REAL Spring 2026 Cumulative Smithrud Biochem (CHEM 3040) Final
1/99
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
100 Terms

C
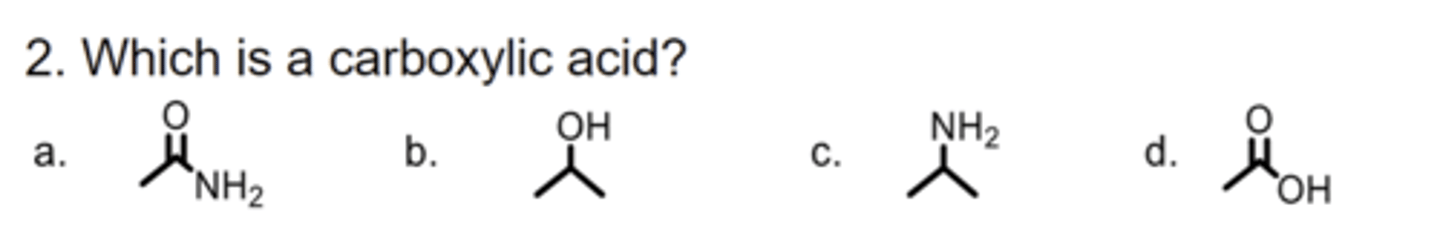
D
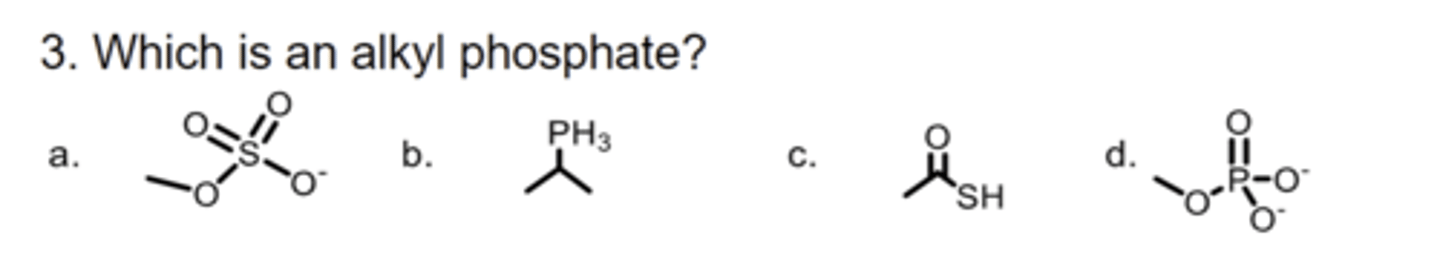
D

B

A

A
7. Which bond has shared electrons? (a) noncovalent (b) shared (c) covalent (d) polar (e) dipolar
C
8. Which bond type is not strongly affected by the environment? (a) ionic (b) H-bond (c) covalent(d) dipole-dipole (e) dispersion
C
9. Which type of bond does not generally form between proteins and ligands when they bind (a)salt bridge (b) H-bond (c) covalent (d) dipole-dipole (e) dispersion
C
10. Which bond gives more possible conformations (a) carbon-carbon (b) carbon-nitrogen (c)phosphodiester (d) van der Waals interactions
D
11. Rank the length of the bonds from longest to shortest: 1-amide bond, 2-carbon nitrogen doublebond, 3-carbon nitrogen single bond, 4-H-bond between amides
a. 1 > 3 > 4 > 2
b. 3 > 1 > 4 > 2
c. 3 > 4 > 1 > 2
d. 1 > 4 > 3 > 2
e. 4 > 3 > 1 > 2
E
12. Which is the weakest bond in the gas phase?
a. dipole-dipole
b. hydrogen
c. dispersion
d. ionic
C
13. Which is the strongest bond in the gas phase?
a. dipole-dipole
b. hydrogen
c. dispersion
d. ionic
D

C

A

D

