Kekule's model suggests that 2-R-benzene should have 4 isomers
- but in reality it only has 3 (delocalised model)
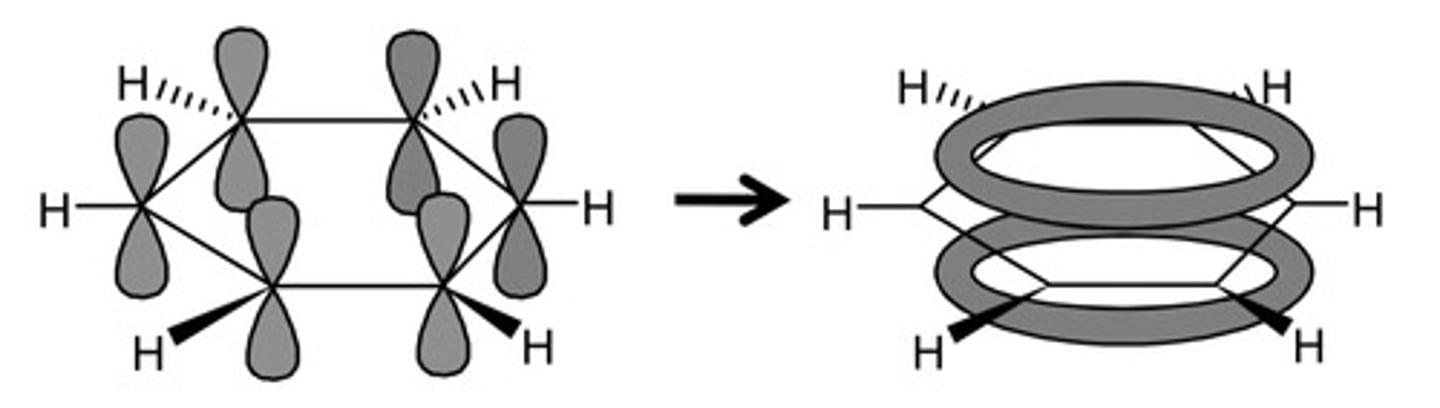
delocalised model for benzene
- each C atom form 3 σ-bonds
- the unused electron in the p orbital form a ring structure above and below the carbon atoms
- known as delocalised since the 6 electrons in the pi bond are spread out over the ring
draw the isomers of 2-bromobenzene using both the kekule and delocalised model in order to explain the limitation of the kekule model
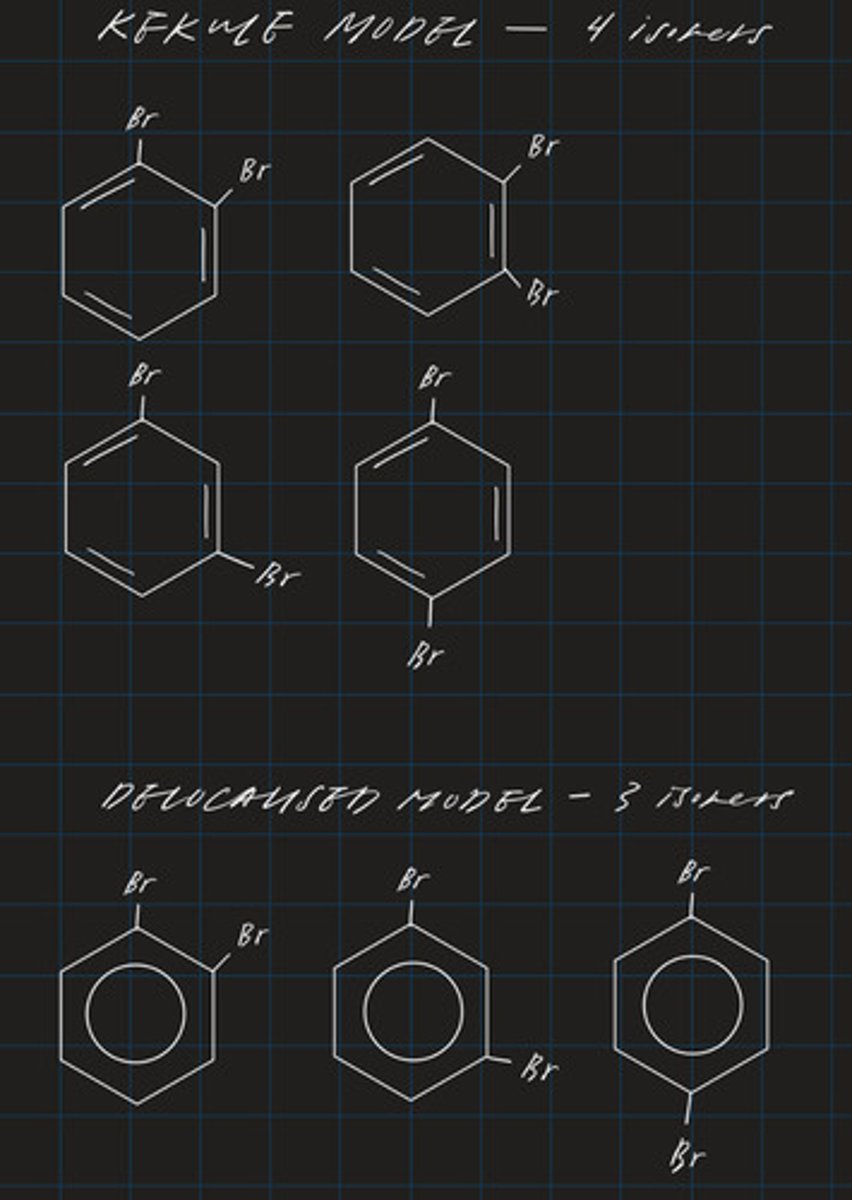
evidence for the delocalised structure of benzene includes
∙ only one structure for 1,2-dichlorobenzene
∙ analysis of X-ray diffraction images of benzene
explain why each of these makes the delocalised structure more likely to be correct than the Kekule model (2)
- isomers of dichlorobenzene should exist according to Kekule model where one has a single bond between C atoms bonded to Cl but it doesn't
- X-ray suggest that benzene is a regular hexagon where all bonds are the same length
explain why benzene is more stable than alkene against electrophilic attack
- benzene does not have a concentrated area of high electron density
- electrons are distributed across its ring structure
- so benzene is kinetically stable
write an equation for the nitration of benzene with nitric acid
C₆H₆ + HNO₃ -> C₆H₅NO₂ + H₂O
state the condition and catalyst required for the nitration of benzene
- concentrated sulphuric acid (react with nitric acid to form NO₂+)
- concentrated nitric acid
- at 55°C
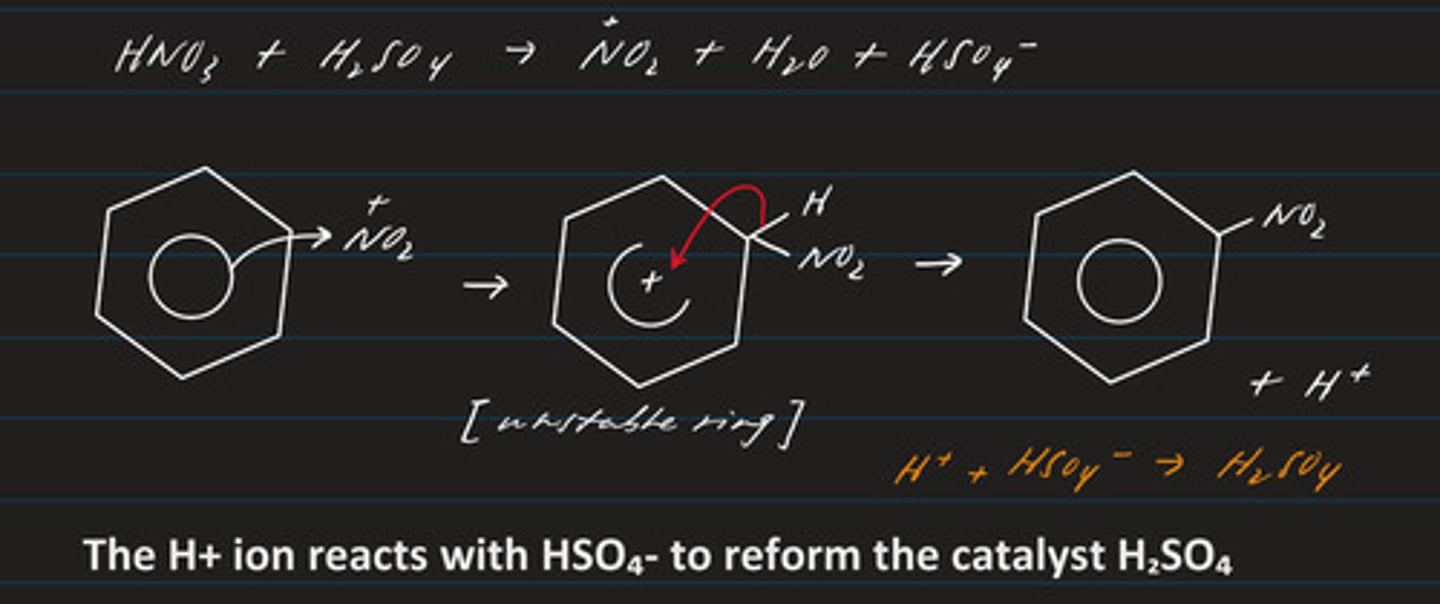
write an equation for the generation of the electrophile NO₂+ (sulphuric acid + nitric acid)
2H₂SO₄ + HNO₃ ⇄ 2HSO₄⁻ + H₃O+ + NO₂+
draw the mechanism regarding the nitration of benzene
- electrophilic substitution (electrophile: NO₂+)
- there is a substitution of hydrogen
- product: nitrobenzene
explain why the nitration of benzene must be done below 60°C
higher temp cause a second nitro group to be substituted onto a different position on the ring (2, 4, 6)
benzene reacts with a nitrating mixture of concentrated nitric and sulfuric acids. which species is least likely to be present in the nitrating mixture?
A. NO₃⁻
B. H₃O⁺
C. HSO₄⁻
D. NO₂⁺
A: NO₃⁻
write an equation for the halogenation of benzene with Cl₂
C₆H₆ + Cl₂ → C₆H₅Cl + HCl
state the conditions and catalyst required for the halogenation of benzene
- catalyst AlCl₃ / FeBr₃ (halogen carrier since chlorine is non polar)
- rtp
write an equation for the generation of the electrophile Cl⁺ (chlorine + aluminium chloride)
Cl₂ + AlCl₃ → AlCl₄⁻ + Cl⁺
draw the mechanism regarding the chlorination of benzene
- electrophilic substitution (electrophile: Cl⁺)
- product: chlorobenzene
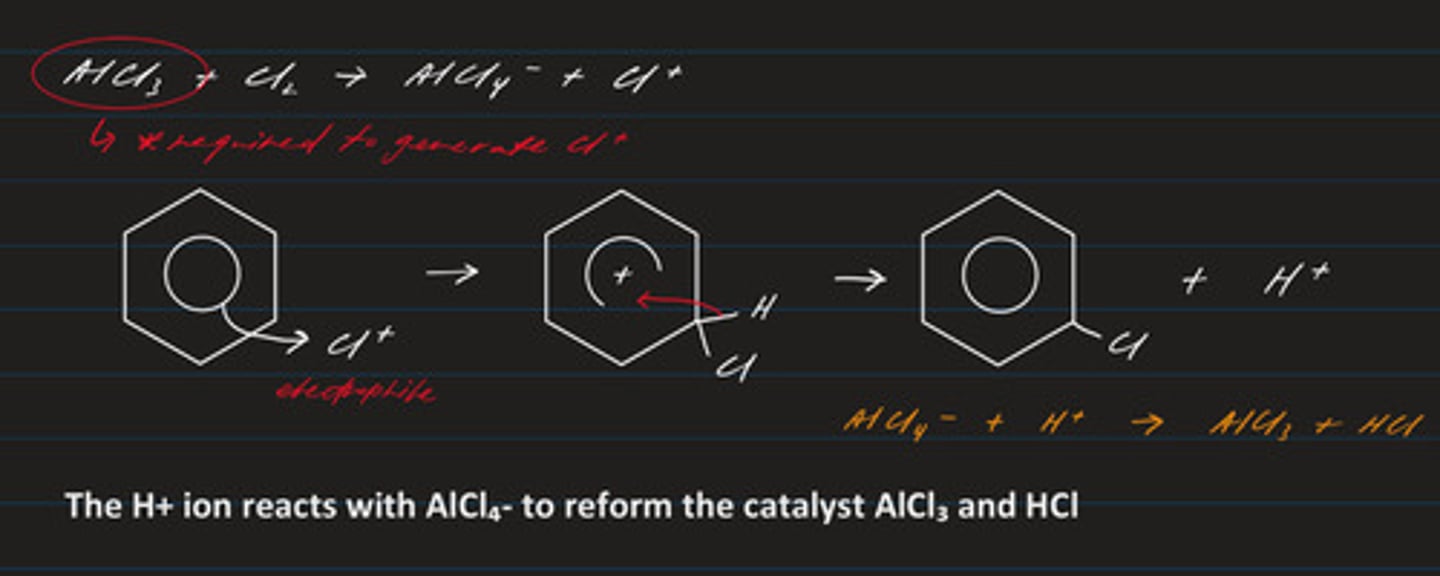
write the equation regarding the formation of the electrophile required in the mechanism for the chlorination (halogenation) of benzene
AlCl₃ + Cl₂ → AlCl₄⁻ + Cl⁺
state the condition and catalyst required for the hydrogenation of benzene
- nickel catalyst
- 200C, 30atm
write a displayed formula for the hydrogenation of benzene
