Chemistry Exam 4 Study things
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
What is organic chemistry and why does it matter/what are organic molecules?
Organic chemistry: is the studies of the compounds of the element carbon.
It matters because:
Medicines, materials, and most biologically active molecules are organic molecules
Organic molecules react with receptors on your tongue(taste), nose(smells), and brain(everything you brain does)…..those receptors in your cells are made of organic compounds too
What are the 3 most important things to remember about carbon bonds in organic compounds?
1) Carbon will form four bonds to satisfy its octet
2) Carbon always forms covalent bonds
3) Its covalent bonds can be polar when it bonds to elements from the right or left side of the periodic table(C-H bonds are NOT polar)
What is a hydrocarbon? What are the types of hydrocarbons?
A molecule that only contains carbon (C) and hydrogens (H).
Alkanes
Cycloalkanes
Alkenes
Alkynes
Aromatic hydrocarbons
What is an alkane?
Contain only C-C single bonds.
What is a cycloalkane?
Alkanes with C atoms bonded in rings
What are alkenes?
Contain at least one C-C double bonds
What are alkynes?
Contain at least one C-C triple bond
What are aromatic hydrocarbons?
Contain a benzone ring or a derivative of a benzene ring.
What are saturated hydrocarbons?
Have the maximum number of hydrogen atoms attached to each carbon atom(“saturated with hydrogen”)
Are alkanes and cycloalkanes with single C-C bonds
What are unsaturated hydrocarbons?
Have fewer hydrogen atoms attached to the carbon chain than alkanes
Are alkenes with C-C double bonds
Are alkynes with C-C triple bonds
What is the structural formula of organic compounds?
(Lewis structure): Where you show every bond and every atom
Make sure every C has 4 bonds
What is the condensed structure include?
Shows every bond and every atom except H-C bonds. Do not show lone pairs
H atoms that are bonded to the same carbon are group together
Groups that are repeating can be shown with () and subscript
Extra info:
You don’t need to. number carbons in a straight line
Atoms and molecules can rotate and flip
What is the condensed formula include?
Typically, doesn’t show bonds(C-C bonds some time included)
Atoms that are bonded are group together
Groups that are repeating can be shown with () and subscript
Groups that are the same that are bonded to the same carbon can be condenced using ()
What is a bond-line/skeletal formula include?
Shows all bonds except between C-H, show all atoms EXCEPT H and C
This type of formula assumes a carbon atom at any location where lines intersect or end; hydrogens are not drawn
Hydrogens are assumed based on the “4-bonds to carbon” rule (Every corner and every end represents a carbon)
Each carbon in the structure is assumed to be bonded to the correct number of hydrogens to equal 4 bonds.
What is a parent chain/main chain?
The longest continuous chain of carbon atoms in any direction.
Functional Groups: atom or group of atoms in a molecule that defines the chemical properties of the molecule.
Example: Alcohol (-OH)
What are substituents?
An atom or group of atoms that has been been substituted for a hydrogen atom in an organic compound(aka “a branch”)
1) Alkyl groups = R group
2) Halo groups = X group
F/Cl/Br/I (fluoro/chloro/bromo/iodo)
3) NO2 nitro
What are the steps to naming organic compounds?
Identify and name the parent the (longest chain of carbon atoms)
Identify the functional group and add its suffix to the parent chain name
Identify and name the types of substituents
Number the parent chain giving the functional group the lowest number and add location of the functional group to the parent chain
Number the substituents where they are bonded on the parent chain
Write the name as a single word.
What is naming the alkanes/parent chains?
Remember alkanes is only single bonds
Methane: CH4, 1 carbon
Ethane: C2H6, 2 carbons
Propane: C3H8, 3 carbons
Butane: C4H10, 4 carbons
Pentane: C5H12, 5 carbons
Hexane: C6H14, 6 carbons
Heptane: C7H16, 7 carbons
Octane: C8H18, 8 carbons
Nonane: C9H20, 9 carbons
Decane: C10H22, 10 carbons
What is an alkyl group?
