CHEM 1311 Midterm 3
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
ideal gas law formula
PV=nRT
change in energy formula
ΔE=q+w
When heat is absorbed, it is called?
endothermic
When heat is released, it’s called?
exothermic
work (w) is positive (+) when..?
work is done ON the system
work (w) is negative (-) when..?
when the SYSTEM does work
Endothermic
ΔE>0 (energy gain)
q is positive (+)
Exothermic
ΔE<0 (energy loss)
q is negative (-)
Calorimetry is?
measuring heat
Heat capacity (C)
C = q/Δt
Specific heat capaity (c or s)
q needed to raise T by 1 degree Celsius
c = q/mΔt
Specific heat capacity for H2O
4.184 J/C*g
heat energy formula
q=mcΔT
heat energy for a calorimeter
mcΔT = CcalΔT + mCH2O ΔT
Charles’s Law
V1/T1 = V2/T2
V is directly proportional to T
As gas temperature increases, gas volume increases
Boyle’s Law
P1V1 = P2V2
P is inversely proportional to V
As gas pressure increases, gas volume decreases
Avogadro’s Law
V1/n1 = V2/n2
V is directly proportional to n
As the number of moles of gas increases, gas volume increases
Combined Law
(P1V1)/T1 = (P2V2)/T2
V is directly proportional to T/P
Moles formula
mass/molar mass
1 atm equals?
760 torr
760 mm Hg
101.3 kPa
14.7 psi
Dalton’s Law of Partial Pressure: For a mixture of gases in a container…,
Ptotal = P1 + P2 +P3 +…
density formula
d = ((MW)P) / (RT)
root mean square velocity of gases (rms)
urms = rt(3RT/M) in m/s
Graham’s Law
The rate of effusion and diffusion of a gas are inversely proportional to the square root of its molar mass.
Lighter gas molecules move faster and effuse/diffuse quicker than heavier molecules. The law applies to gases at constant temperature and pressure
As mass increases, speed decreases
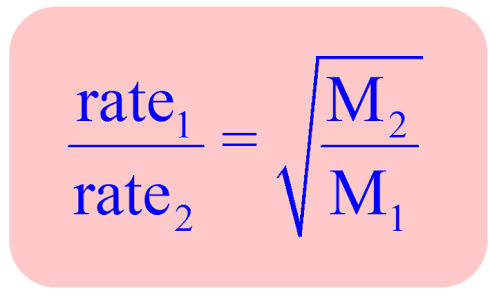
kinetic molecular theory
If temp ↑, then kinetic energy ↑, and rate of collisions ↑ and vice versa
KMT: If volume is decreased..?
everything is the same, but collisions ↑ (V decreased)
KMT: If temp is increased…?
Kinetic energy ↑, velocity ↑, collisions ↑
KMT: If number of moles increase…?
everything is the same but collisions ↑
Kinetic energy formula
KE = 1/2mv²
Gases behave ideally at?
High temps and low pressures
What is the standard temperature and pressure (STP)?
temp: 273 K
pressure: 1 atm
volume: 22.4L
what are intermolecular forces?
forces between MOLECULES
London dispersion forces (LDF)
anything with electrons
nonpolar molecules will ONLY have LDFs
anything with just C and H is nonpolar
Dipole-dipole
polar covalent bond
POLAR
hydrogen bonding
H bonded with N, O, F
ion-dipole
ionic bonds
strength of IMFs from weakest to strongest
LDF < dipole-dipole < hydrogen bonding < ion-dipole
Stronger IMFs mean?
higher melting/boiling point
heavier/bigger elements and molecules
higher viscosity
lower vapor pressure
Larger molecules mean?
Higher: surface tension, viscosity, boiling point
stronger IMFs
more molecular mass → attractive forces stronger
enthalpy of vaporization is greater for bigger molecules
A less polar molecule would have a…?
lower viscosity
If a molecule is nonpolar, it will have…?
a lower surface tension
work formula
w = -PΔV
Enthalpy formula
ΔH = ΔE + w
ΔHrxn
ΔHrxn = ΔHproducts - ΔHreactants
ΔH =
ΔH = [(energy of bonds broken) - (energy of bonds formed)]
What is Hess’s Law?
ΔH = ΔH1+ΔH2+ΔH3+…
qcalorimeter
=CcalorimeterΔT
mole fraction formula using pressure
X1 = P1/PTotal
mole fraction formula using moles
X1 = n1/nTotal
KEavg
3/2 RT
What is the triple point on a phase change diagram?
the temp and pressure where all three phases are in equilibrium
What is the critical point?
The point at the end of the liquid-gas boundary, beyond this point substances become supercritical fluids
solid → gas
sublimation
gas → solid
deposition
solid → liquid
melting (fusion)
liquid → solid
freezing
liquid → gas
vaporization
gas → liquid
condensation