paper 1 chem
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
requirements for 2 solutions to mix
- the energy required to break the intramolecular forces must be similar or less than the energy required to form intermolecular forces between the 2 solutions
- eg water and hexane do not mix well as water has strong hydrogen bonding but hexane only has weak LDF
- so only new LDFs can form between them which is not energetically favorable
explain why 2-methylpentane can dissolve in hexane but not water. (3)
- 2-methylpentane cannot form hydrogen bonds with water so insoluble
- soluble in hexane as LDF in both compounds are similar in strength and magnitude hence they mix well
explain why potassium bromide can dissolve in water but not in hexane. (3)
- KBr is ionic so when dissolved in water, the ions (K+ and Br-) become hydrated
- this is when the oxygen end of water is attracted to K+, while the hydrogen end of water is attracted to Br-
- the energy released from this hydration causes the ionic lattice to break apart
- doesn't dissolve in hexane since LDF forces are much weaker than the ionic forces holding KBr together
the common oxidation numbers of chromium are +2, +3, +6.
give a reason, in terms of ionisation energies, why chromium can show variable oxidation numbers. (1)
Cr configuration is [Ar] 3d5 4s1
gradual increase in successive ionisation energy due to similar shielding in 3d5 and 4s1 orbital
![<p>Cr configuration is [Ar] 3d5 4s1</p><p>gradual increase in successive ionisation energy due to similar shielding in 3d5 and 4s1 orbital</p>](https://knowt-user-attachments.s3.amazonaws.com/31886615-f35a-4380-bd7a-ef724a8a381b.jpg)
2Mg(NO₃)₂ → 2MgO + 4NO₂ + O₂
explain, in terms of all relevant oxidation number, why this is redox. (3)
to find the oxidation number of 2Mg(NO₃)₂:
consider Mg = +2 and NO₃⁻: as O is -6, N must = +5
N is reduced from +5 to +4
O is oxidised from -2 to 0
explain why [Zn(H₂O)₆]²⁺ is colourless. (2)
- zinc has a full 3d subshell
- so d-d transitions cannot take place
the data show the enthalpy changes of reaction when 1 mol of different acids is neutralised by NaOH solution.
comment on the relative enthalpy changes of reaction, using the data below and include any relevant equations. (6)
HCl: -58 kJ mol-1
HNO₃: -58 kJ mol-1
H₂SO₄: -115 kJ mol-1
CH₃COOH: -56 kJ mol-1
HCl and HNO₃
- same value as they are strong acids that completely dissociate into ions in solution
- H⁺ + OH⁻ → H₂O
H₂SO₄
- H₂SO₄ is diprotic so 1 mol provides 2H⁺
- hence value is 2x that of HCl / HNO₃
CH₃COOH
- weak acid so only partially dissociates into ions
- CH₃COOH ⇌ CH₃COO⁻ + H⁺
- some energy is needed to break OH bonds (endothermic) to release H⁺ so enthalpy change of neutralisation is less than strong acids
glycolic acid (HOCH₂COOH) has an acid dissociation constant of 1.5x10⁻⁴ compared with a value of 1.7x10⁻⁵ mol dm-3 for ethanoic acid (CH₃COOH).
give a possible explanation as to why the value of Ka for glycolic acid is 10x larger than that of ethanoic acid. (2)
- the hydroxyl group -OH in glycolic acid is electron withdrawing, which means it pulls the e- density away from the rest of the molecule
- while the methyl group -CH₃ in ethanoic acid is electron donating, which means it pushes e- towards the rest of the molecule
- for an acid to be strong, its anion (eg CH₃COO⁻, HOCH₂COO⁻) must be stable
- an electron withdrawing group is able to spread out the negative charge of the COO⁻, making it more stable so its comfortable existing as a negative ion
- a stable anion has a weaker O-H bond so H⁺ can be lost more easily
2MnO₄⁻ + 5C₂O₄²⁻ + 16H⁺ → 2Mn²⁺ + 10CO₂ + 8H₂O
the reaction starts slowly, then rate of reaction increases, before it decreases again. explain this sequence. (3)
- initial rate is slow because both reacting species are negatively charged
- rate of reaction increase due to formation of Mn²⁺ ions which act as an autocatalyst
- the rate decreases as reactants are used up