An alkane missing an H usually represented by an R.
We name them like a parent alkane, but change the ending(-ane) to -yl (alkyl).
What is methyl formula and substituent look like?
-CH3

What is the ethyl formula and substituent look like?
- CH2CH3

What is the propyl formula and substituent look like?
-CH2CH2CH3
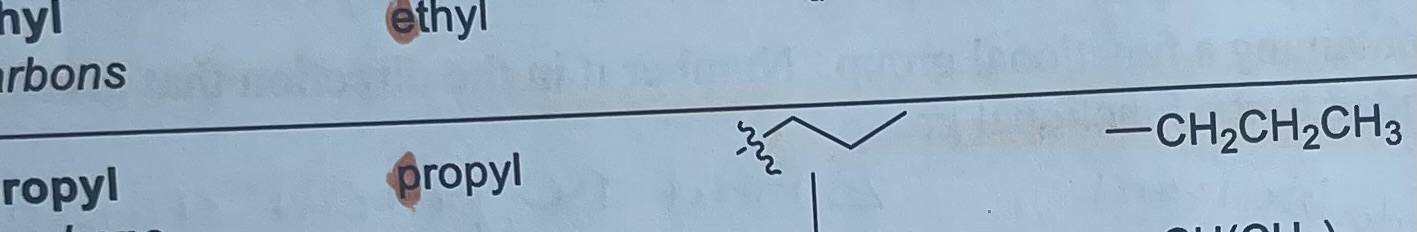
What is the formula and substituent look like for isopropyl?
-CH(CH3)2

What is the formula and substituent for butyl?
-CH2CH2CH2CH3

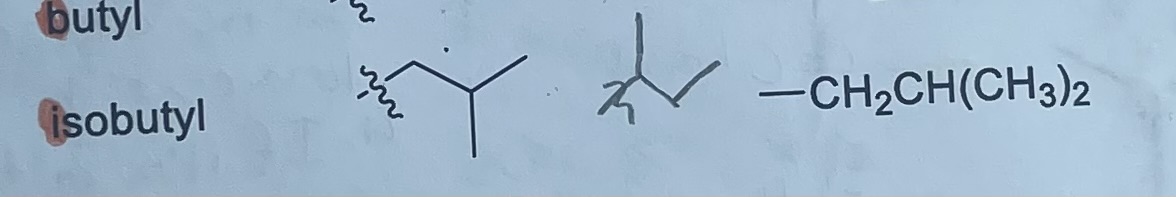
What is the formula and substituent for isobutyl?
-CH2CH(CH3)2
What is the formula and substituent of sec-butyl?
-CH(CH3)CH2CH3
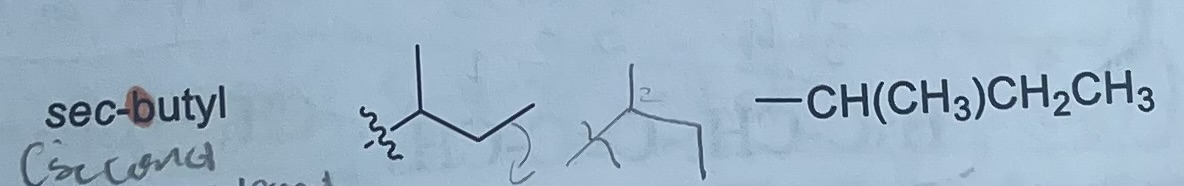
What is the formula and substituent of tert-butyl?
-C(CH3)3
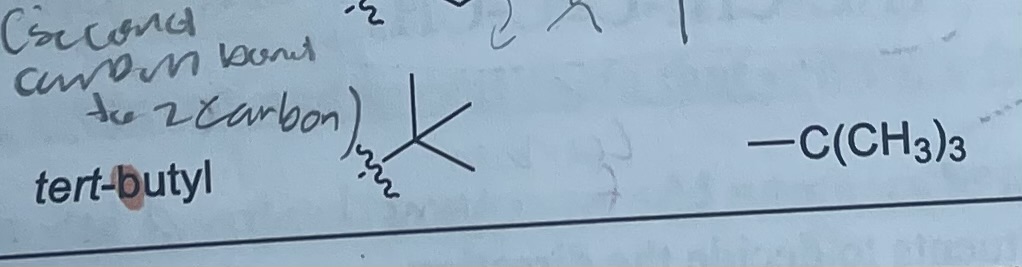
What is the formula and substituent of pentyl?