- addition and reduction
- product: cyclohexane
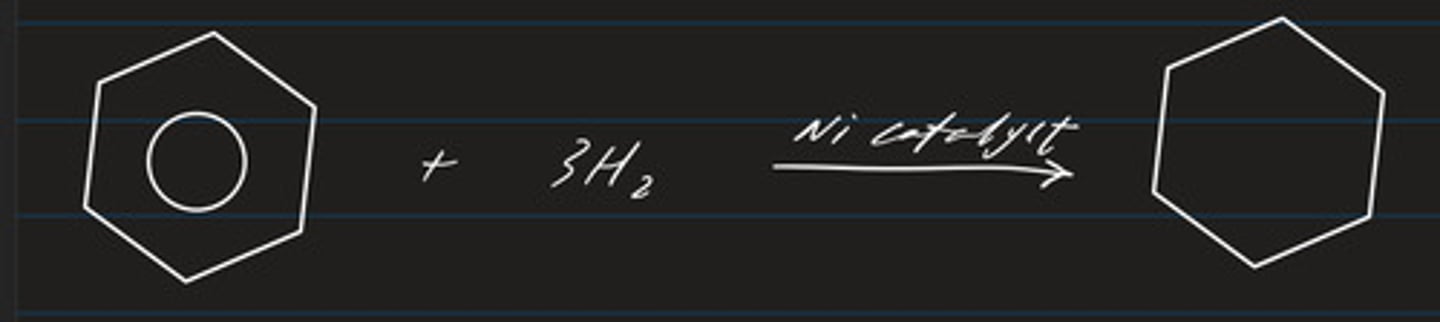
explain why a phenol is more reactive than a benzene
- phenols contain a hydroxyl -OH group
- lone pair of electrons on oyxgen overlap with the pi-bonding system in the benzene ring (high electron density)
- activates the benzene ring, making it more reactive
- more susceptible to electrophilic attacks
write an equation for the acylation of benzene with an acyl chloride
C₆H₆ + CH₃COCl → C₆H₅COCH₃ + HCl
state the conditions and catalyst required for Friedel Crafts acylation (CH₃C=O)
- heat under reflux in dry solvent (ether)
- acyl chloride
- aluminium chloride catalyst
write the equation regarding the formation of the electrophile required in the mechanism for the acylation of benzene
CH₃COCl + AlCl₃ → AlCl₄⁻ + CH₃CO⁺
draw the mechanism regarding the acylation of benzene
- electrophilic substitution (electrophile: RCO⁺)
- product: phenyl ketone or aldehyde
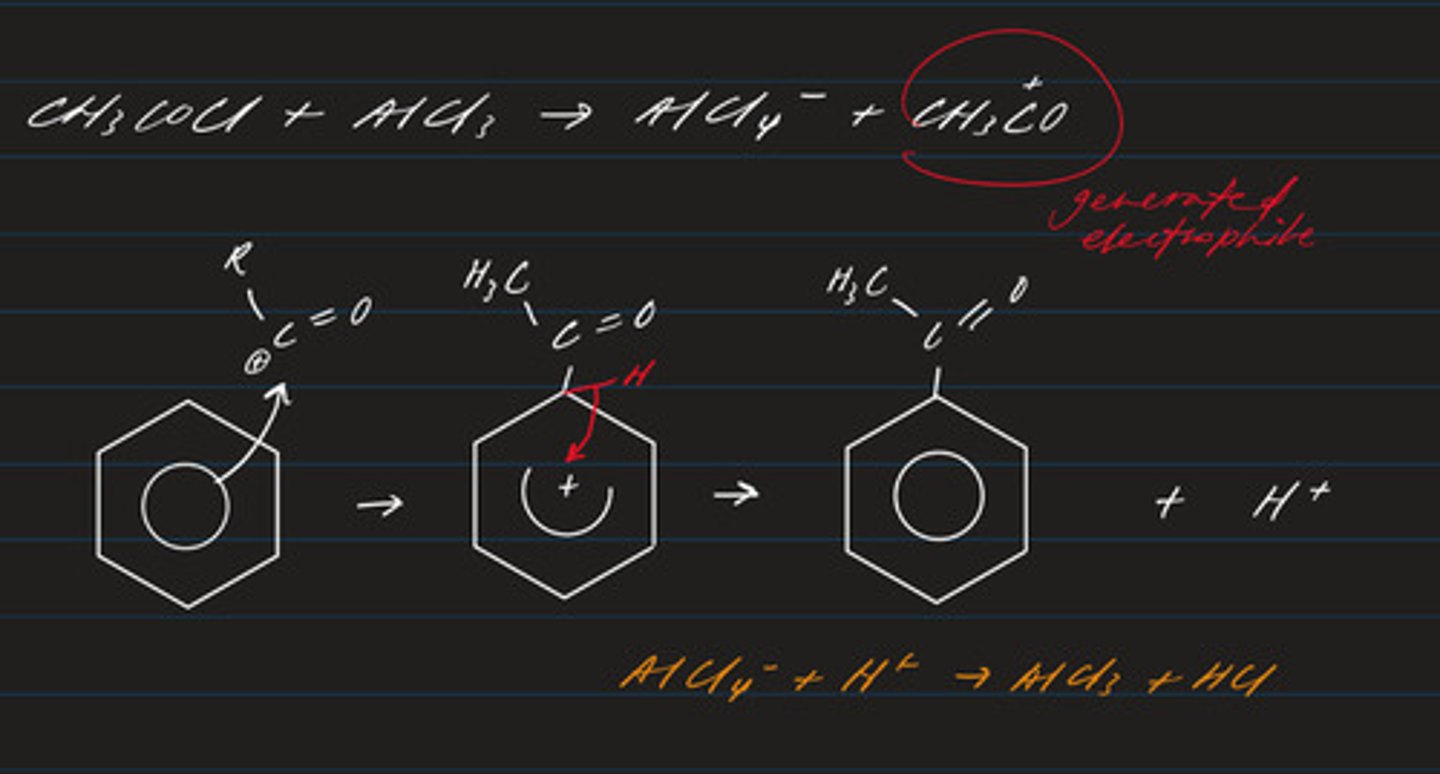
write an equation for the alkylation of benzene with a haloalkane
C₆H₆ + C₂H₅Cl → C₆H₅C₂H₅ + HCl
state the conditions and catalyst required for Friedel Crafts alkylation (eg CH₃)
- heat under reflux in a dry solvent (ether)
- halogenoalkane
- aluminium chloride catalyst
write the equation regarding the formation of the electrophile required in the mechanism for the alkylation of benzene
CH₃Cl (R) + AlCl₃ → AlCl₄⁻ + CH₃⁺
draw the mechanism regarding the alkylation of benzene
- electrophilic substitution (electrophile: R⁺)
- product: alkylbenzene
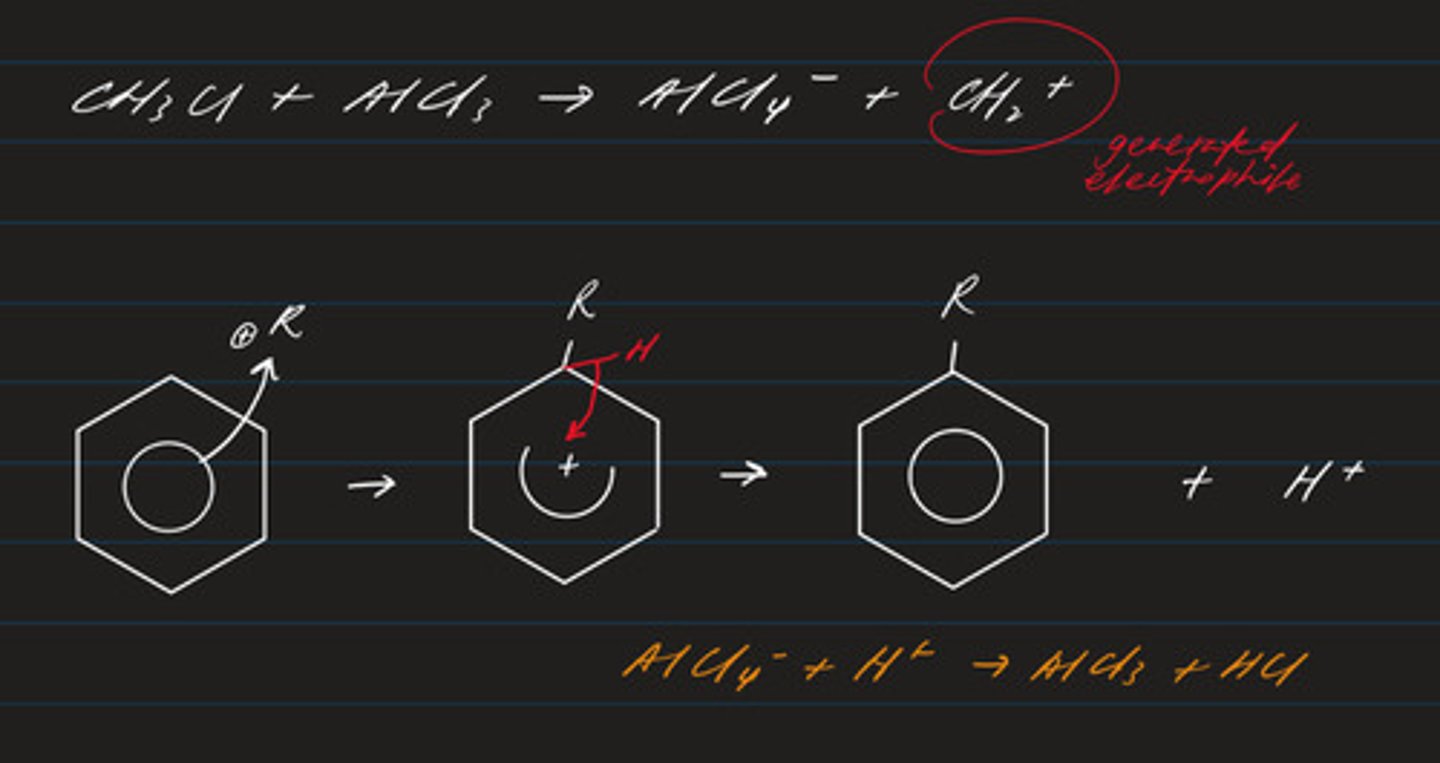
predict the shape of an AlCl₃ molecule and the Cl-Al-Cl angle. (2)
- trigonal planar
- 120°
state the compound and identify the catalyst which could be used to react with benzene in step 1.
compound: CH₃(CH₂)₃CHCl(CH₂)₆CH₃ as the 'R' group
catalyst: AlCl₃
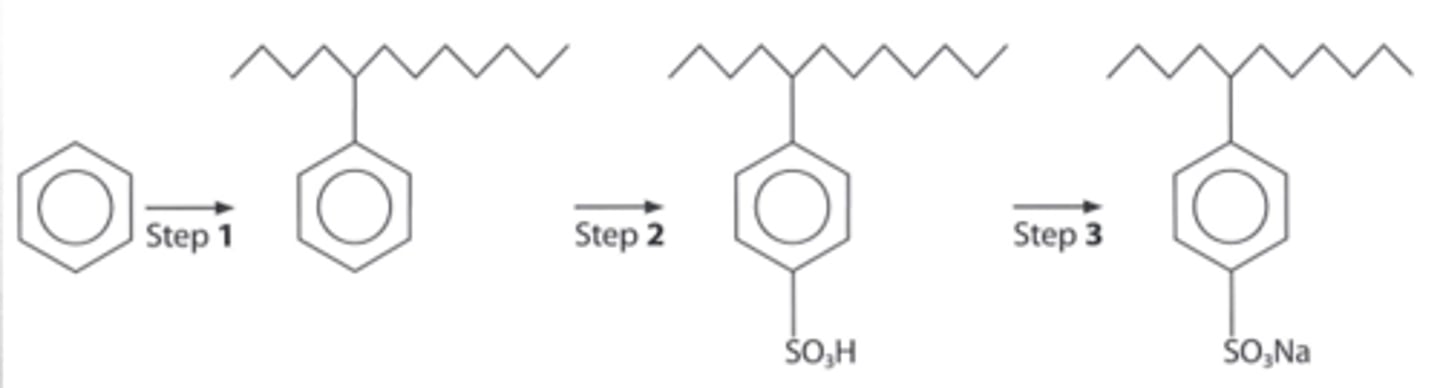
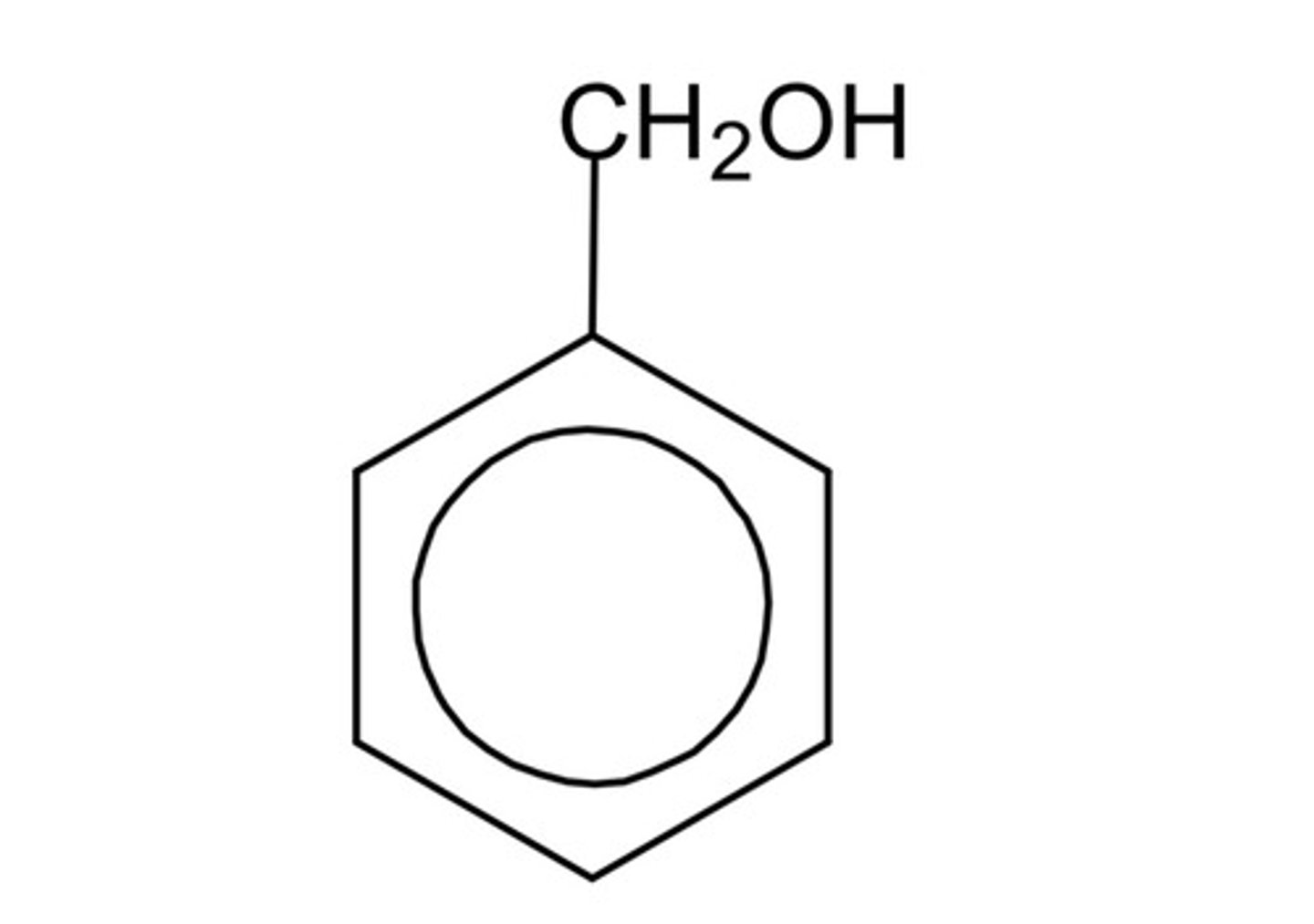
differentiate between a phenol and an alcohol with a benzene ring
- in a phenol, the OH group is directly attached to the benzene ring
- in the photo, the OH group is attached to an alkyl group rather than a benzene ring, so it is NOT a phenol
write the equation for the complete combustion of phenol. (2)
C₆H₅OH + 7O₂ → 6CO₂ + 3H₂O
give the expected observation when phenols or phenylmethanol undergo incomplete combustion. (1)
black smoke / soot
identify another type of organic compound which will also produce the same observation as phenols when undergoing incomplete combustion. (1)
alkenes
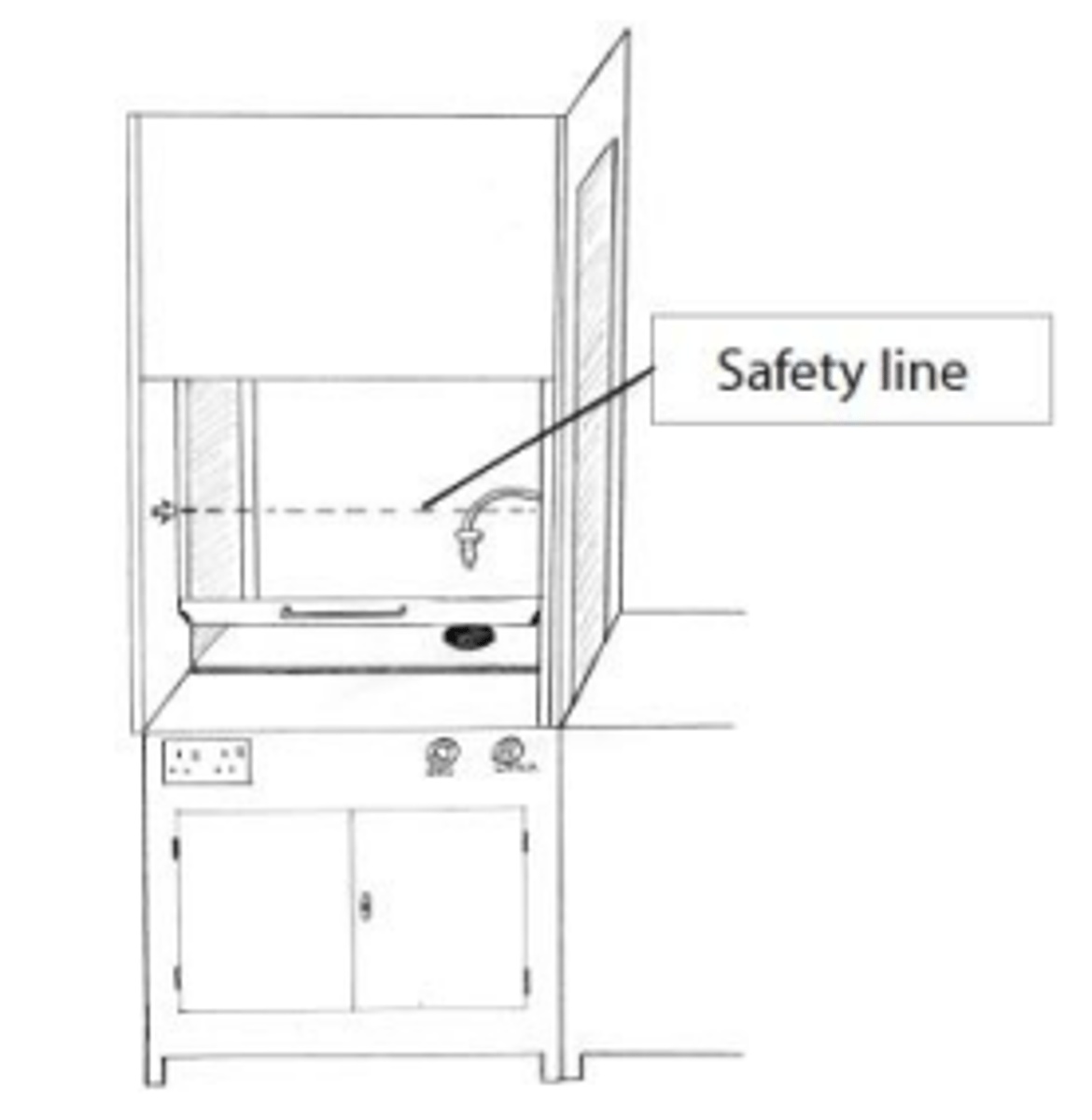
explain why the front window of the fume cupboard must be below the safety line even with the exhaust fan on. (2)
- if the window was above the safety line the exhaust system will not be strong enough to draw in the fumes
- so the toxic fumes will escape into the laboratory
explain why benzene is unable to react with a bromine molecule
- insufficient electron density
- unable to polarise the bromine molecule
- require a halogen carrier (to generate electrophile) to react with bromine
what does the likelihood of further substitution of benzene depend on?