E
18. What type of bond requires random shifts in e-clouds to form?
a. H-bond
b. ionic
c. salt bridge
d. dipole-dipole
e. dispersion
E
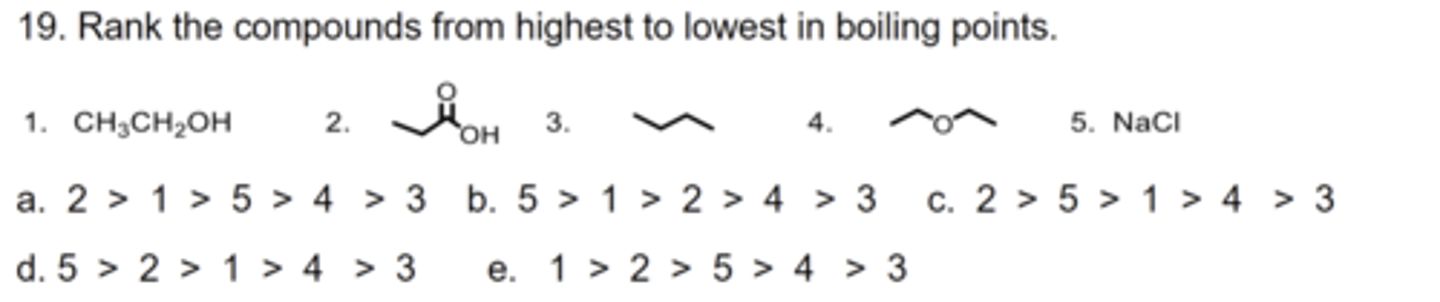
D
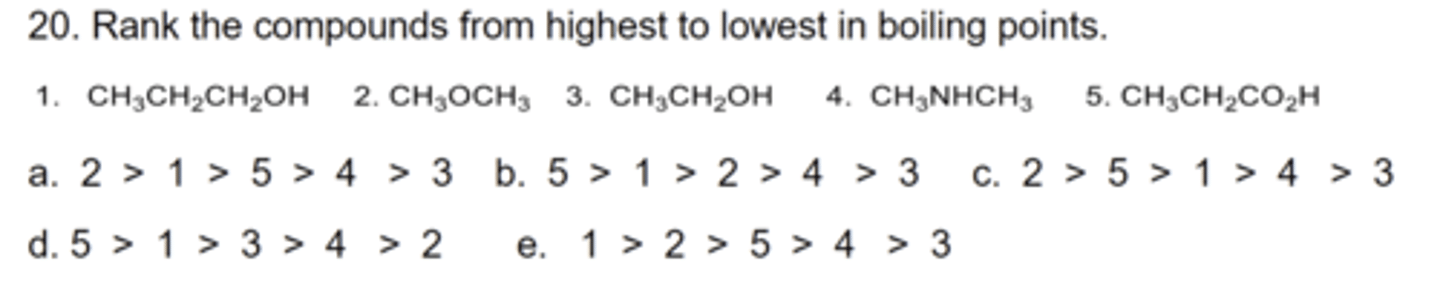
D
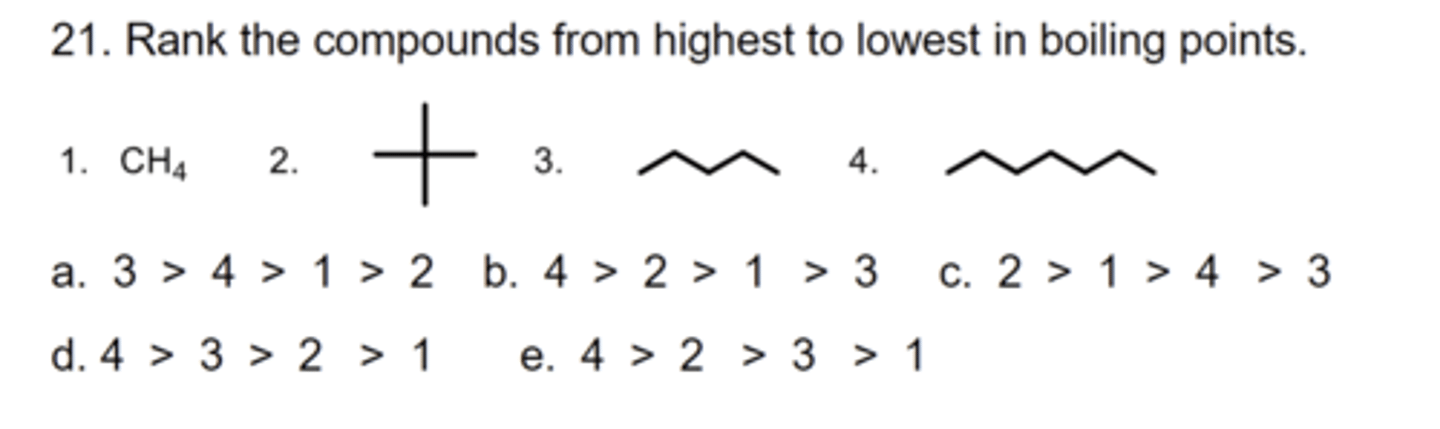
D
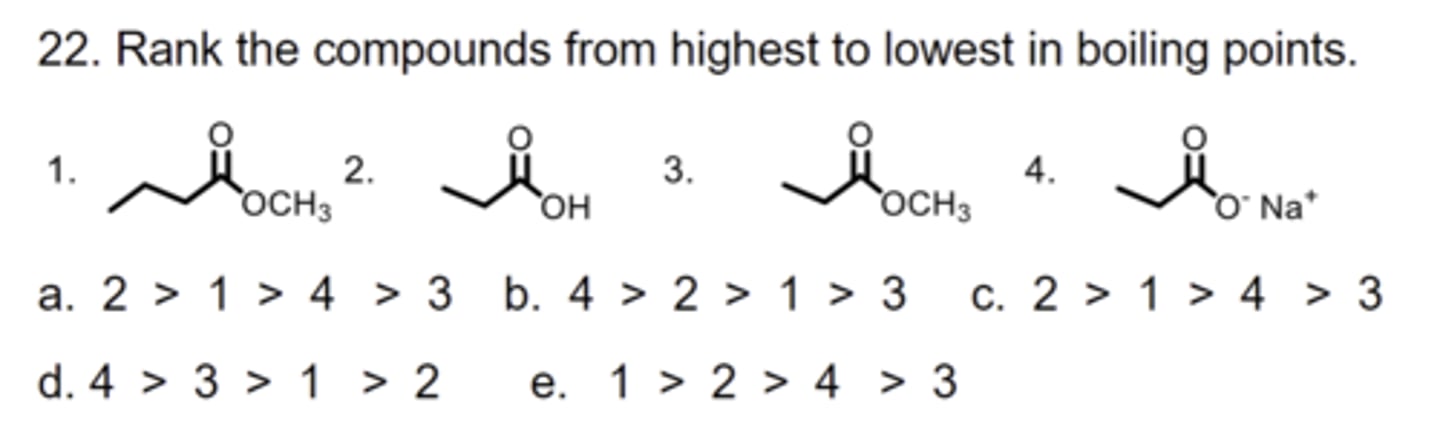
B
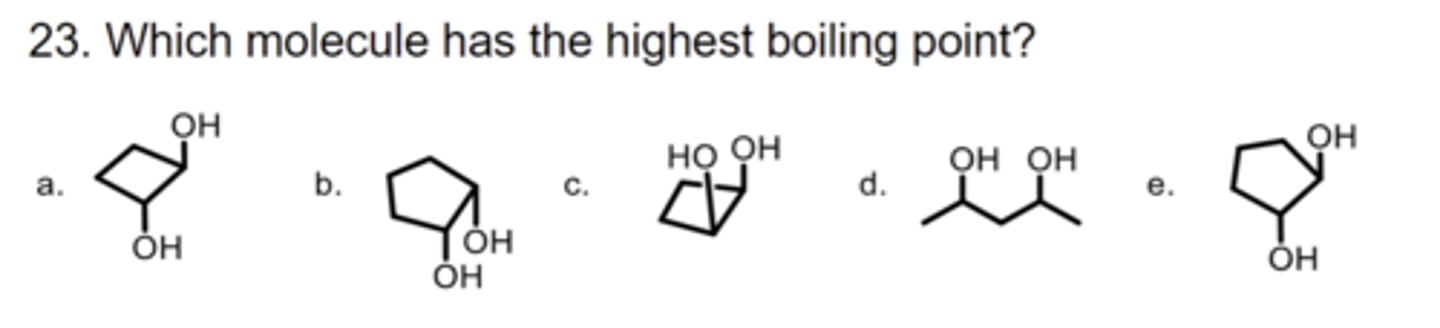
E
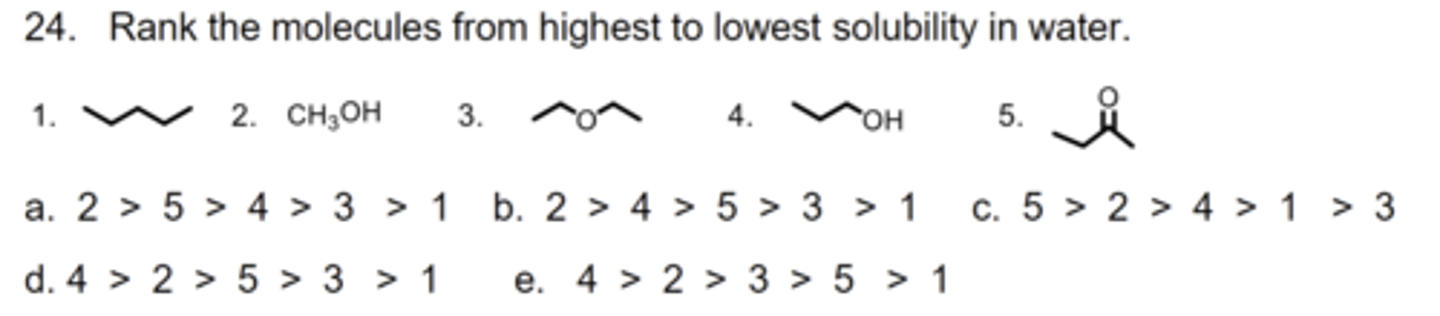
B
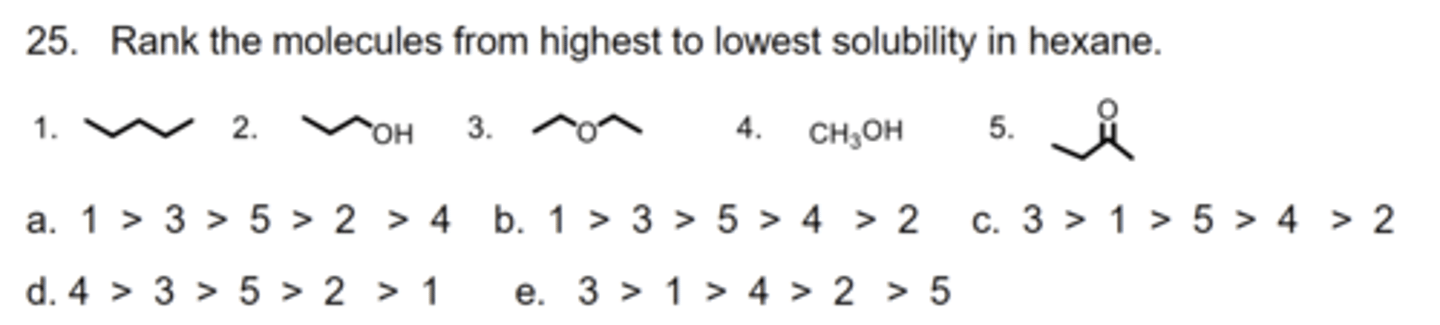
A
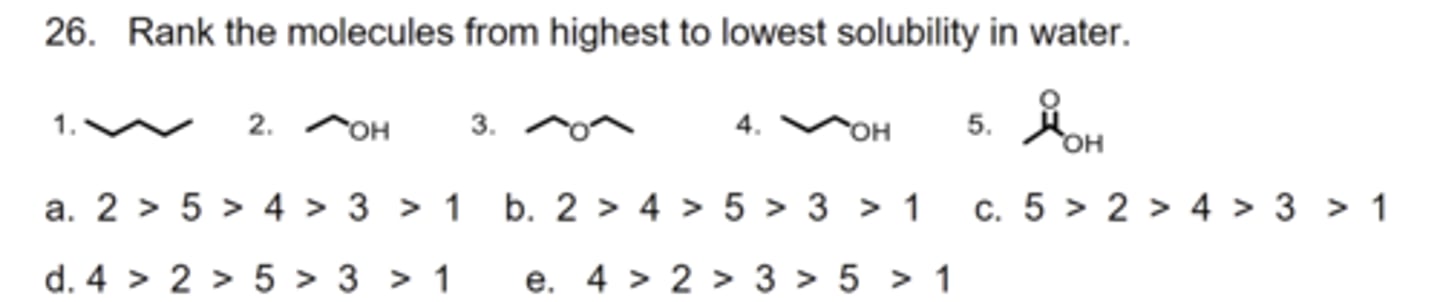
C
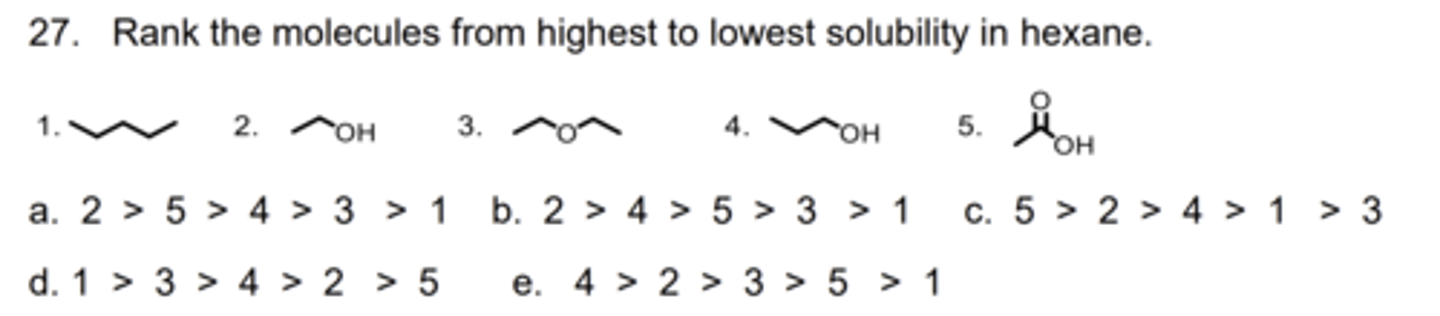
D