give a reason why a solid is not included in the expression for Kc. (1)
because the concentration of a solid is constant
LiCl theoretical lattice energy: -845
LiCl experimental lattice energy: -848
MgI₂ theoretical lattice energy: -1944
MgI₂ experimental lattice energy: -2327
comment on the theoretical and experimental lattice energy values, giving the reasons for any differences and similarities. (6)
- LiCl is almost 100% ionic
- while difference in theoretical and experimental value suggest that MgI₂ has some covalent character
- since Mg²⁺ has a higher charge density than Li⁺ so it has a larger polarising power
- while I⁻ is larger than Cl⁻ so it is more easily polarised
give a reason why the catalysis of Fe²⁺ is possible. (1)
due to variable oxidation number in TM
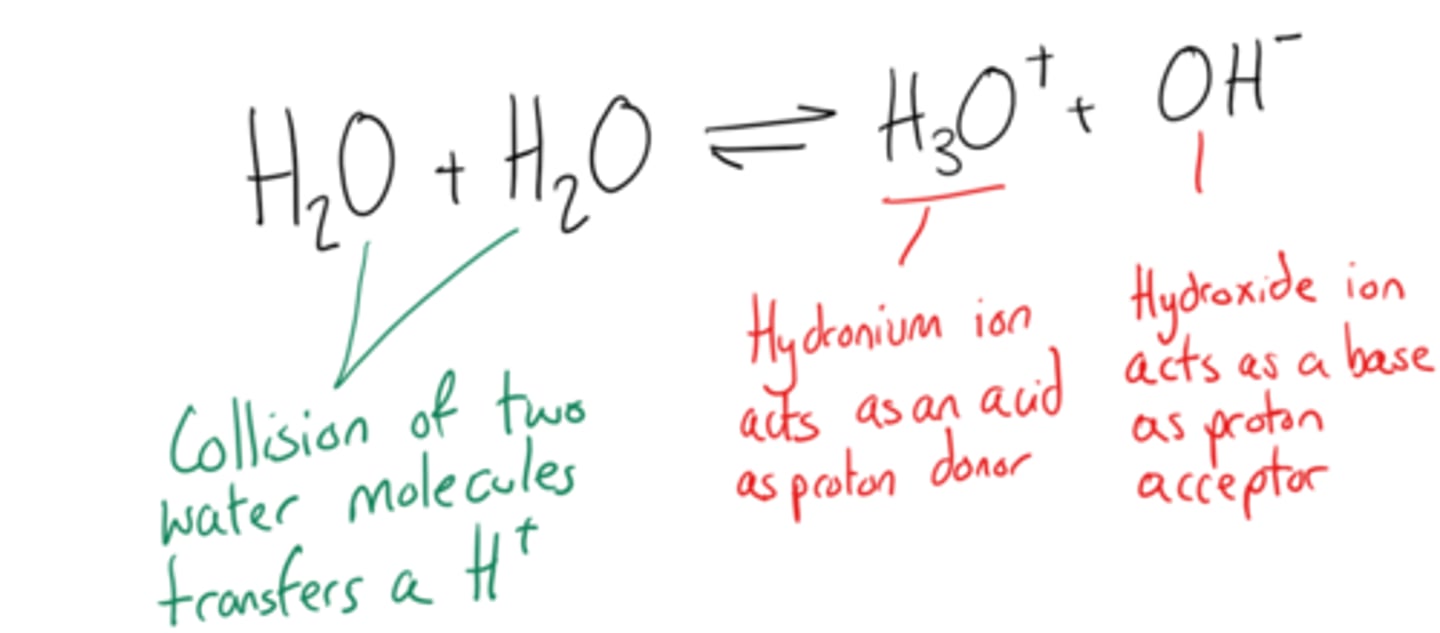
predict, with reason, whether water is acidic, alkaline or neutral.
neutral since [OH⁻] = [H⁺]
predict, with reason, the sign of the enthalpy change for the ionisation of water. (1)
- positive (endothermic)
- as bond breaking requires energy
compare and contrast the origin of colour of a copper(II) complex with the origin of colour of a copper(II) ion flame test. (6)
- difference in energy levels determines colour of complex ion and colour in flame test
- in a flame test, the heat energy cause electrons to become promoted to an excited state
- the return of excited electron to lower energy state result in colour
- in a complex ion, d orbitals are split in energy by ligands
- d-d transitions cause different frequency of visible light to be absorbed
- light that is not absorbed is transmitted to give colour
rank the lattice energies of the hydrides NaH, KH and MgH₂
MgH₂ > NaH > KH
lattice energy of MgH₂ should be 2x more than that of NaH and KH because higher charge of Mg²⁺ and 2H⁻
NaH is more exothermic than KH because Na⁺ is smaller than K⁺, leading to stronger electrostatic attraction with H
one of the systems controlling the pH of blood is the carbonic-acid hydrogencarbonate buffer system.
H₂CO₃ ⇌ H⁺ + HCO₃⁻
explain how this buffer system helps to control the pH of blood when extra CO2 is present due to strenuous exercise. (3)
- carbon dioxide dissolved in the blood forms carbonic acid (H₂CO₃)
- which causes the equilibrium to shift the the RHS so more H⁺ is formed
- reservoir of HCO₃⁻ combine with H⁺ to control blood pH
[Cr(H₂O)₆]³⁺ + EDTA⁴⁻ ⇌ [CrEDTA]⁻ + 6H₂O
the Kc is 2.51 x 10²³
by considering the equilibrium for this reaction and changes in entropy, comment on the value of the equilibrium constant. (3)
- Kc is larger which suggest that the equilibrium lies to the right
- 2 mol of reactant form 7 mol of products
- this increases entropy of the system
give 2 reasons why you would expect the ∆S(sys) to be positive. (2)
- more gas moles on the right
- gas and liquid is formed from solids
a commercial nitric acid solution, HNO₃, has a concentration of 15.9 mol dm-3.
a 15cm3 sample was made up to 100cm3 by adding deionised waetr.
calculate the pH of this solution. (2)
initial vol: 15
final vol: 100
initial conc: 15.9
final conc: ?
so 15 x 15.9 = 100 x ?
so final conc = 2.385
name 2 properties of ions that affect their enthalpy change of hydration
- ionic radius
- ionic charge
HCl is addded to aqueous Na₂CO₃.
the reaction takes place in 2 steps because CO₃²⁻ is diprotic.
the equation taking place in the first step is Na₂CO₃ + HCl → NaHCO₃ + NaCl.
write the equation for the reaction taking place during the second equivalent point. (1)
HCO₃⁻ + H⁺ → CO₂ + H₂O
Na₂CO₃ + HCl → NaHCO₃ + NaCl.
explain how the solution can act as a buffer. (3)
- large resevoir of acid and its conjugate base (CO₃²⁻ and HCO₃⁻
- so CO₃²⁻ can react with added H⁺ (CO₃²⁻ + H⁺ → HCO₃⁻)
- while HCO₃⁻ can react with added OH⁻ (HCO₃⁻ + OH⁻ → CO₃²⁻ + H₂O)
define the term orbital (2)
- a region where theres a 95% chance of finding an electron
- contain a pair of e- with opposite spins