- the functional group that is already on the ring (eg OH)
- and whether it is electron releasing (OH, NH2, CH3) or electron withdrawing (NO2)
explain why the substitution of phenol is easier than with benzene.
- OH is electron releasing
- electron density greatest at 2, 4, 6 positions
- multioke substituions are able to take place
compare the conditions and the products of the reaction for the bromination of phenol and of benzene. (4)
- both are electrophilic substitution
PHENOL (more reactive)
- forms 2,4,6-tribromophenol (multiple substitutions occur)
- react with bromine water at RTP
BENZENE (less reactive)
- forms bromobenzene (one substitution)
- requires a halogen carrier catalyst (eg FeBr₄)
the bromination of the benzene ring occurs much more readily in phenols compared to the bromination of benzene. explain this increased reactivity. (2)
- the OH group in phenols are electron withdrawing
- the lone pair from the oxygen will overlaps with the delocalised ring of electrons of the benzene ring
- increase electron density of ring
- ring is more susceptible to electrophilic attack
- bromine is more easily polarised
state the reagent and condition required for the bromination of phenol
- bromine water
- in the cold
write a displayed formula for the bromination of phenol in bromine water (tri-substitution)
2,4,6-tribromophenol
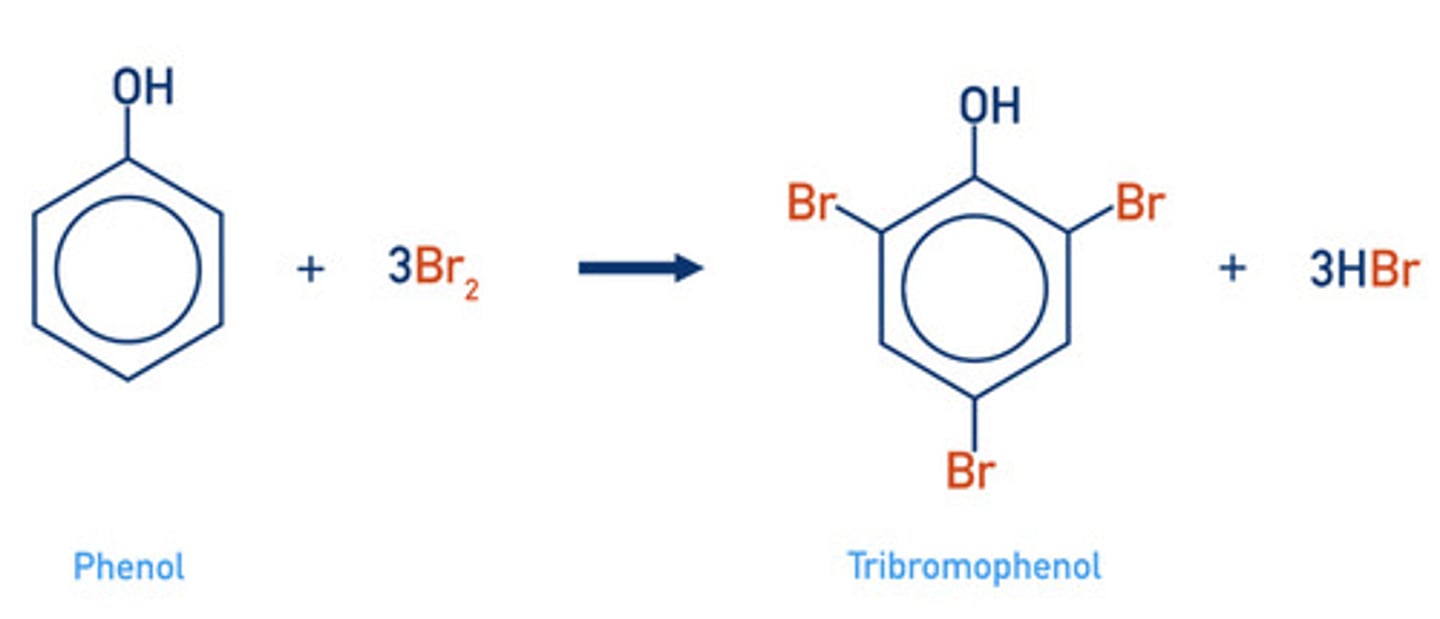
state the scent and 1 observation of tribromophenol
antiseptic smell
white precipitate
write a displayed formula for the bromination of phenol (mono-substitution)
2- or 4-bromophenol
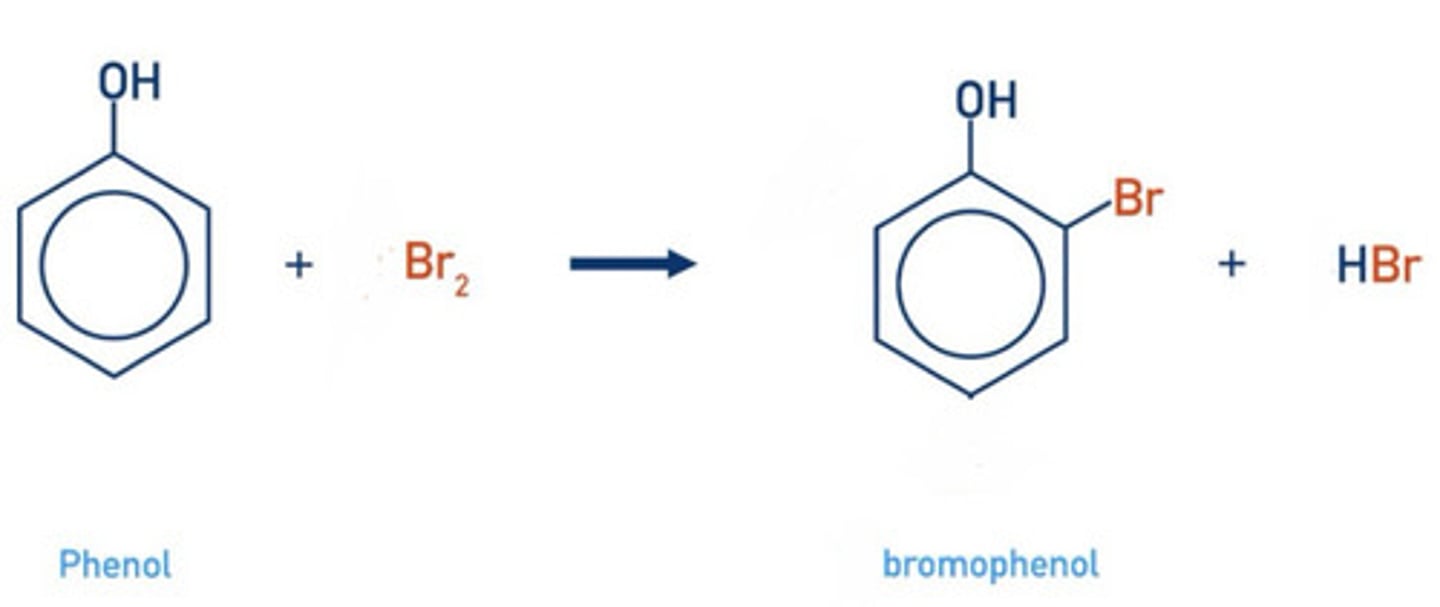
what type of reaction is the bromination of phenol?
electrophilic substitution
a mixture of 2- and 4-nitrophenol can be separated by steam distillation.
the boiling temperature of 2-nitrophenol is 215°C and 4-nitrophenol is 279°C
explain, in terms of intermolecular forces, why 4-nitrophenol has a higher boiling temperature than 2-nitrophenol. (2)
- they can both form hydrogen bonds
- but in 4-nitrophenol the hydrogen bonds join molecules in a straight chain (at both ends of the ring) so are stronger
explain why phenols (acidic) are classified as weak acids
- produce phenoxides and lose H+
- due to electron-withdrawing -OH group
- negativity on O- is delocalised around ring structure
- phenoxides are fairly stable
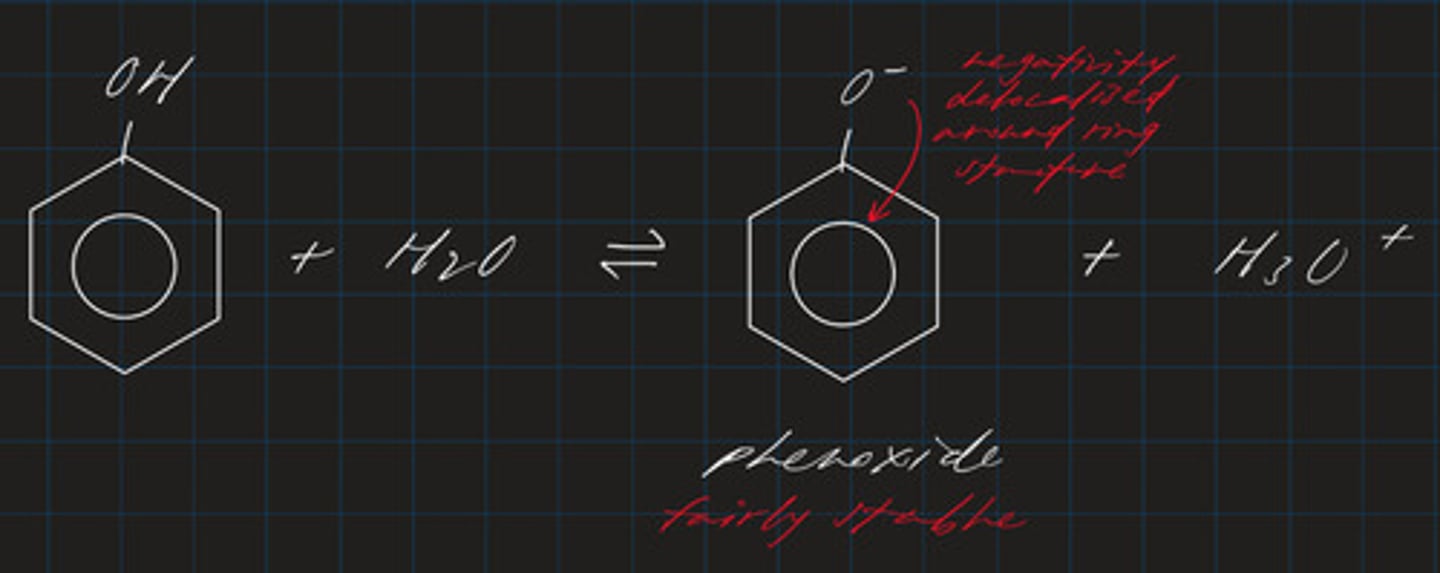
what can phenoxides react with?