28. Which is more soluble in water: NaCl or ethanol?
a. NaCl, full charges bind stronger to water
b. Ethanol, its H-bonds form stronger bonds to water
c. NaCl, it has two charged groups
d. Ethanol, breaking NaCl's ionic bond costs more energy
D
29. Which is more soluble in water: ethanol or acetic acid
a. Ethanol, it has more hydrogen bond acceptors
b. Ethanol, it has more hydrogen bond donors
c. Acetic acid, it has more hydrogen bond acceptors
d. Acetic acid, it has more hydrogen bond donors
C
30. Which is more soluble in water: ethanol or diethyl ether
a. Ethanol, it has more hydrogen bond acceptors
b. Ethanol, it has a hydrogen bond donor
c. Diethyl ether, it has more hydrogen bond acceptors
d. Diethyl ether, it has a hydrogen bond donor
B

A
32. Which system has the highest entropy?
a. ice
b. water 25 C
c. water at 75 C
d. water at 40 C
C
33. Which state has the highest entropy?
a. gas
b. liquid
c. solid
A
34. Which system has the lowest entropy?
a. ice
b. water 25 C
c. water at 75 C
d. water at 40 C
A
35. Which state has the lowest entropy?
a. gas
b. liquid
c. solid
C
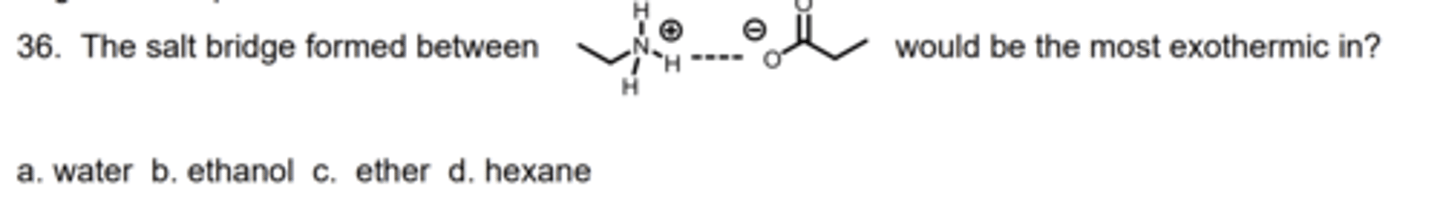
D

A
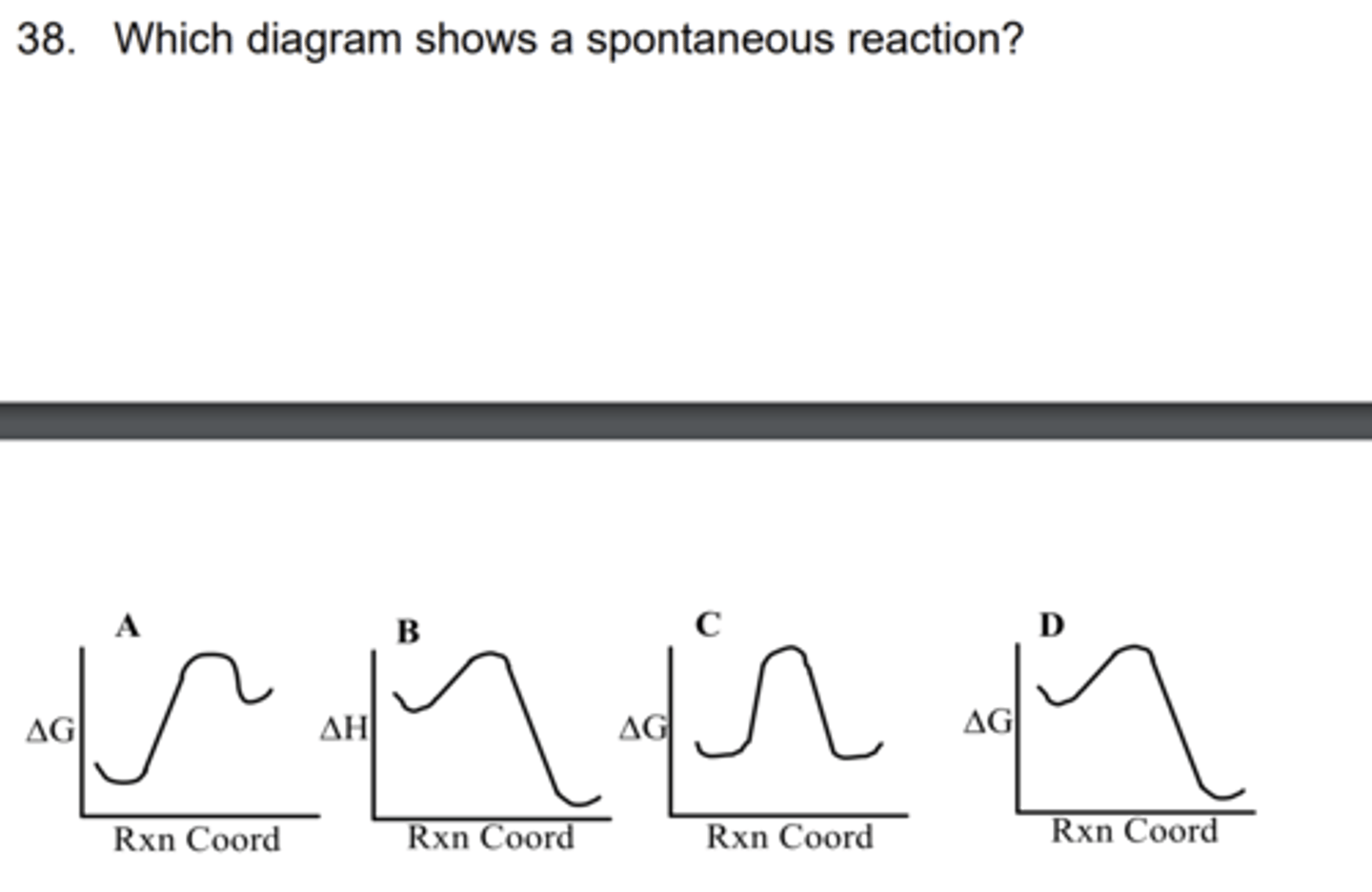
D
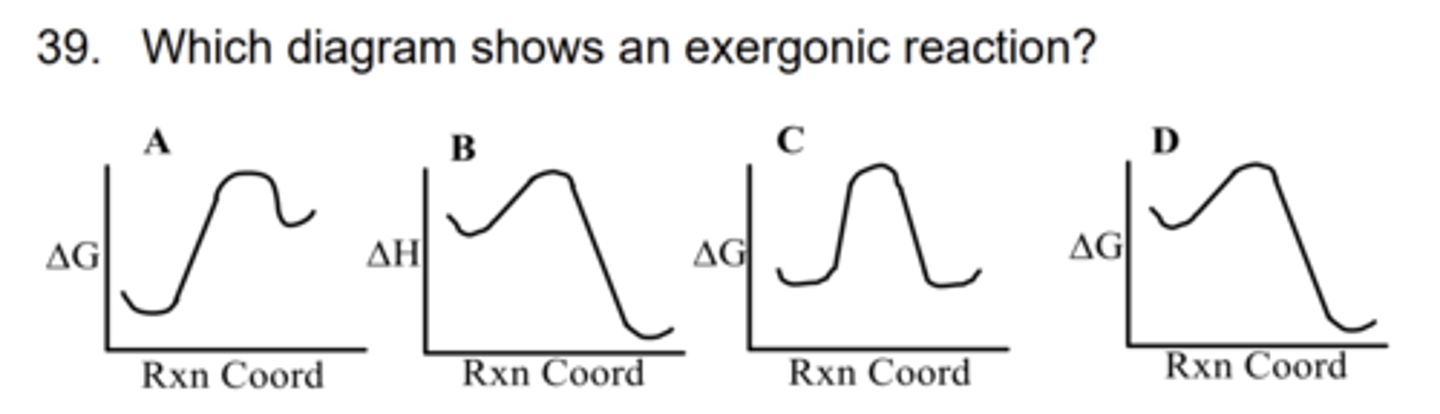
D
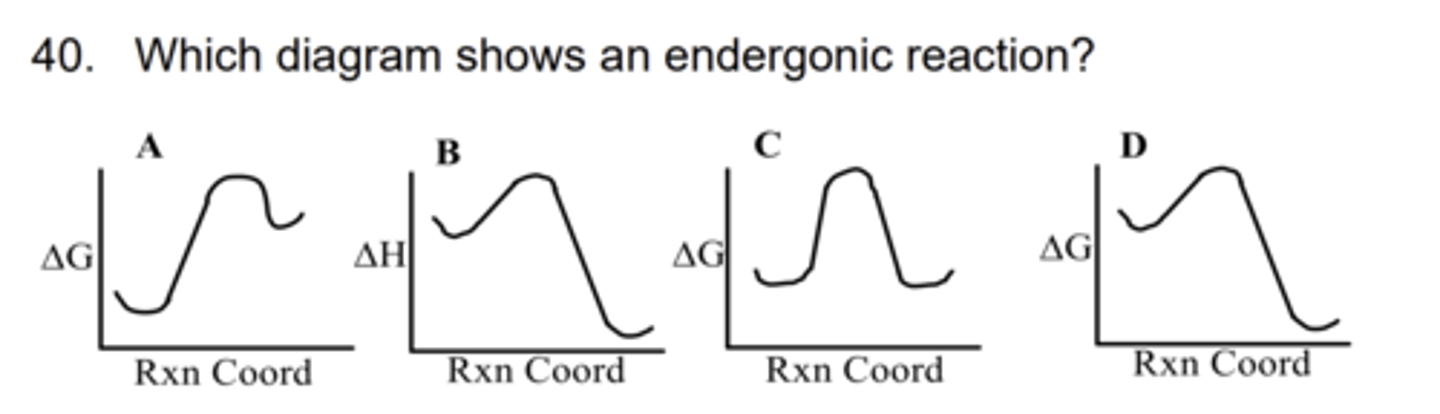
A
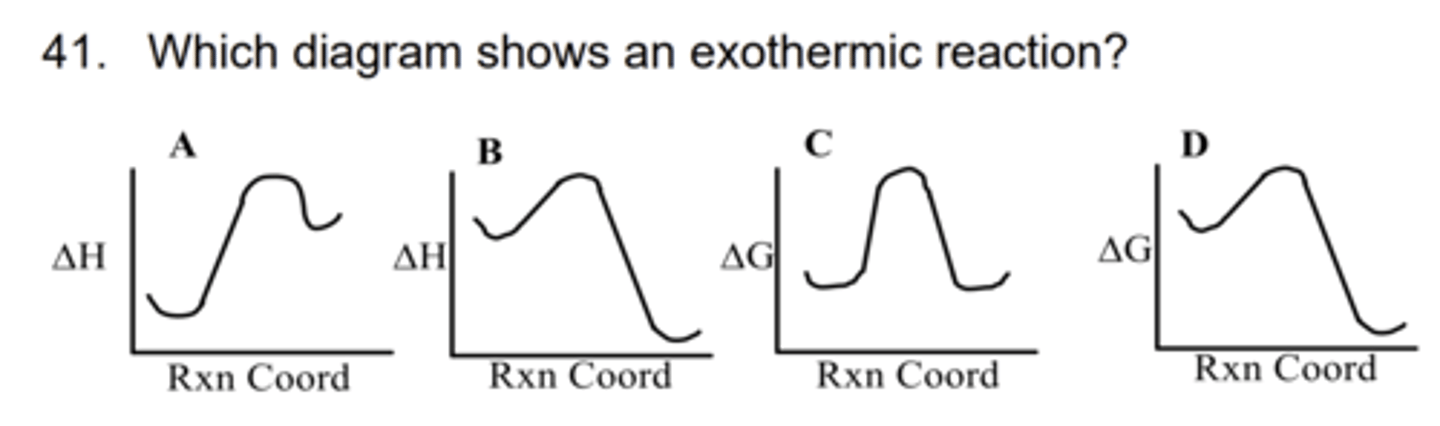
B
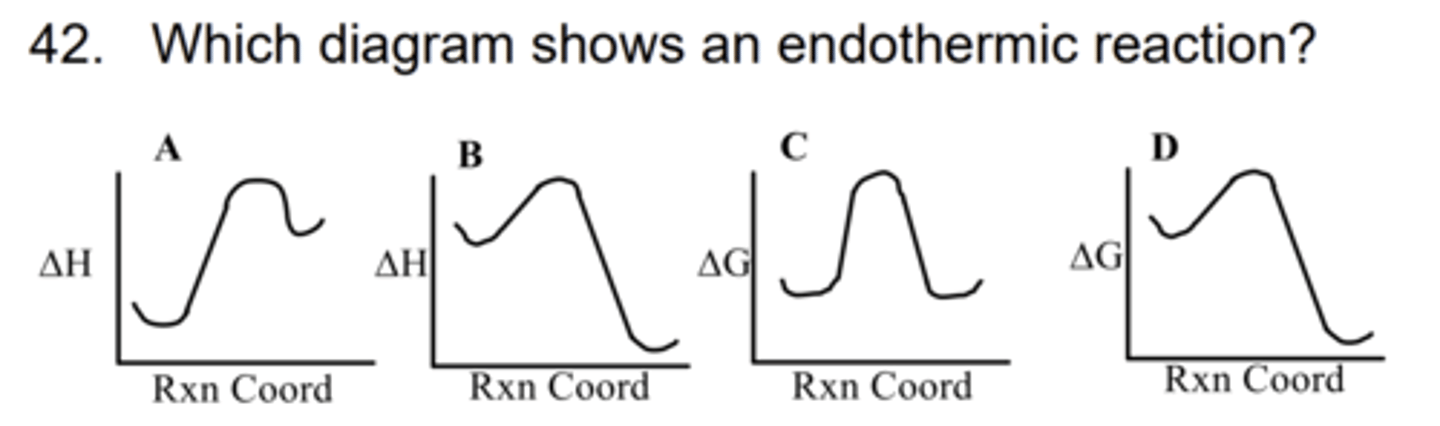
A
43. Which statement is consistent with the 2nd law of thermodynamics?
a. The entropy of the surrounding must increase
b. A hot object will spontaneously warm a cold object
c. The entropy of the system must decrease
d. The entropy of the universe must stay the same
e. Both answers a and b
B
44. What is the first law of thermodynamics?
a. systems go to their lowest energy states
b. energy can be created not destroyed
c. energy cannot be created only destroyed
d. energy cannot be created or destroyed
e. energy is constant in the system
D
45. Which outcome breaks the second law of thermodynamics?
a. the universe loses energy
b. a puzzle falls to pieces when dropped
c. the universe gains matter
d. a cold object spontaneously heats a warm object when in contact.