-CH2CH2CH2CH2CH3
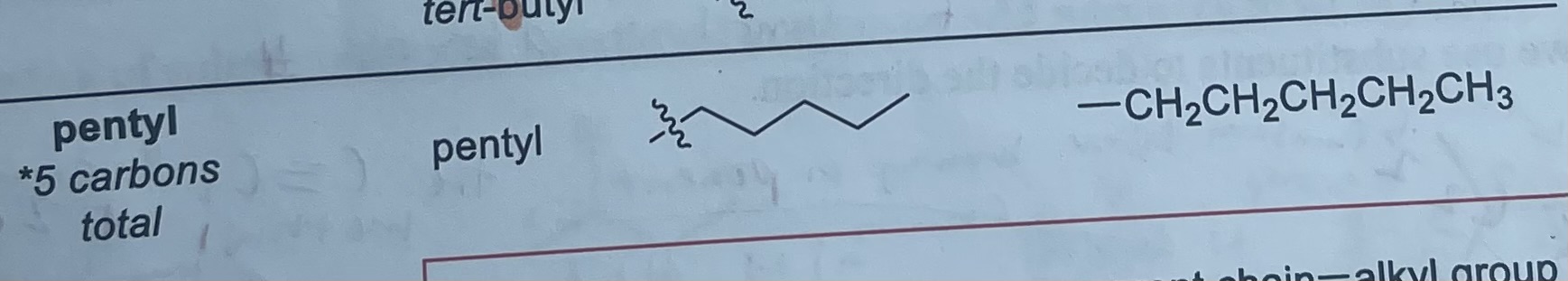
What are the other substituents not containing carbon called and what are they?
Halo groups = X group
F/Cl/Br/I (fluoro/chloro/bromo/iodo)
NO2 nitro
What do you do when 2 or more identical substituents are present?
Use the prefix(di(2), tri(3), tetra(4)
NOT USED FOR ALPHABETICAL ORDER
What are the steps in numbering the parent chain giving the function group the lowest number?
Identify the first carbon containing a functional group. Number it in the direction that gives it the lowest number to the carbons bonded to the functional group.
Find the parent chain
a. The longest chain that contains the functional group
In the direction that gives it the lowest number to the carbons
a. If it’s a tie number by which substituent side comes first.
How do we number parent chains of alkanes?
Has no functional groups
Number the chain so the carbon atoms with the most substituent closest to the end gets the lowest number.
If the number of the substituents are the same us alphabetical order gets priority.
What are the rules for naming organic compounds? And what do we use hyphens and commas for?
Write the name as a single word:
The substituents are written before the name of the parent chain
If two or more substituite groups are present, write them in alphabetical order
bromo
butyl
sec-butyl
tert-butyl
chloro
ethyl
fluoro
iodo
isobutyl
isopropyl
methyl
nitro
pentyl
propyl
We use hyphen to separate locations (numbers) from names, and use commas to separate numbers.
What are isomers(structural/constitutional isomers)?
Have the same chemical formula but are attached differently
Isomers are different molecules but have different properties
Constitutional isomers: have a different connection of atoms, but same formula.
Stereoisomers: are same connection, different shape(cis and trans molecules).