- bases (eg sodium-containing compounds)
- can produce salts (eg sodium phenoxide) which is more soluble than phenols
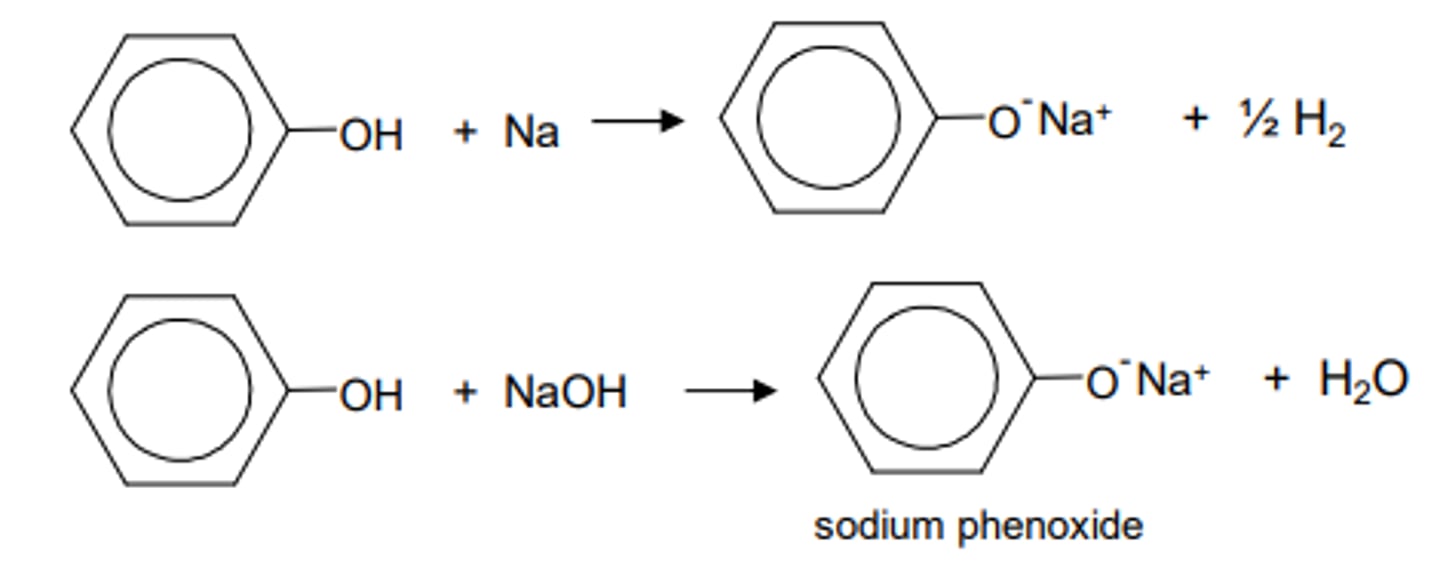
what is the expected product when phenol undergoes bromination?
A) bromobenzene
B) 2-bromophenol
C) 4-bromophenol
D) both B and C
D
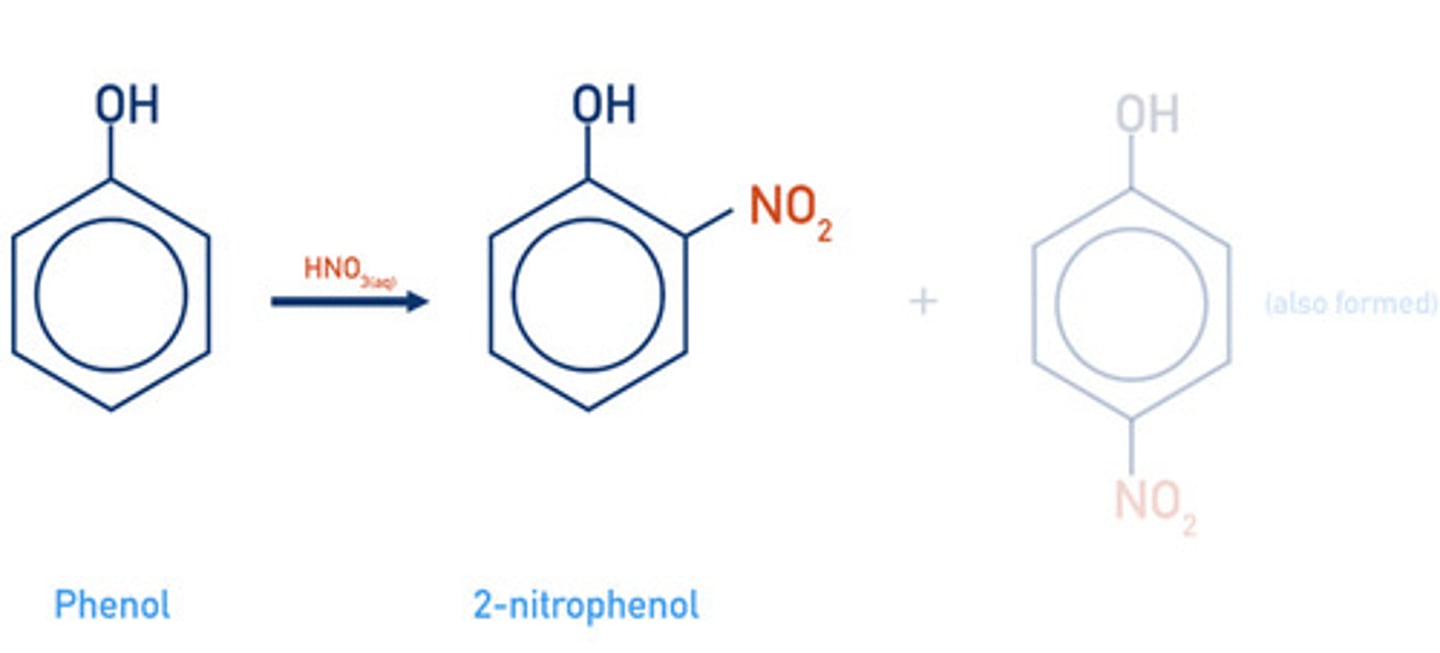
state the conditions required for the nitration of phenol
- dilute nitric acid
- rtp
write a displayed formula for the nitration of phenol with dilute nitric acid (mono-substitution)
2- or 4-nitrophenol
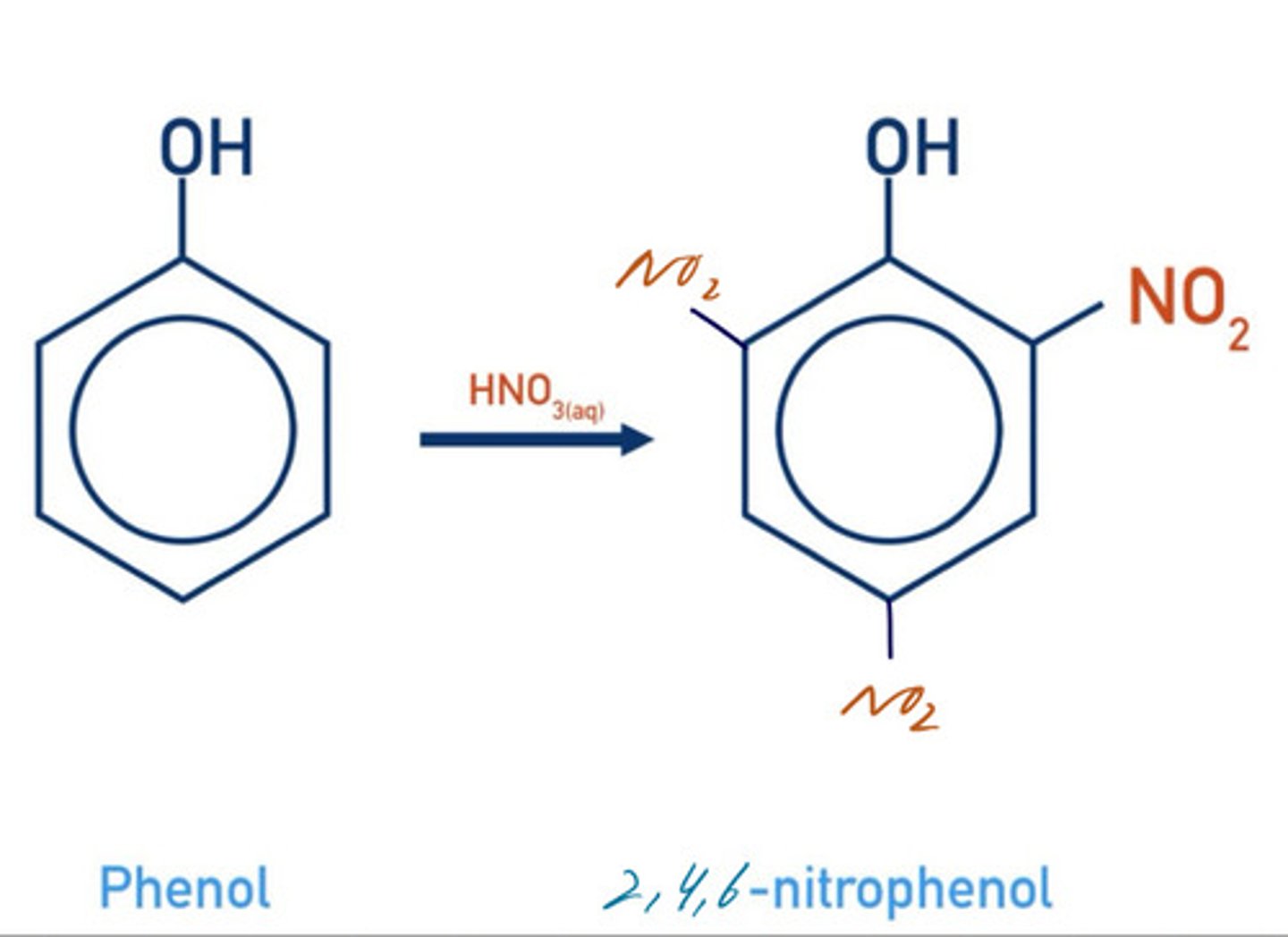
write a displayed formula for the nitration of phenol with concentrated nitric acid (tri-substitution)
2,4,6-trinitrophenol
the boiling temperature of 2-nitrophenol is 215˚C while 4-nitrophenol is 279˚C.
explain why 4-nitrophenol has a higher boiling temperature than 2-nitrophenol. (2)
- they both form hydrogen bonds
- but in 4-nitrophenol the hydrogen bonds join the molecules in a straight chains so they are stronger
- while 2-nitrophenol forms intramolecular hydrogen bonds so fewer intermolecular hydrogen bonds
what is the mass of 4-nitrophenol produced from 0.94g of phenol if the yield of the isomer is 15%?
0.21g
selecting only from these compounds, devise a synthetic pathway for converting benzene into phenylethene, clearly identifying compounds A and B and stating the appropriate conditions for each step. (5)
step 1: acylation of benzene using ethanoyl chloride + aluminium chloride (catalyst) + heat
A is a phenylethanone
step 2: reduction of A with LiAlH₄ + dry ether
B is phenylethanol
step 3: dehydration of B with conc. phosphoric acid

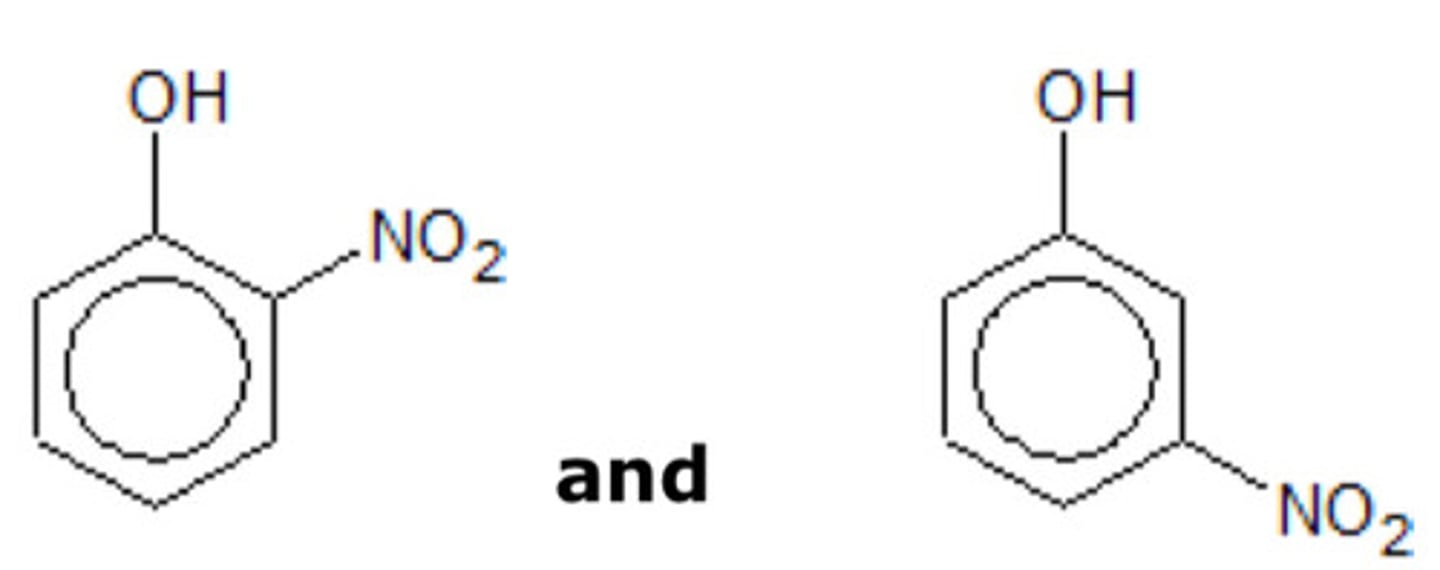
draw 2 structural isomers of 4-nitrophenol with a benzene ring. (1)
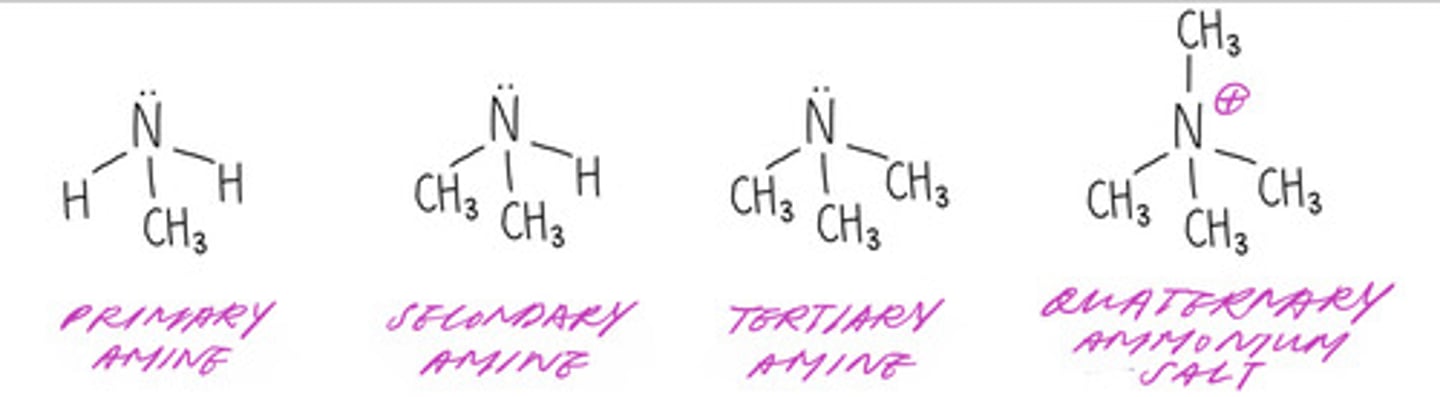
the classification of amines (NH2)
(quaternary ammonium salt is NOT an amine)
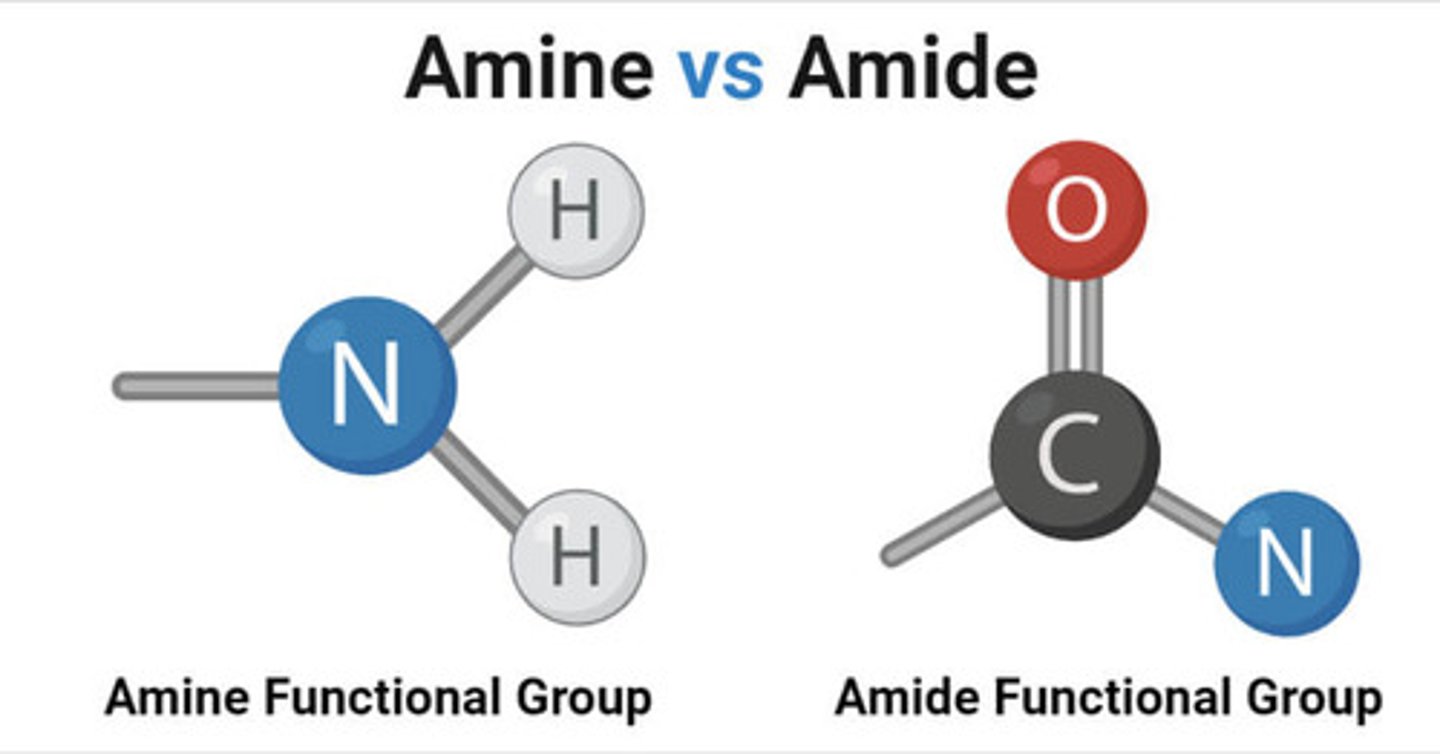
differentiate between the amine vs amide functional group
state the base properties of primary aliphatic amines
CH₃NH₂ + H₂O → CH₃NH₃⁺ + OH⁻
- act as bronsted-lowry bases
- lone pair on nitrogen available for formation of dative covalent bond with an H+
- they are also stronger bases than ammonia due to the positive inductive effect of R groups
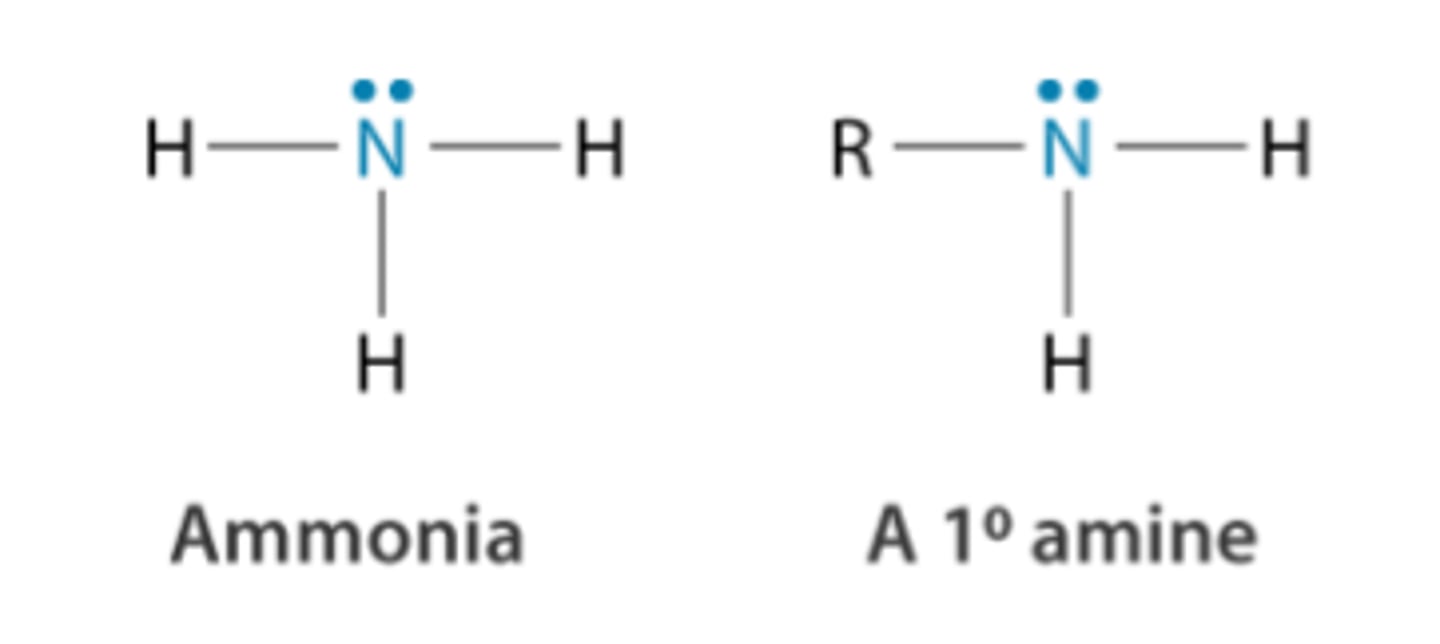
reaction of amines with water to form alkaline solution