D
46. Which is true for a spontaneous process in a system?
(a) heat is consumed
(b) heat is released to the surrounding
(c) heat can be consumed or released
(d) heat stays constant
C
47. Which is true for a spontaneous process in a system?
(a) the system becomes more
disordered
(b) the system could become more or less disordered
(c) the system becomes more
ordered
(d) disorder of the system remains constant
B
48. Which is true for a spontaneous process in a system?
(a) the surrounding becomes more disordered
(b) the surrounding could become more or less disordered
(c) the surrounding becomes more ordered
(d) disorder of the surrounding remains constant
B
49. When change happens, the entropy of the universe must
(a) increase
(b) decrease
(c) increase or decrease
(d) stay constant
A
50. When change happens, the entropy of the surrounding must
(a) increase
(b) decrease
(c) increase or decrease
(d) stay constant
C
51. Do proteins have high heat capacities / why?
a. yes, they have many conformations
b. no, they have very stable structures
c. no, they have many conformations
d. yes, they have very stable structures
A
52. Entropy is a measure of
a) probability
b) disorder
c) heat distribution
d) answers (a) and (b)
e) all the above
E
53. All processes must be favorable under a specific condition if they are
a. exothermic
b. endothermic
c. exergonic
d. endergonic
e. more free
C
54. The bond between Na+ and Clis weakest in
a. hexane
b. water
c. methanol
d. acetone
B
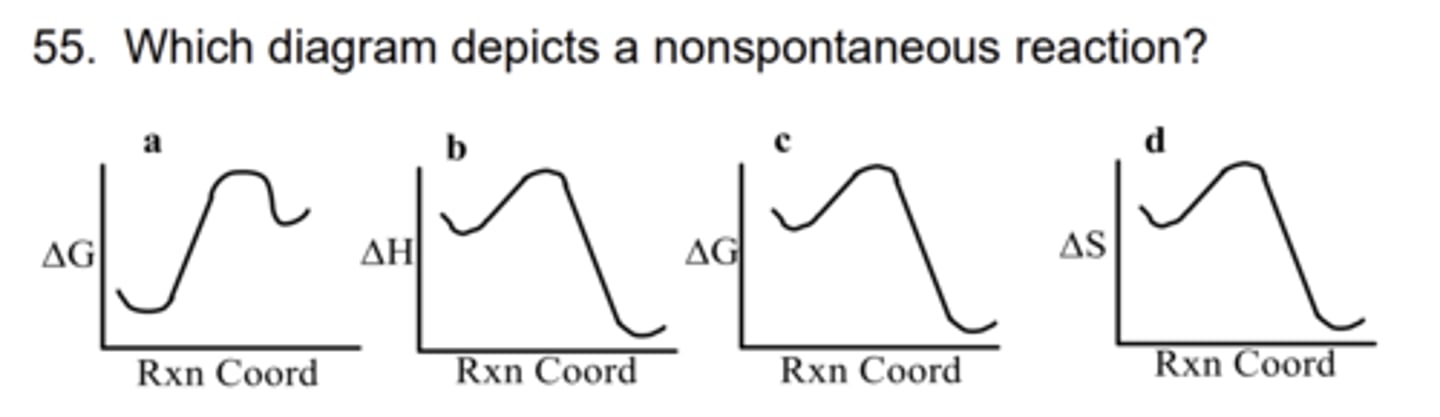
A
56. Which functional group can form a off-faced stack arrangement?
a. carboxylic acid
b. amides
c. alcohol
d. arenes
e. amines
D
57. The Gibbs free energy for a chemical process was -8 kcal/mol and the heat produced was -5
kcal/mol. What was the TS term for this process?
a. -3 kcal/mol
b. 3 kcal/mol
c. -13 kcal/mol
d. 13 kcal/mol
B
58. The enthalpy for a chemical process was 2 kcal/mol and TS was 5 kcal/mol. What was the
Gibbs free energy (G) term for this process?
a. -3 kcal/mol
b. 3 kcal/mol
c. -7 kcal/mol
d. 7 kcal/mol
A
59. The Gibbs free energy for a chemical process was -5 kcal/mol and TS was -2 kcal/mol. What
was the enthalpy for this process?
a. -3 kcal/mol
b. 3 kcal/mol
c. -7 kcal/mol
d. 7 kcal/mol
C
60. The Gibbs free energy for a chemical process was 4 kcal/mol and the heat produced was 3
kcal/mol. What was the TS term for this process?
a. -7 kcal/mol
b. 7 kcal/mol
c. -1 kcal/mol
d. 1 kcal/mol
A
61. The Gibbs free energy for a chemical process was -13 kcal/mol and TS was -2 kcal/mol.
What was the enthalpy for this process?
a. 15 kcal/mol
b. 11 kcal/mol
c. -11 kcal/mol
d. -15 kcal/mol
D
62. A thermometer measures
(a) thermo energy
(b) average kinetic energy
(c) potential energy
(d) heat
B
63. Which reaction can be coupled to A + B = C + D G = 3 kcal/mol?
a. C + E = F G = -2 kcal/mol
b. F + B = G G = -5 kcal/mol
c. F + G = H G = -3 kcal/mol
d. D + F = G G = -5 kcal/mol
D
64. The bond between Na+ and Clis strongest in
a. hexane
b. water
c. methanol
d. acetone
A
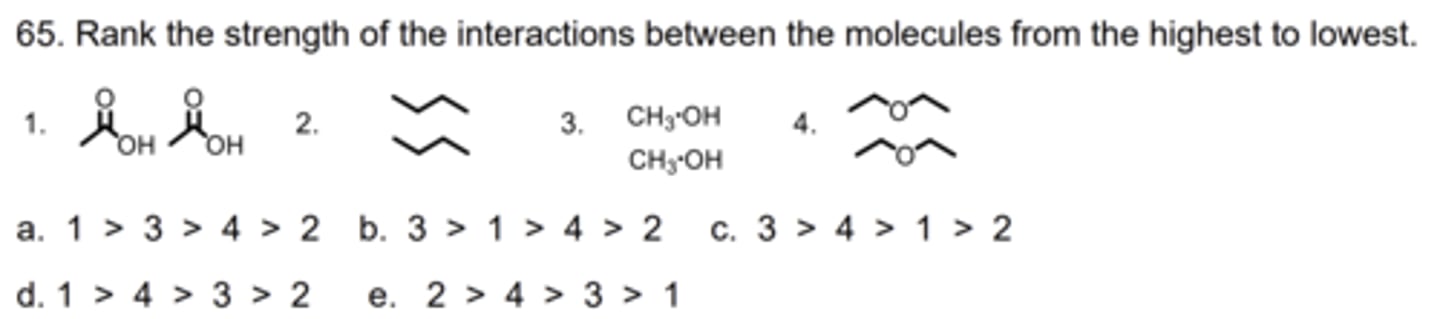
A