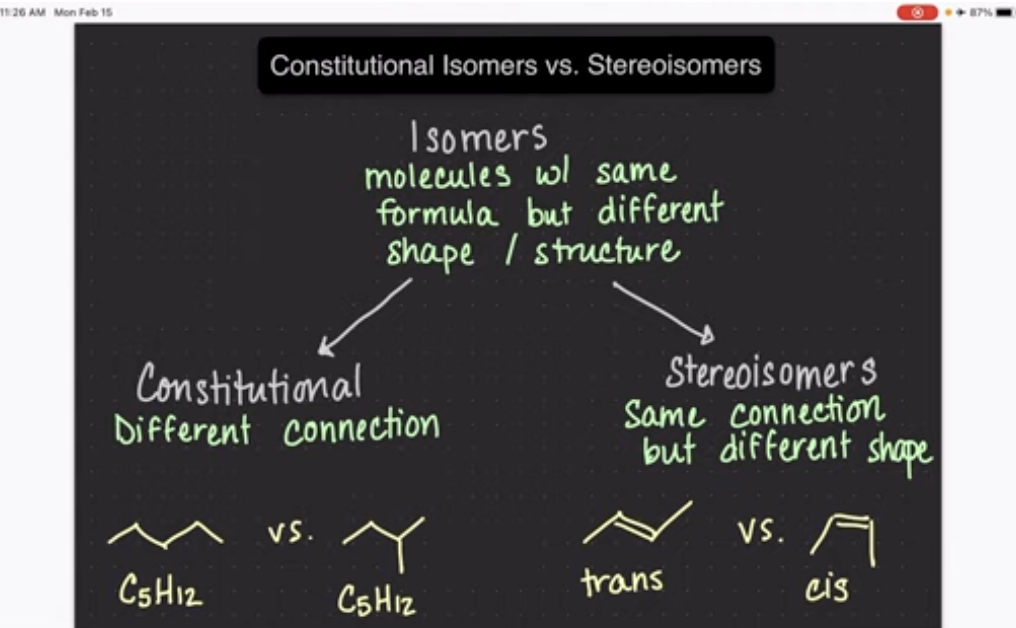
What are functional groups?
Functional groups are an atom or group of atoms in a molecule that defines the chemical properties of the molecule, example: alcohol (-OH).
What is the meaning of R?
“R” usually represents the carbons that’s not part of the functional group (specifically hydrocarbons).
What is an alpha carbon?
An alpha carbon is a carbon directly bonded to the functional group
What are the polar functional groups/draw them out(alkoxy, hydroxyl, carbonyl, carboxyl, acyl, amino, and thiol)?
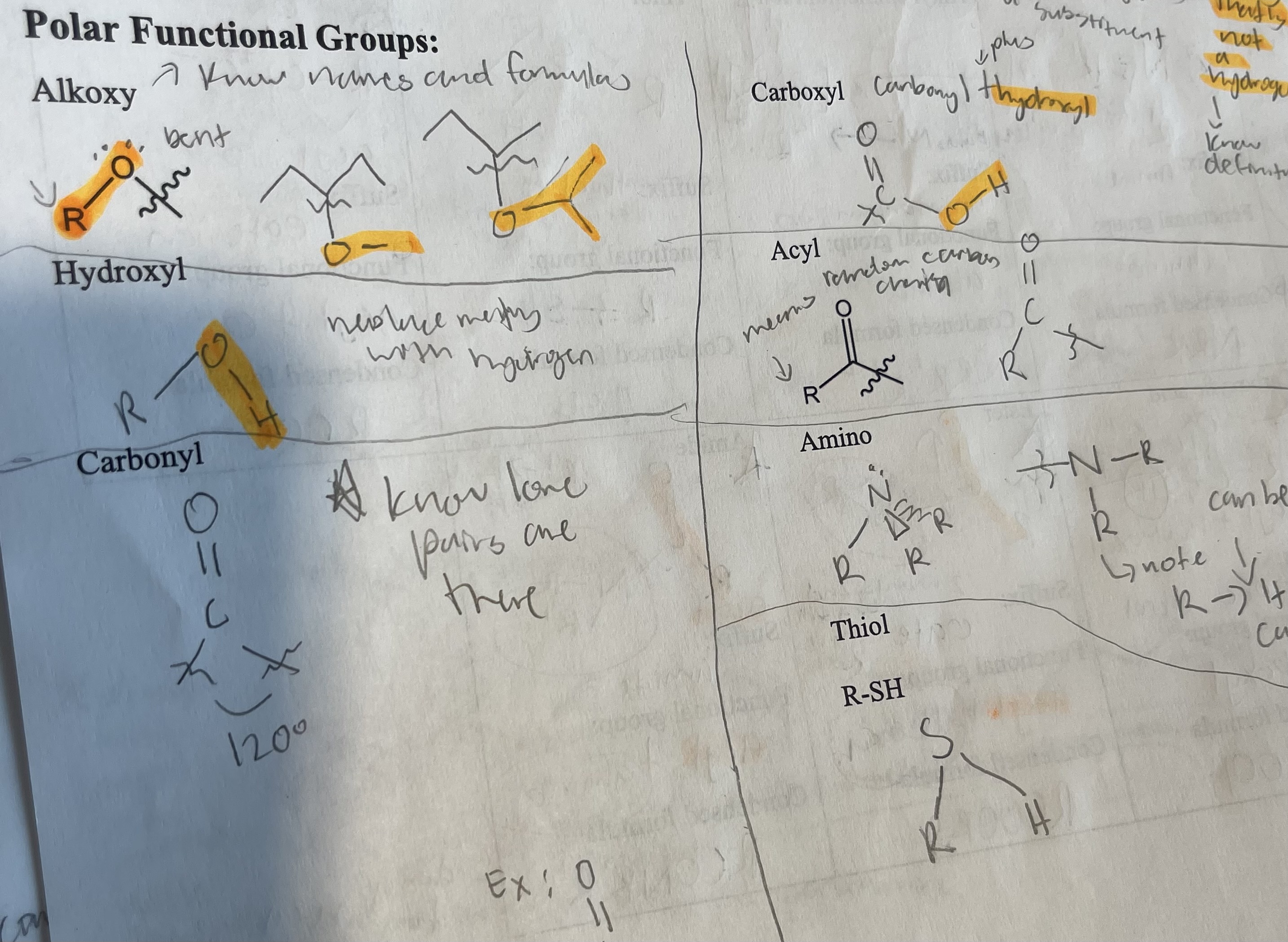
What are the steps of naming compounds with functional groups?
Identify the functional group
Label the alpha carbon
Identify and number the parent chain (longest carbon chain) giving the alpha carbon the lowest number possible
Name the parent chain
Change the suffix of the parent chain
Label in the name where the alpha carbon (location of the functional group) if there is more than one location where it could be
Name the side groups.
When do you NOT name the location of the functional groups?
When naming smaller compounds or ring structures exclude the position of the alpha carbon (location of the functional group)
Similar to alkenes and alkynes majority of the polar organic compounds do not name the location of the functional group (alpha carbon) for 2 or less carbons on the parent chain