CH₃NH₂ + H₂O ⇌ CH₃NH₃⁺ + OH⁻
explain why phenylamine is a weaker base than butylamine (3)
- lone pair on nitrogen atom
- interaction overlap of lone pair and delocalised pi electrons of the ring
- less able to accept protons
- while butyl group push electrons towards lone pair (positive inductive effect)
- more able to accept protons
compare and contrast the basicity of phenylamine and butylamine. (4)
- both are basic due to their lone pair on the nitrogen atom (able to accept protons)
- however in phenylamine lone pair on N overlaps with delocalised pi ring of electrons
- so less able to accept protons = weaker base
- butyl group is electron releasing (positive inductive effect)
- so more able to accept protons = stronger base
reaction of amines with acids to form ammonium salts
the ionic salts are solid crystals
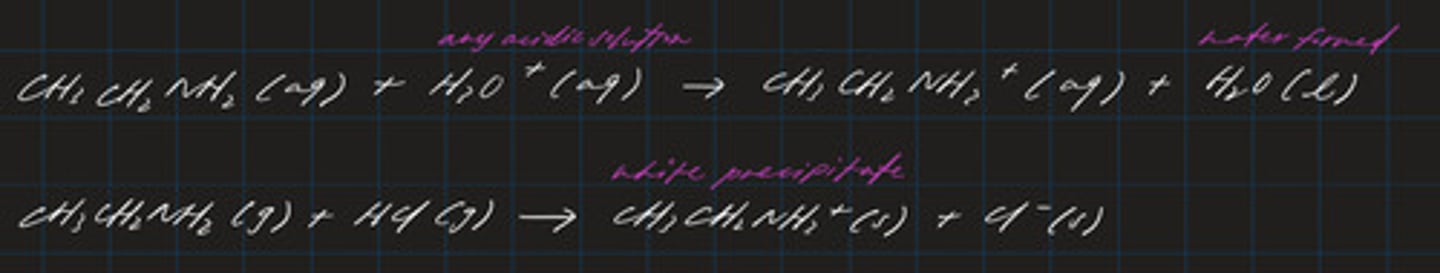
reaction of primary amines with ethanoyl chloride to form secondary amides (N-substituted)
- an addition-elimination reaction
- forms the CONH group
- unlike the reactions between ethanoyl chloride and water / ethanol, HCl is NOT produced
- this is because HCl immediately reacts with excess conc methyl amine to give methylammonium chloride
- otherwise, HCl can be formed instead
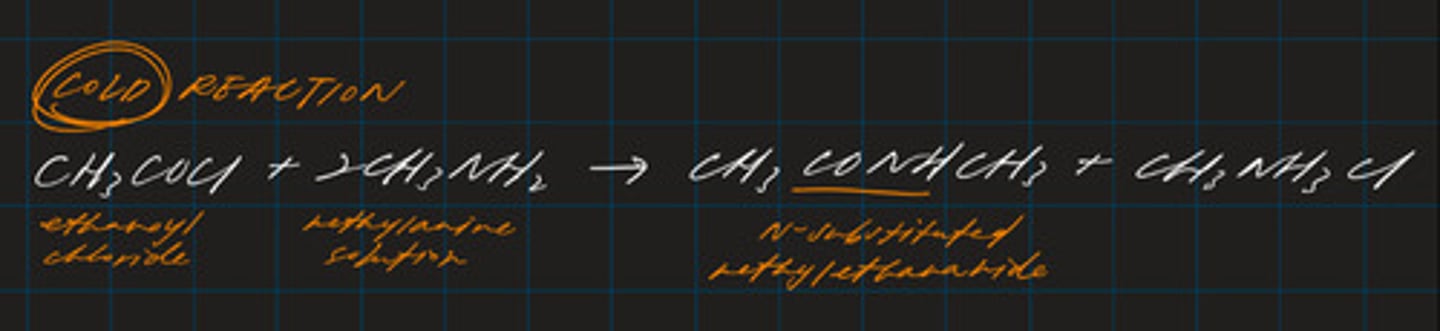
write an equation for the reaction between propanoyl chloride and pentylamine. name the amide formed. (2)
CH₃CH₂COCl + CH₃CH₂CH₂CH₂CH₂NH₂ → CH₃CH₂CONHCH₃CH₂CH₂CH₂CH₂ + HCl
N-pentylpropanamide
2C₄H₉NH₂ + CH₃COCl → C₄H₉NHCOCH₃ + C₄H₉NH₃⁺Cl⁻
explain how this equation illustrates that butylamine acts as a nucleophile and as a base. (4)
- nucleophiles are electron pair donors
- so the amine group attacks as a nucleophile
- it attacks the delta-positive C of the acyl chloride
- which produces hydrogen chloride
- its a base since amine group reacts with the acid to produce the salt C₄H₉NH₃Cl
give the name and structural formula of the compound needed to react with butylamine, C₄H₉NH₂, to form CH₃CH₂CONHC₄H₉. (2)
- the -CONH- group is formed, hence it is reacting with an acyl chloride
- CH₃CH₂ suggest that it is reacting with propanoyl chloride
- formula of CH₃CH₂COCl
give the reagent and condition for the formation of propylamine (primary amine) from a halogenoalkane
- alcoholic ammonia
- heat and under pressure