A
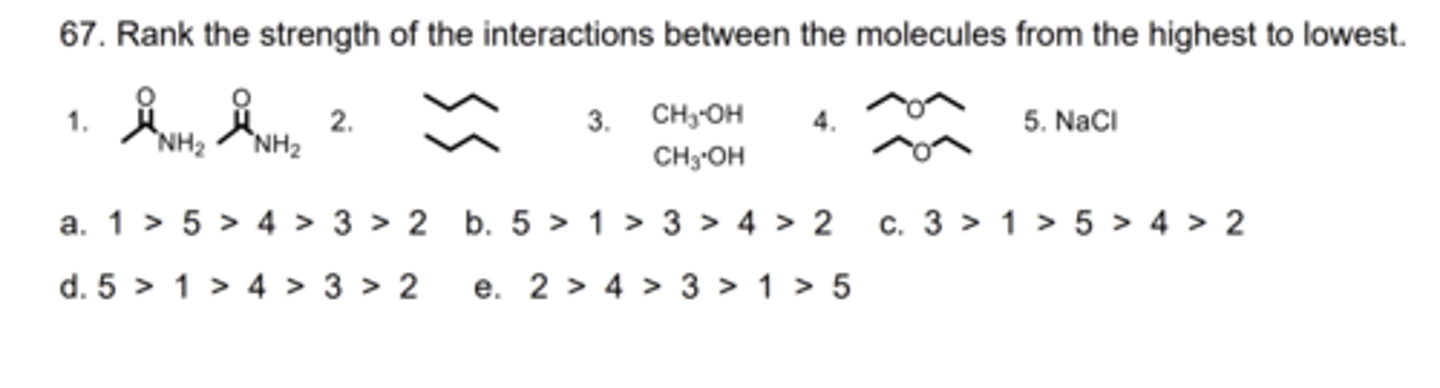
B
68. Which electrostatic interaction is less affected by going from the gas phase to water?
a. ionic
b. H-bond
c. dipole
d. dispersion
D
69. Which functional group can form a T-stack arrangement?
a. carboxylic acid
b. arene
c. alcohol
d. amides
e. amines
B
70. Which sign/parameter shows an exothermic process?
a. S
b. H
c. -G
d. -S
e. −H
E
71. Which sign/parameter shows an exergonic process?
a. S
b. H
c. -G
d. -S
e. −H
C
72. Which sign/parameter shows an endergonic process?
a. G
b. H
c. -G
d. -S
e. −H
A
73. Which sign/parameter shows a gain in freedom during a process?
a. S
b. H
c. -G
d. -S
e. −H
A
74. Which sign/parameter shows a loss of freedom during a process?
a. S
b. H
c. -G
d. -S
e. −H
D
75. Which sign/parameter shows an endothermic process?
a. S
b. H
c. -G
d. -S
e. −H
B
76. Which parameter gives the distribution of heat in a system?
a. w
b. H
c. G
d. S
e. q
D
77. Which is the first law of thermodynamics?
a. Energy can be created and destroyed
b. Energy cannot be created or destroyed
c. Energy can be destroyed and not created
d. Energy can be created and not destroyed
B
78. Which statement is consistent with the 2nd law of thermodynamics?
a. The entropy of the universe cannot be destroyed
b. The entropy of a system always increases
c. Cold objects will spontaneously give energy to warm objects
d. Freedom of motion of a system can decrease
D
79. A thermometer measures
a. pressure
b. heat
c. average kinetic energy
d. molecular conformations
C
80 Which is not wasted heat?
a. Conformational changes
b. Increased molecular vibrations
c. Breaking H-bonds
d. Expanding glassware
D
81. Which system has the highest entropy?
a. CH4 at 30 C
b. H2O at 30 C
c. H2O at 50 C
d. Sand at 30 C
A
82. A spontaneous reaction
a. Has a positive free energy
b. Makes less stable molecules
c. Makes more stable molecules
d. Happens immediately once the materials are mixed
e. Both c and d
C
83. Which reaction can be coupled to A + B = C + D G = 3 kcal/mol?
a. C + E = F G = -2 kcal/mol
b. F + B = G G = -5 kcal/mol
c. F + G = H G = -3 kcal/mol
d. D + F = G G = -5 kcal/mol
D
84. Which reaction can be coupled to A + B = C + D G = 3 kcal/mol?
a. C + E = F G = 4 kcal/mol
b. F + B = G G = -5 kcal/mol
c. F + G = A G = -6 kcal/mol
d. D + F = G G = -2 kcal/mol
C
85. Which are symbols for heat
(a) G
(b) H
(c) w
(d) q
(e) b and d
E
86. Entropy is a measure of
(a) disorder
(b) probability
(c) energy distribution
(d) all answers
D
87. A chemical process gives off heat to the surrounding and the system becomes more ordered.
The process is
a. spontaneous
b. nonspontaneous
c. its spontaneity cannot be determined
C
88. A chemical process takes heat from the surrounding and the system becomes more ordered.
The process is
a. spontaneous
b. nonspontaneous
c. its spontaneity cannot be determined
B
89. A chemical process gives off heat to the surrounding and the system becomes more
disordered. The process is
a. spontaneous
b. nonspontaneous
c. its spontaneity cannot be determined
A
90. The Gibbs free energy for a chemical process was -5 kcal/mol and the heat produced was -7 kcal/mol. What was the TS term for this process?
a. -12 kcal/mol
b. 12 kcal/mol
c. 2 kcal/mol
d. -2 kcal/mol
D
91. Which reaction can be coupled to A + B = C + D G = 3 kcal/mol?
a. C + E = F G = -2 kcal/mol
b. D + F = G G = -5 kcal/mol
c. F + G = H G = -3 kcal/mol
d. F + B = G G = -5 kcal/mol
B

B
93. Fe3+ is called a _____ when bound to a protein and is chemically active.
a. substrate
b. coenzyme
c. cosubstrate
d. cofactor
D
94. A organic molecule that is loosely bound by a protein is called a
a. cosubstrate
b. prosthetic group
c. substrate
d. particle
A
95. A organic molecule that is tightly bound by a protein is called a
a. cosubstrate
b. prosthetic group
c. substrate
d. particle
B
96. Which vitamin is responsible for eye health?
a. A
b. B
c. C
d. D
e. E
f. K
A
97. Which vitamin is used to make collagen?
a. A
b. B
c. C
d. D
e. E
f. K
C
98. Which vitamins are water soluble?
a. A
b. B
c. C
d. D
e. E
f. K
B C
99. Which vitamin is responsible for blood clotting?
a. A
b. B
c. C
d. D
e. E
f. K
F
100. Which vitamin is responsible for bone health?
a. A
b. B
c. C
d. D
e. E
f. K
D