WHAT ARE THE EXCEPTIONS TO THE 2 OR LESS RULE?
Ketones:
Since the alpha carbon cannot be on the end of the chain; ketones with 4 or less carbon do not name the location of the alpha carbon.
WHAT ARE THE CERTAIN FUNCTIONAL GROUPS YOU NEVER NAME THE LOCATION?
Aldehydes, carboxylic acids, carboxylate ions, esters, amides, and acyl.
How do we name compounds containing an acyl group: Ester and amides?
The acyl group will always be the parent chain
How do we name esters?
Identify the Acyl and alkoxy portion
The 2nd word comes from changing the name the acyl portion as the parent chain using the suffix
The 1st word is the alkoxy portion named as an alkyl group
How do we name compounds with alkyl groups attached to nitrogen(like amines, ammonium ion, and amides)?
We use “N” as the location in the name instead of a carbon number as their locant number (where alkyl is the name of the alkyl substituent).
How do we classify alcohols and alkyl halides?
Alcohols are classified by the number of alkyl groups directly attached to the hydroxyl or halogen
1 alkyl group on the alpha-carbon = primary (1°)
2 alkyl groups on the alpha-carbon = secondary (2°)
3 alkyl groups on the alpha-carbon = tertiary (3°)
How do we classify amines and amides?
-