- sealed tube
draw the mechanism for the reaction of CH₃CH₂CH₂Cl with an excess of ammonia to form the primary amine. (3)
write an equation for the formation of propylamine, CH₃CH₂CH₂NH₂ from a halogenoalkane
CH₃CH₂CH₂Br + NH₃ → CH₃CH₂CH₂NH₂ + HBr
reaction of primary amines with halogenoalkanes to form secondary amines and so on (nucleophilic substitution)
R'NH₂ + R"X → R'NHR" + HX
where R' is the alkyl group in the amine and R" is the alkyl group in the halogenoalkane
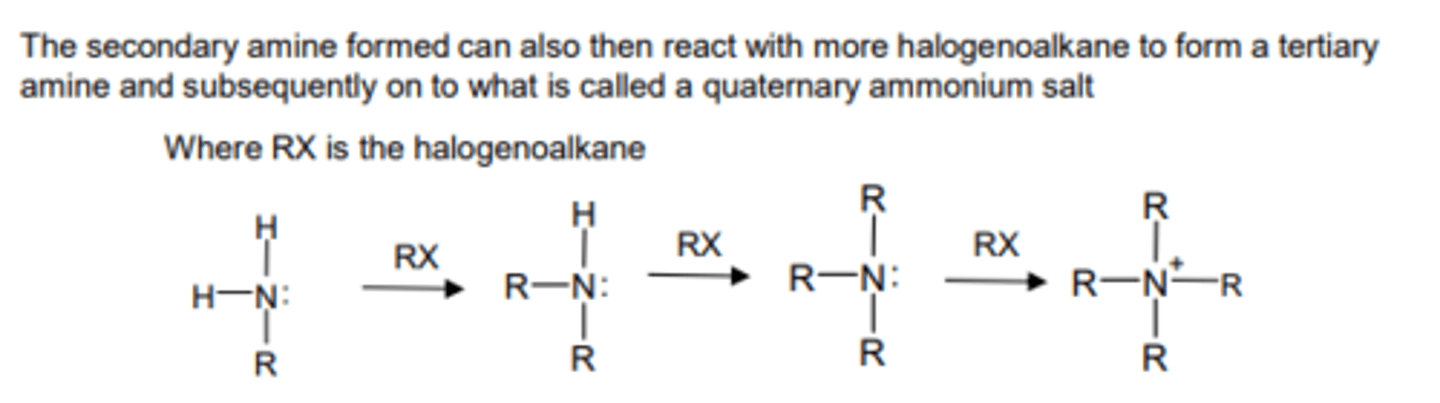
explain why secondary amines are stronger bases than primary amines
- secondary amines have more alkyl groups substituted on the N atom
- hence more electron density is pushed onto N atom (positive inductive effect of R groups)
then, explain why tertiary amines are NOT stronger bases than secondary amines (not examined)
- tertiary amines and quaternary ammonium salts are less soluble in water
- hence they are less strong than secondary amines
state the conditions required to make primary amines from nitriles (-CN)
- LiAl₄ + dry ether
- or hydrogen gas in presence of nickel catalyst
write an equation for the formation of propylamine, CH₃CH₂CH₂NH₂ from a nitrile
CH₃CH₂CN + 4[H] → CH₃CH₂CH₂NH₂
beware of amount of Cs, should only be 3 for propyl-
state the condition and reagent required for the preparation of phenylamine through the reduction of nitroarenes
TWO PART PROCESS
(under the guise that you are starting from a nitrobenzene)
1. Sn (catalyst) + conc HCl + heat under reflux
this will form our -NH3 compound
2. NaOH solution
this will reduce our -NH3 compound into -NH2
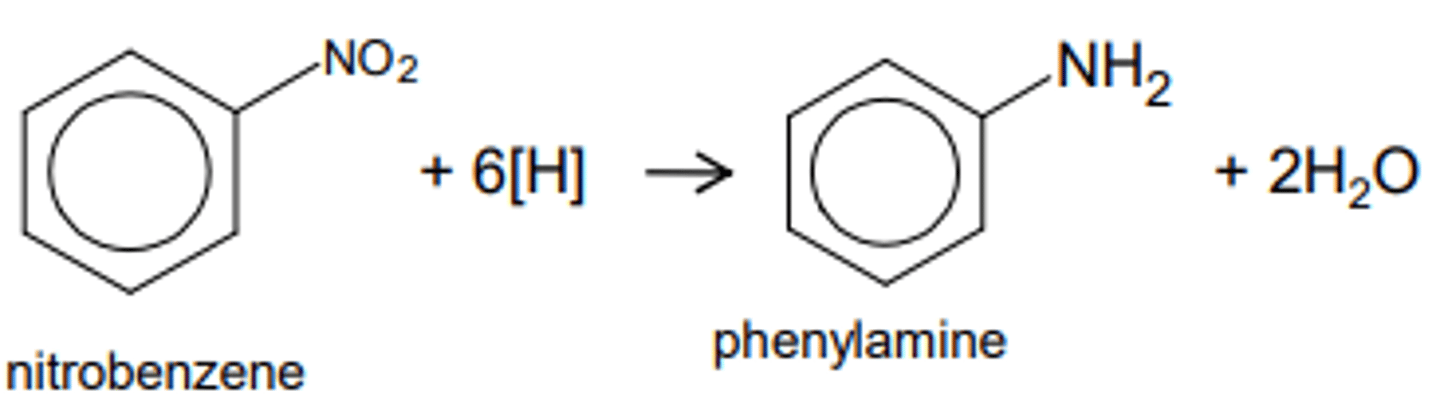
illustrate a flow chart for the preparation of phenylamines through the reduction of nitroarenes (start with a benzene)

write the equation for Reaction 1 to show why the pH of the solution is greater than 7. state symbols are not required. (1)
CH₃NH₂ + H₂O ⇌ CH₃NH₃⁺ + OH⁻
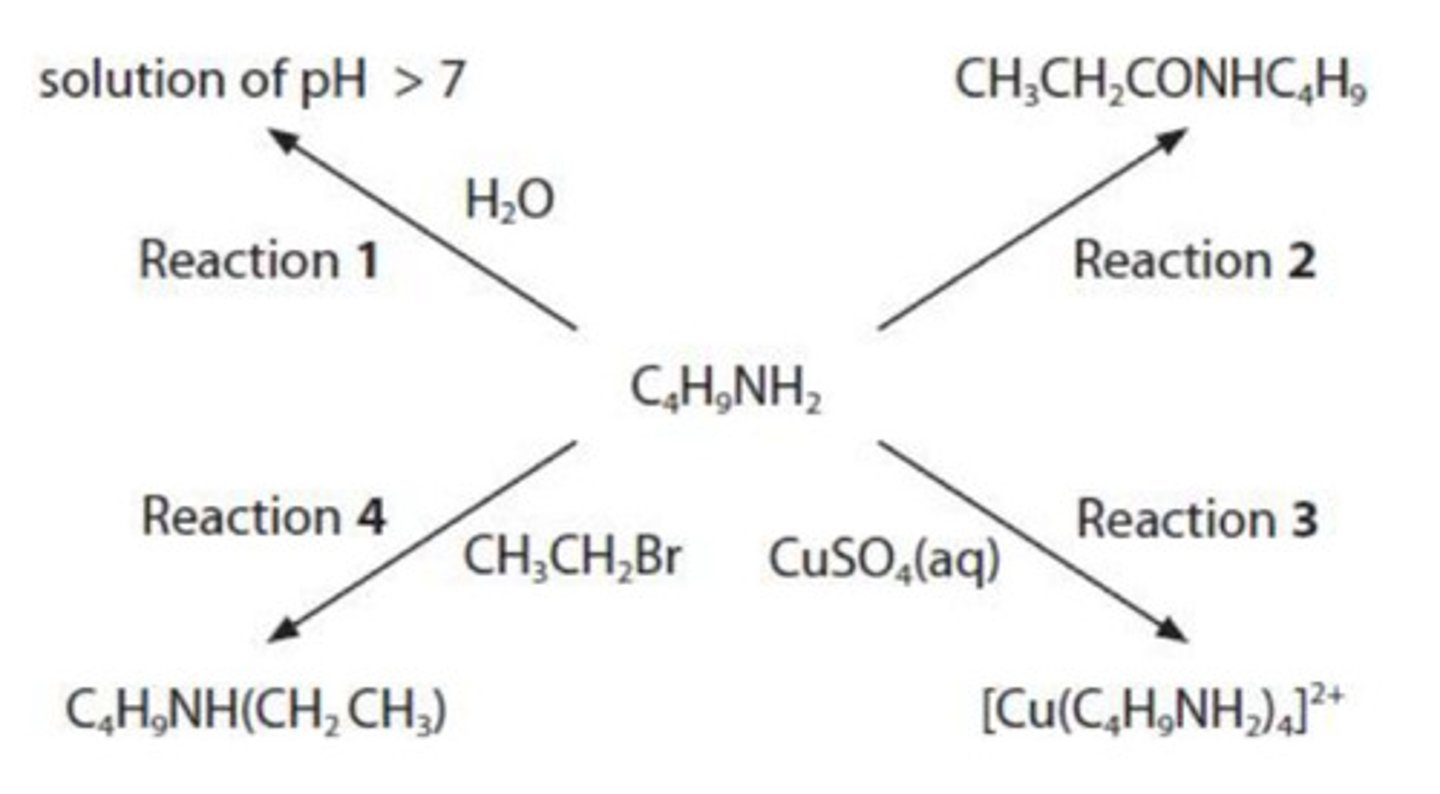
give the name and the structural formula of the compound needed to react with butylamine in Reaction 2.
name: propanoyl chloride
structural formula: CH3CH2COCl
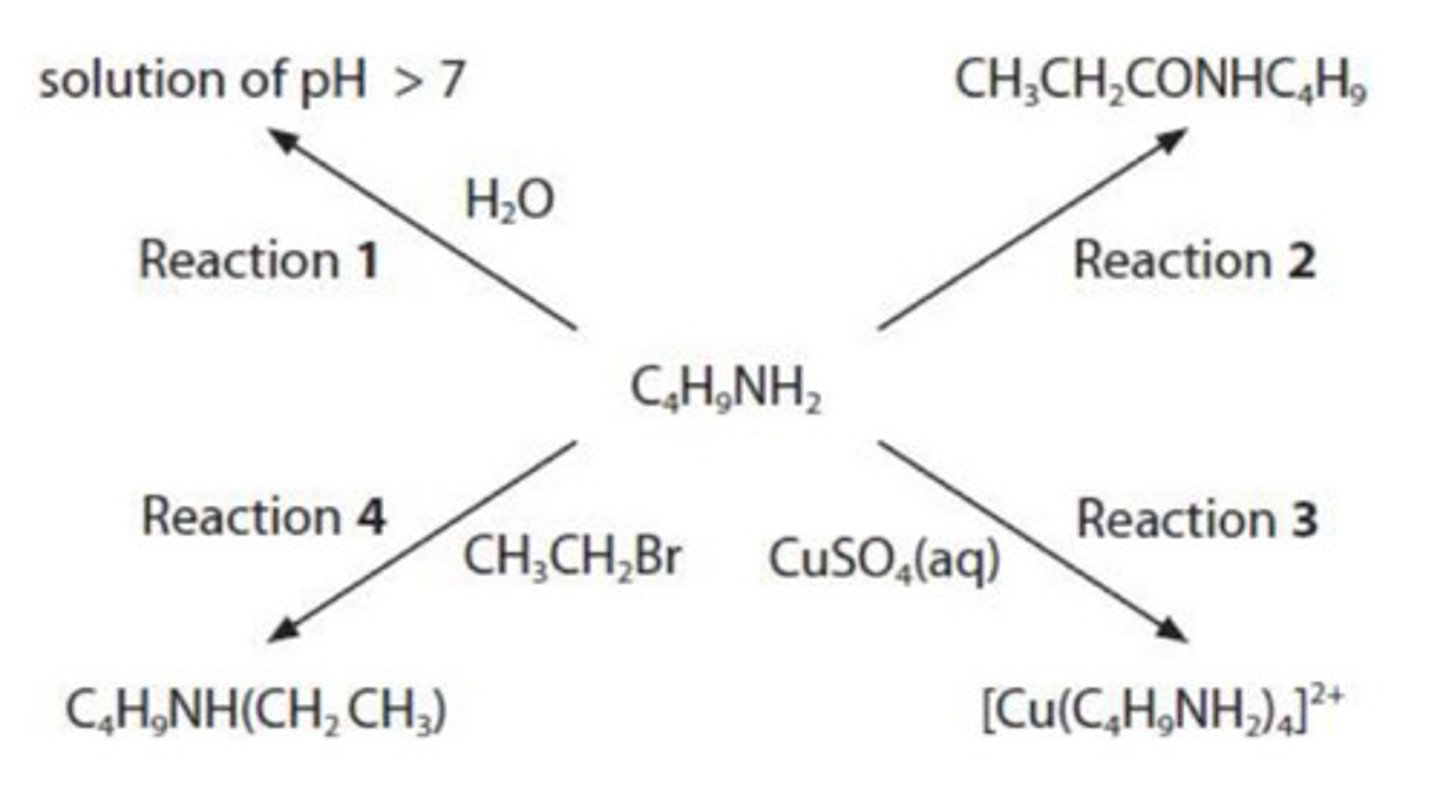
what is seen when excess butylamine is used in Reaction 3?
blue solution
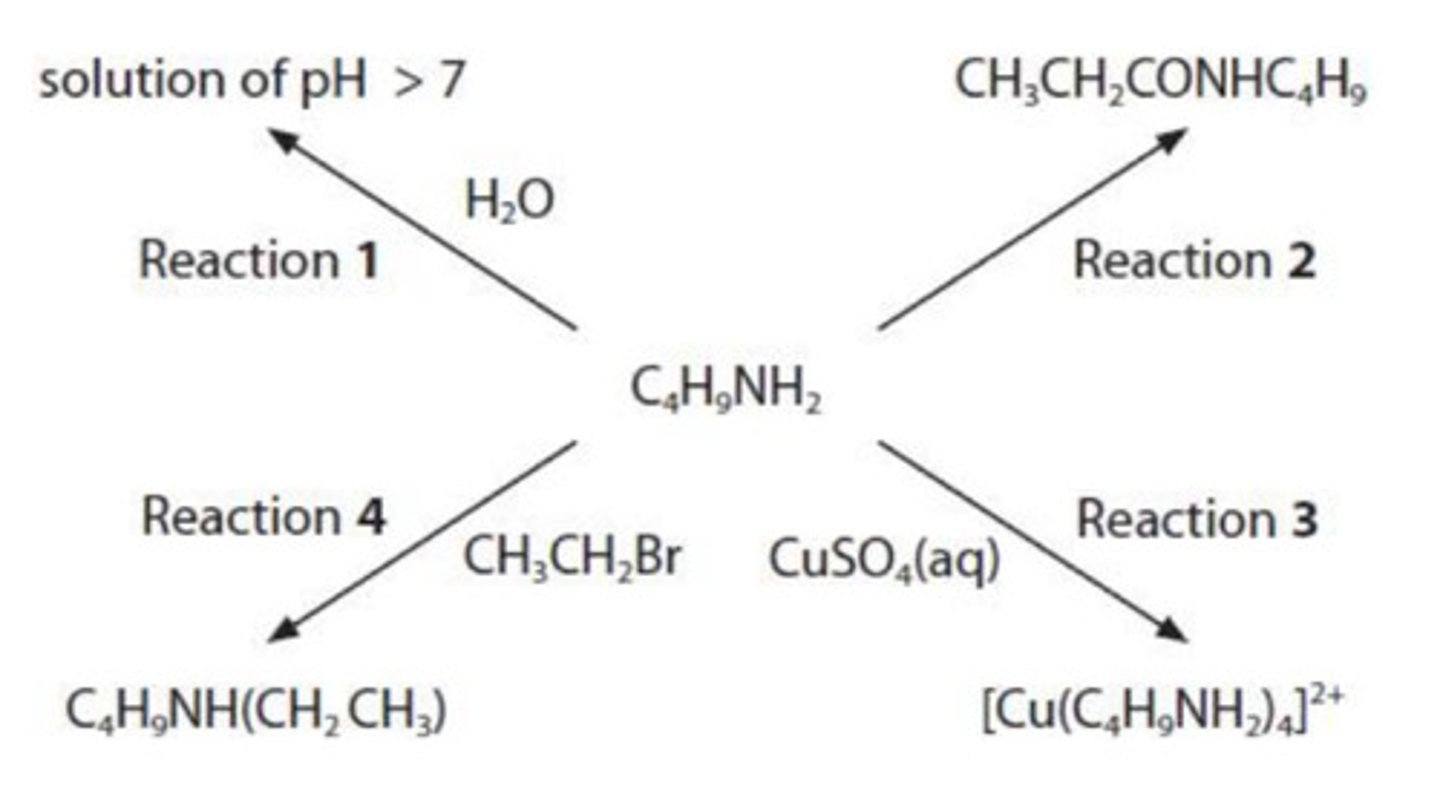
what is the type and mechanism for the reaction in Reaction 4?
