AQA A level Chemistry - Le Chatelier's Principle, Equilibrium, Kc
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
what are the characteristics of dynamic equilibrium?
occurs in a closed system
forwards and reverse reactions occur at the same rate
concentrations of reactants and products remain constant
what is a homogenous reaction?
reaction where all reactants and products are in the same phase (state)
define Le Chatelier’s Principle
when a system in equilibrium is disturbed, the position of equilibrium will move in a direction to reduce the effect of a disturbance
how do you include Le Chatelier’s Principle in exam questions?
to oppose the change in x
to reduce the x or remove x etc.
what is the effect of increased pressure on the position of equilibrium?
shifts position of equilibrium to side with fewer gas moles
to reduce pressure
to oppose the change
opposite for decreased
what is the effect of increased temperature on the position of equilibrium?
shifts position of equilibrium in direction of endothermic reaction
to reduce temperature/oppose the change
opposite for decrease
what is the effect of increased concentration of a reagent/product on the position of equilibrium?
position of equilibrium shifts to side with lower concentration of stuff (side conc wasn’t increased on)
in order to oppose the change
what is the effect of a catalyst on the position of equilibrium?
no effect on position of equilibrium
as it increases the rate of the forwards and reverse reactions equally
what is the equation for the Haber Process?
N2 + 3H2 ←> 2NH3
delta H = -92
what is the temperature used for the Haber process and why?
450C
compromise temperature as:
higher temperature increases rate of reaction
as well as shifts position of equilibrium to left :(
what is the pressure used for the Haber process and why?
20MPa
compromise pressure as:
higher pressure shifts equilibrium to the right and increases the yield
but is also very expensive
what is the catalyst used for the Haber process?
finely divided Fe (s)
what is the equation for the manufacture of ethanol from ethene?
C2H4 + H2O ←> C2H5OH
delta H = -42
what is the temperature used for the manufacture of ethanol and why?
300-600C
compromise temperature as:
higher temperature shifts position of equilibrium to left as reaction is exothermic
but high temperature = high rate
what is the pressure used for the manufacture of ethanol and why?
5-10 MPa
compromise pressure as:
high pressure shifts equilibrium to the right
but high pressure is ezpeensive
what is the catalyst used for the manufacture of ethanol?
concentrated phosphoric acid
why are compromise pressures and temperatures used?
compromise temperature used to strike a balance between eq position and rate
compromise pressures used to balance between eq position and cost
what is Kc?
ratio of products to reactants at equilibrium at a set temperature
how would you construct an expression for Kc?
find the units by subbing in the units for each concentration of A, B, C, D into expression and simplify

what does it mean if Kc > 1?
concentration of products is greater than concentration of reactants
equilibrium lies to the RHS
what does it mean if Kc < 1?
concentration of reactants is greater that the concentration of products
equilibrium lies to the LHS
which factors is Kc not affected by?
changes in concentration
volume
pressure
catalyst
due to le chateliers principle
which factor is Kc affected by?
changes in temperature
how does an increased temperature affect Kc?
position of equilibrium shifts in the endothermic direction
so Kc increases for forwards endothermic
how does a decreased temperature affect Kc?
position of equilibrium shifts in the exothermic direction
so Kc increases for forwards exothermic
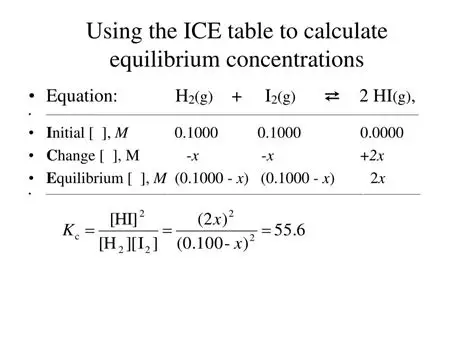
how do you calculate Kc given mole values?
construct I C E table
sub in values given
use values + ratios to work out change in moles from initial to equilibrium - moles always change in same ratio
work out concentrations if necessary
sub values in to expression
might have to do in reverse (find moles) or might have to work it out through titration