nucleophilic substitution
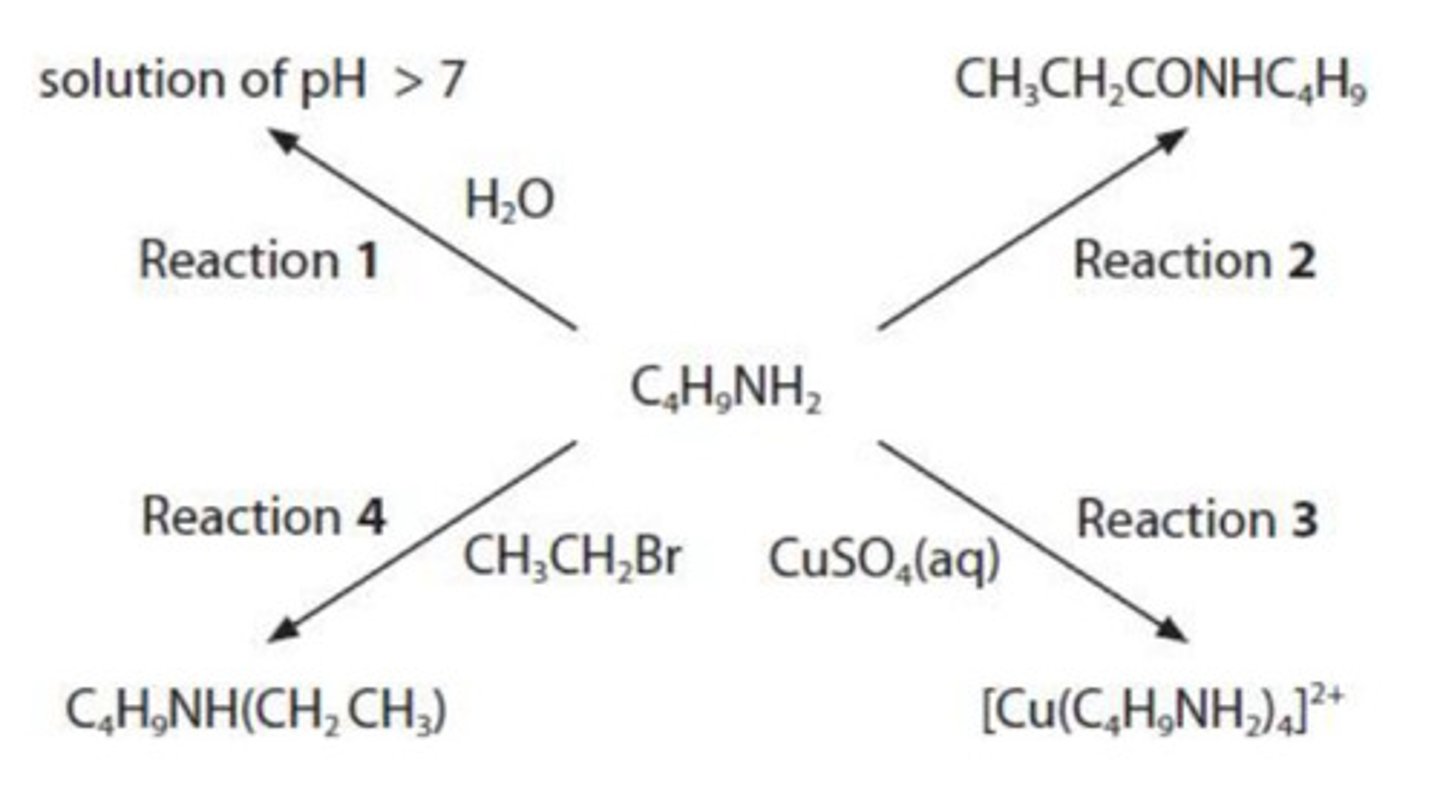
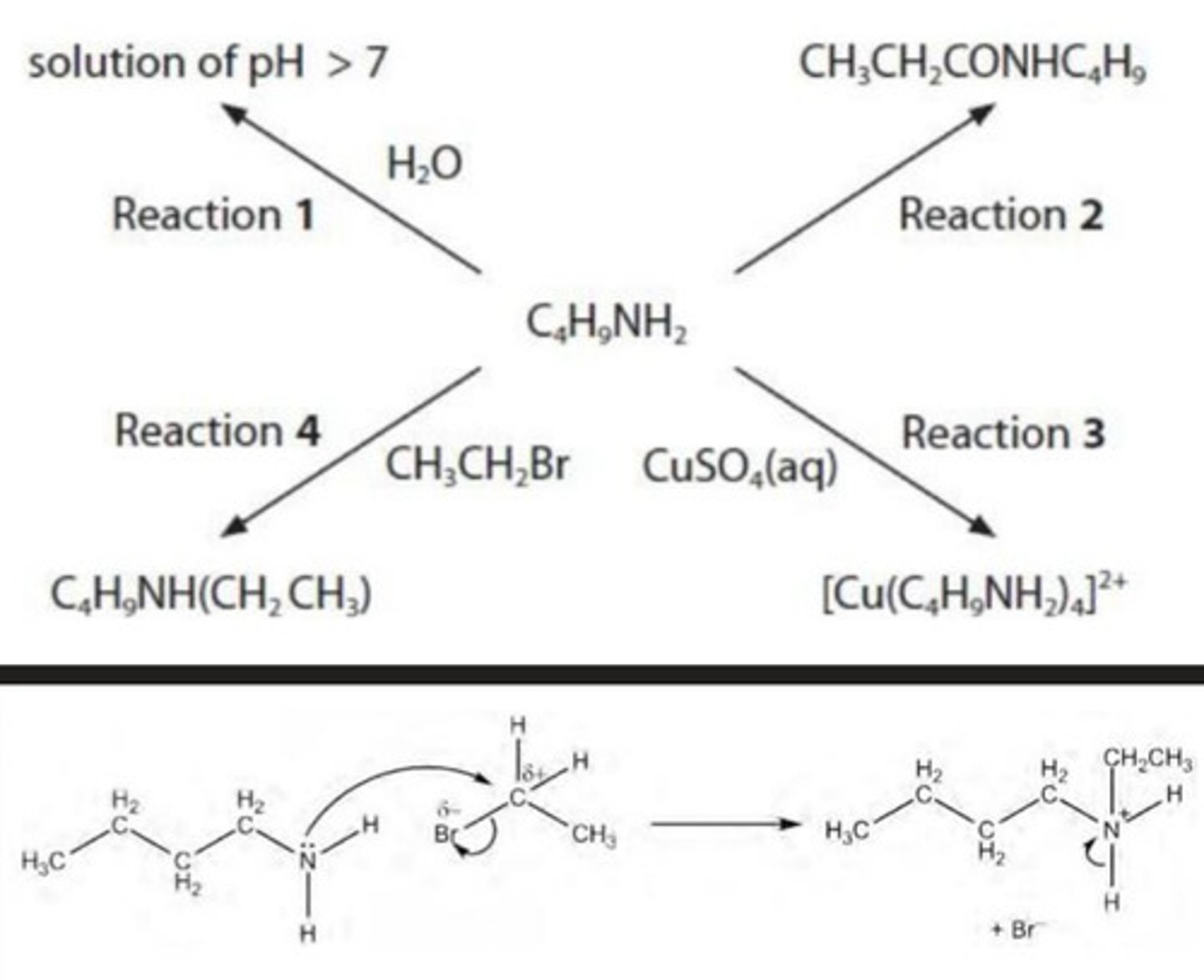
draw the mechanism for Reaction 4.
include curly arrows and relevant lone pairs and dipoles.
phenylamine can be prepared from nitrobenzene.
C₆H₅NO₂ → C₆H₅NH₂
butylamine can be prepared from butanenitrile.
C₃H₇CN → C₄H₉NH₂
compare and contrast these 2 preparations of amines.
- both are reduction reactions
- reagents for preparation of phenylamine are tin (Sn) and conc HCl
- reagents for preparation of butylamine are hydrogen gas and nickel catalyst
- or LiAl₄ + dry ether
state the general formula of an amino acid
RCH(NH₂)COOH
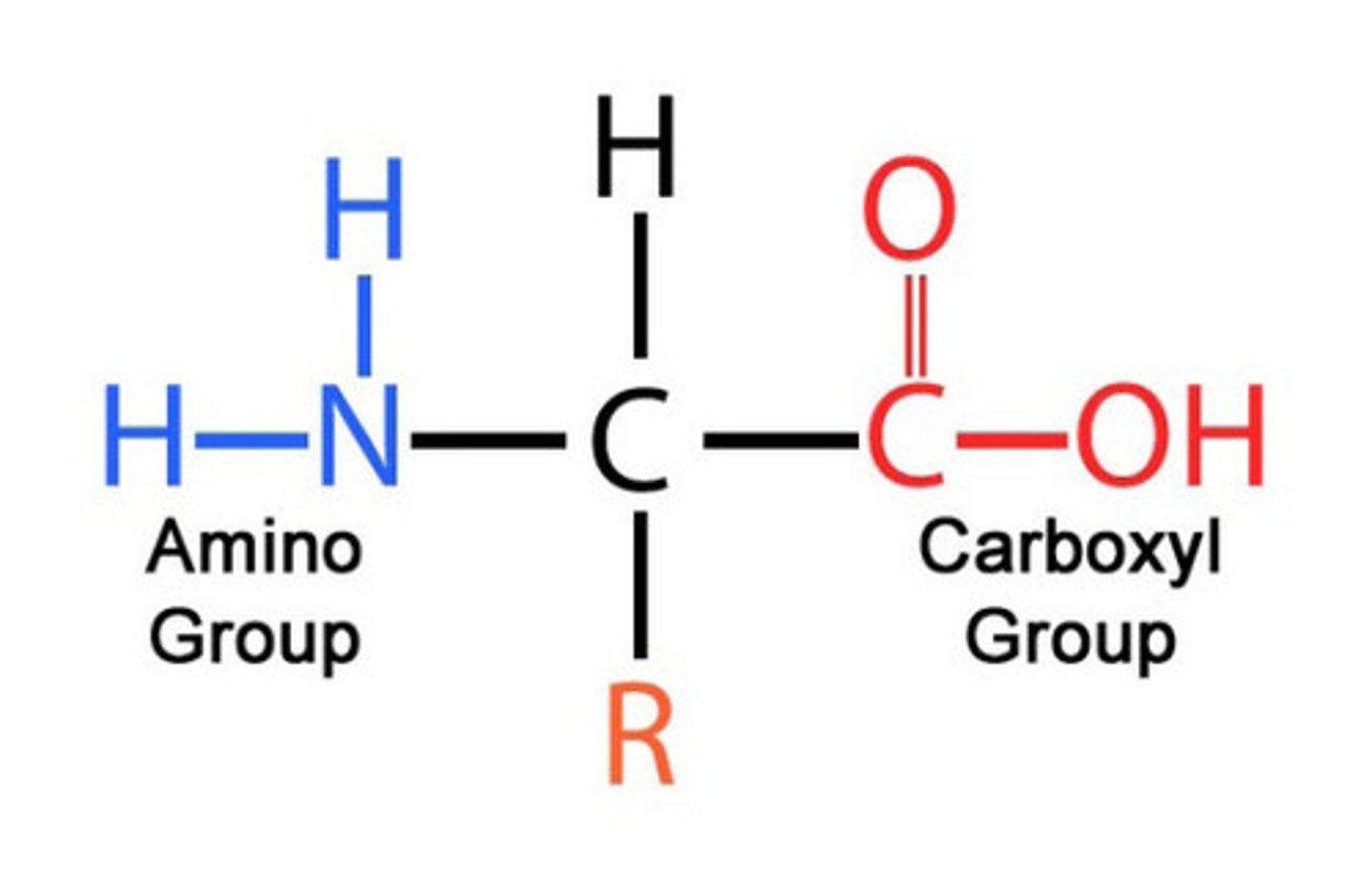
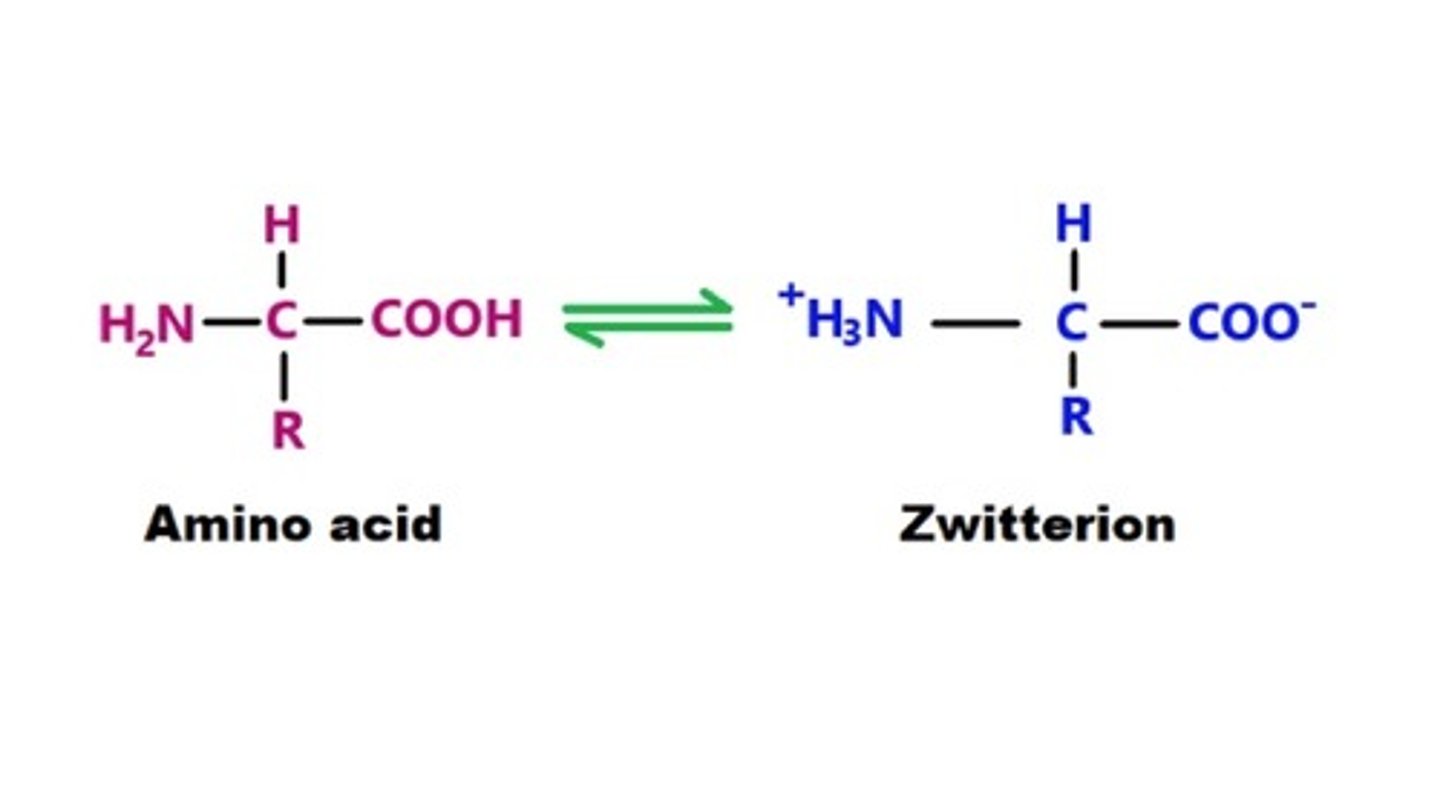
define what a zwitterion is and how it is formed
- a molecule that contains equal +ve and -ve charged functional group
- this occurs at isoelectronic point, which is the pH at which amino acids can exist as a zwitteron
- they are soluble in water
explain why amino acids such as glycine are cristalline solids at RTP. (2)
- amino acids can exist as zwitterions
- ionic bonds form between zwitterions
write a displayed formula for the reaction between an amino acid and an alkali
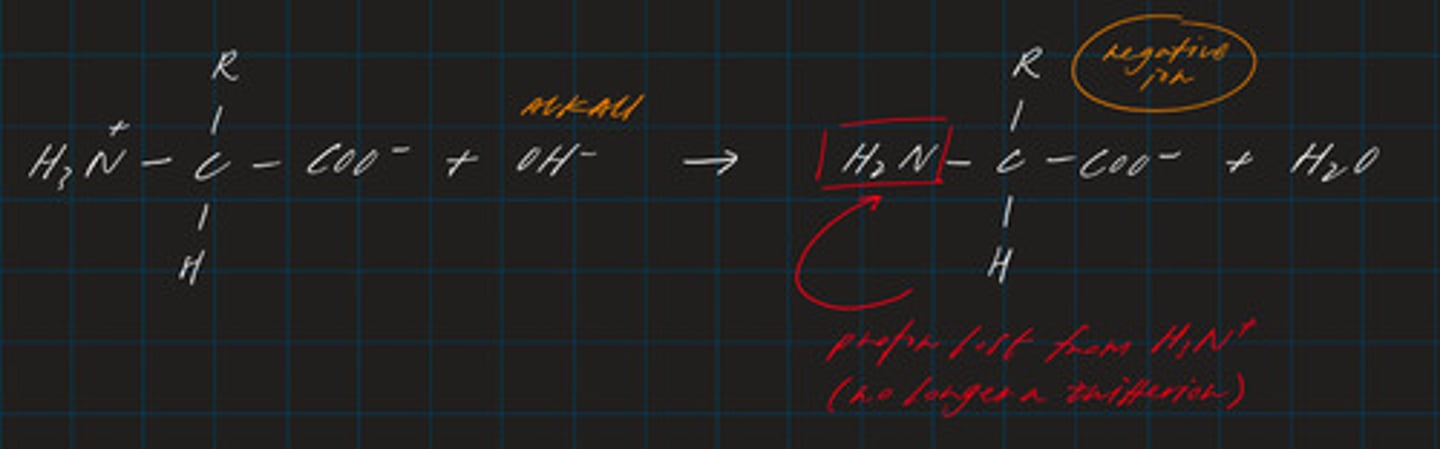
write a displayed formula for the reaction between an amino acid and an acid
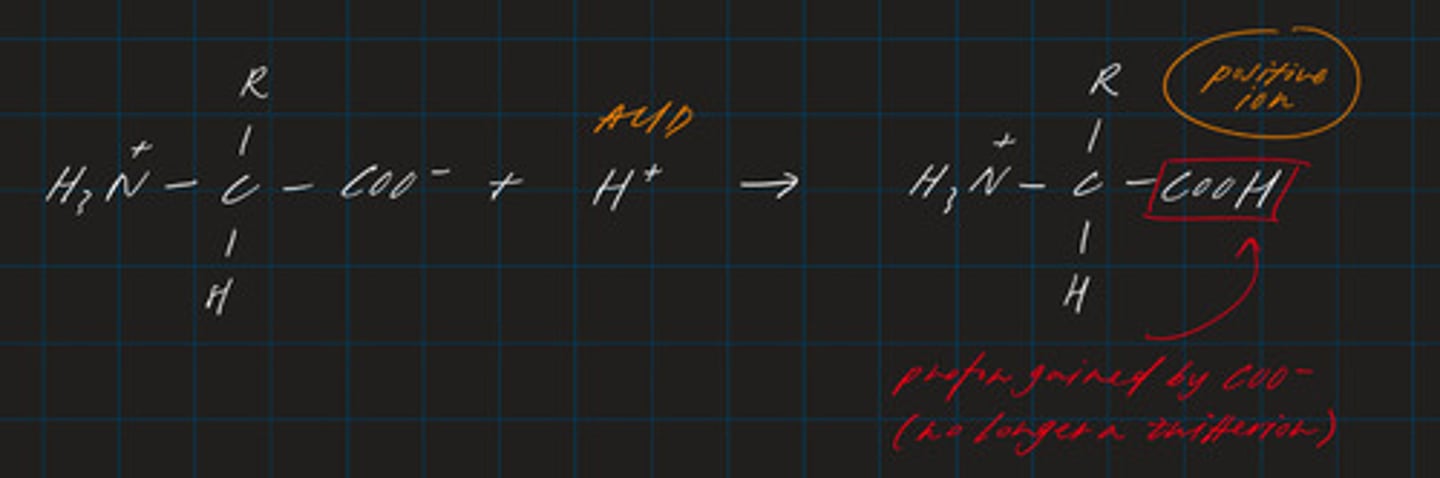
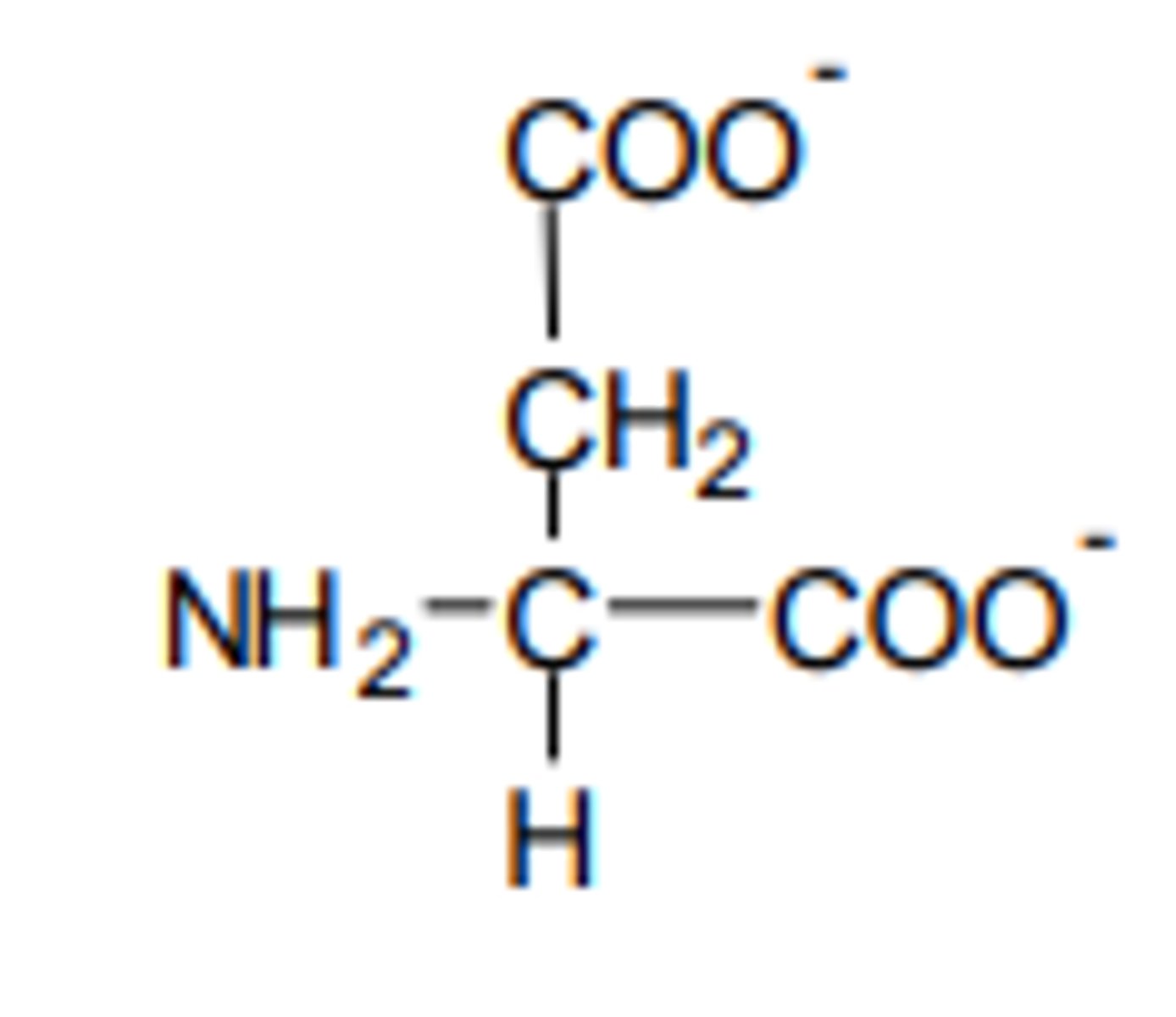
the R group of aspartic acid, an amino acid, consists of a COOH group. illustrate how the zwitterion form of aspartic acid would appear in excess alkali
the extra group on the R group will also react in excess alkali conditions
describe how an amino acid can be identified via chromatography
- spray ninhydrin then heat for visible spots to appear (amino acids are transparent)
- identify the amount they have moved
- Rf value: distance moved by amino acid / distance moved by solvent
- compare unknown amino acid's Rf to known data book value
optical activity of amino acids
- all except glycine are chiral due to 4 different groups around the C
- can rotate plane-polarised light
- similar physical and chemical properties
differentiate between an a-amino acid and a b-amino acid
a-amino acid has both the amine group and carboxyl group joined to the same central carbon
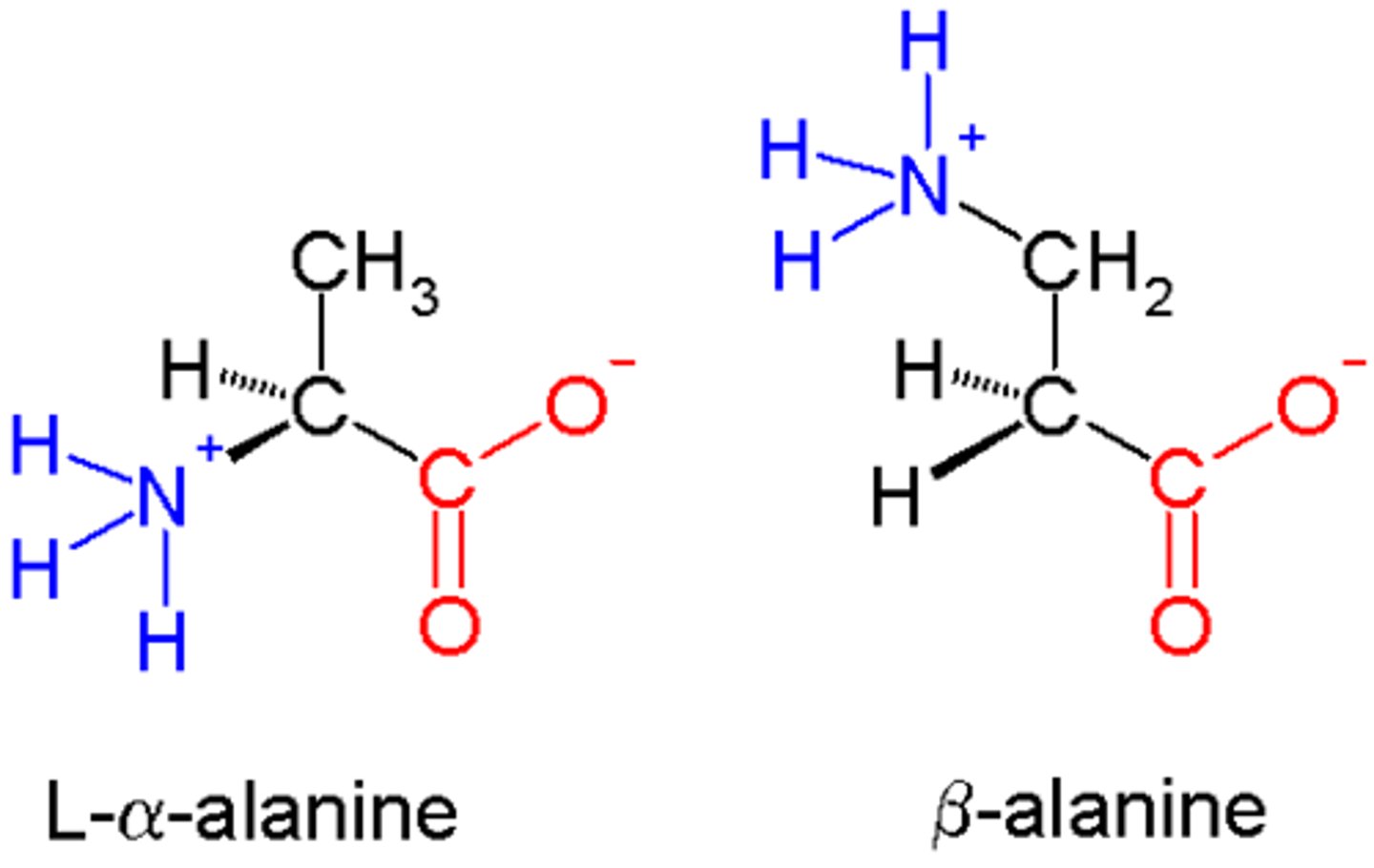
alanine and glycine are amino acids.
compare and contrast their structure and optical activity. (4)
- they are both alpha amino acids / 2-amino acids
- alanine has a chiral centre with non-superimposable mirror images
- alanine can rotate plane-polarised light but glycine cannot
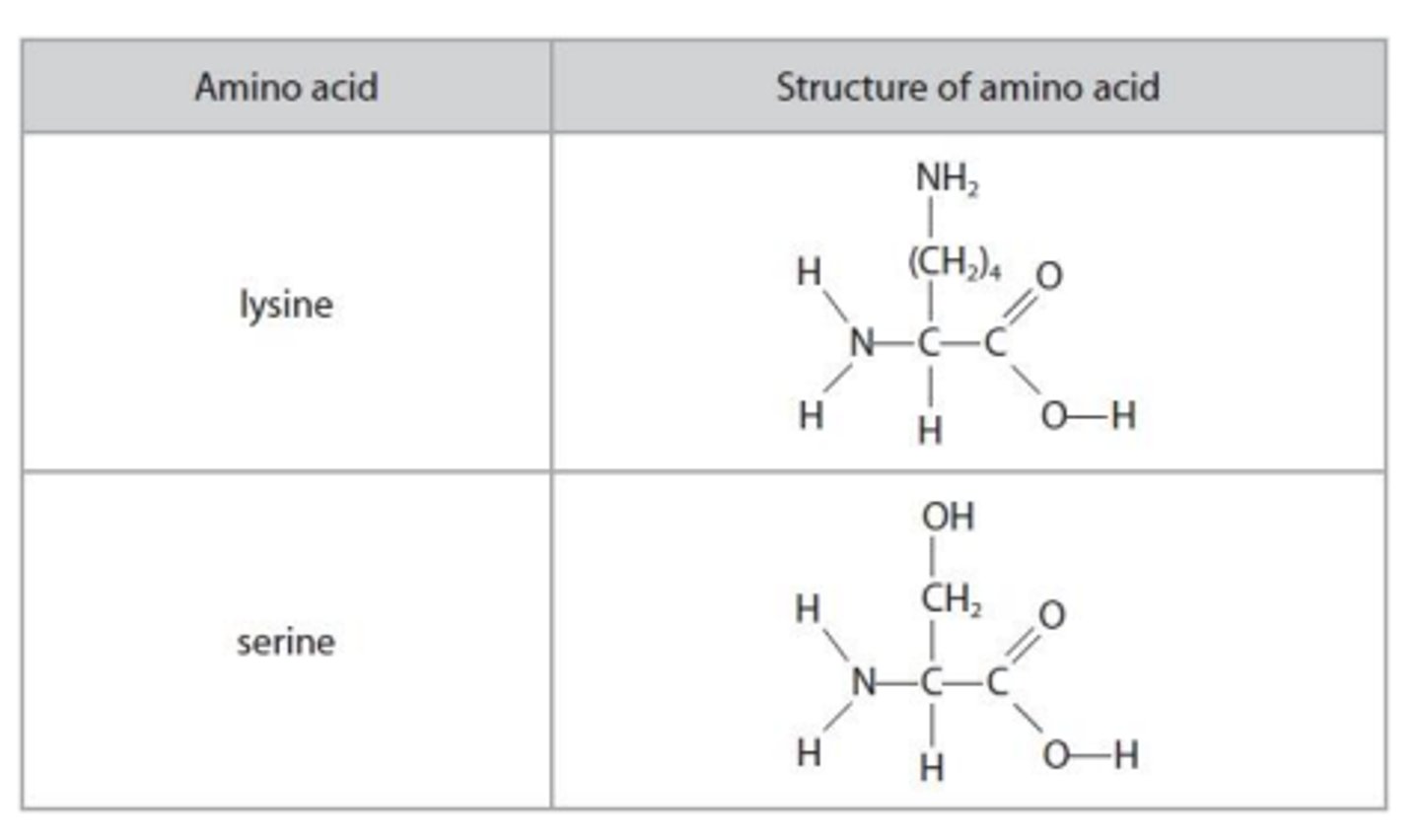
explain the difference in the volumes of 0.010 mol dm–3 hydrochloric acid required to completely react with separate 10.0 cm3 samples of aqueous lysine and of aqueous serine, both of concentration 0.010 mol dm–3 .
- lysine requires twice the volume of HCl
- since lysine has 2 amine group whereas serine only has one