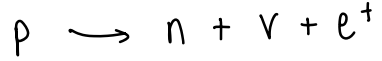f-block elements
1/62
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
What is the abundance of the lanthanides and actinides?
They are the rare earths as they are difficult to extract and purify.
La, Ce and Nd are more common than Pb (lead).
Nd is more common than Au.
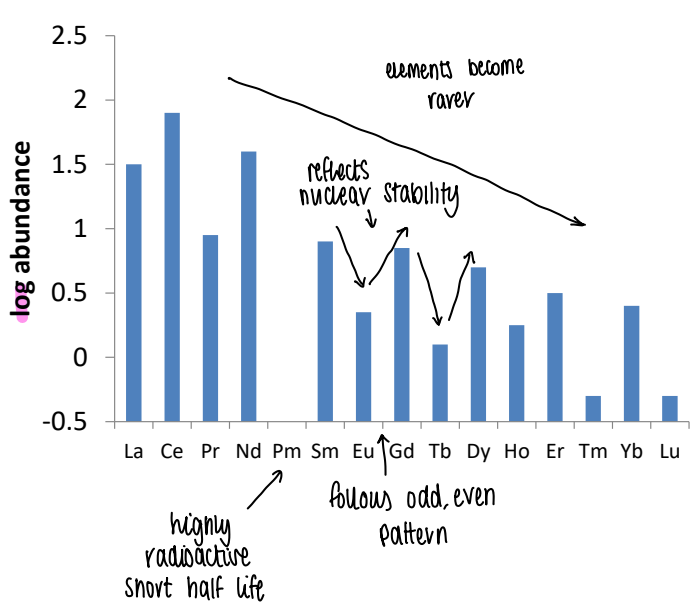
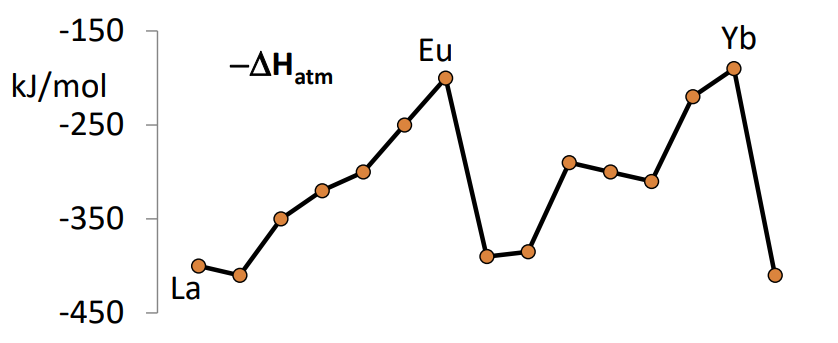
What does the relative abundances of the lanthanides show?
The high, low pattern reflects the alternating nuclear stability.
Pm has a very low abundance due to the very short half life.
Generally they get rarer across the period.
What are the abundances of the actinides/where are they found?
Th is found in the earths crust.
Pa is found only as radioactive decay from U samples.
U is found mostly in minerals, pitchblende and uranitite.
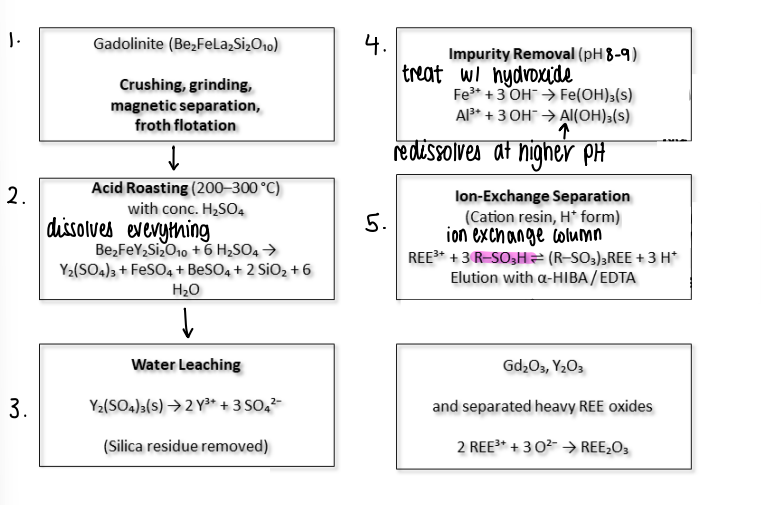
How are lanthanides extracted?
Crushing/grinding of minerals.
Acid roasting to dissolve everything.
Water leaching to remove insoluble silica.
Impurity removal, hydroxide removed insoluble precipitates at pH 8-9.
Ion exchange separation separates different lanthanides.
Heavy rare earth elements oxides are also separated.
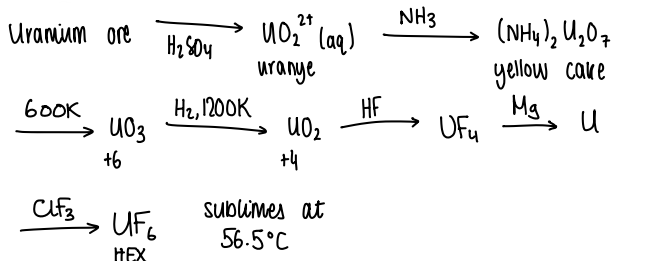
How is uranium extracted?
Extracted on an industrial scale, the isotope 235U is desired, not 238U.
HEX can be separated out due to the difference in rate of diffusion of the isotope.
What are the valence orbitals of lanthanides and actinides?
They have partially filled f orbitals (lanthanides: 4f, actinides: 5f).
F orbitals are similar to d orbitals but ‘lumpier’ due to the higher angular nodality.
Why are f orbitals contracted?
They are shielded by filled orbitals beneath them, but this shielding is limited. This causes the f orbitals to be quickly stabilised by charge on the nucleus causing contraction.
What is the relativistic effect?
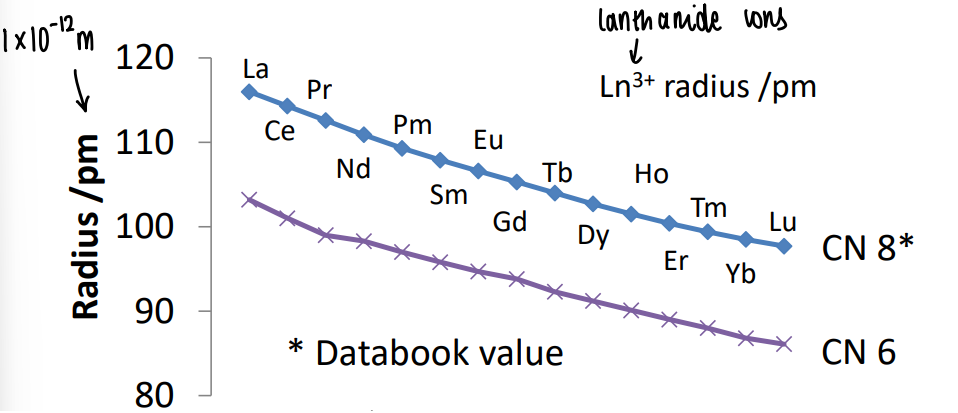
As we progress left to right across the period, the size of the Ln3+ ions get smaller.
It was thought this was due to the increase in effective nuclear charge of the metal, however it was suggested 30% of contraction is due to relativistic effects.
As the electron approaches the nucleus, it speeds up as the nucleus is so strongly positively charged. These speeds are enormous (0.6c), as you approach the speed of light mass increases.
More electron density near the nucleus results in contraction.
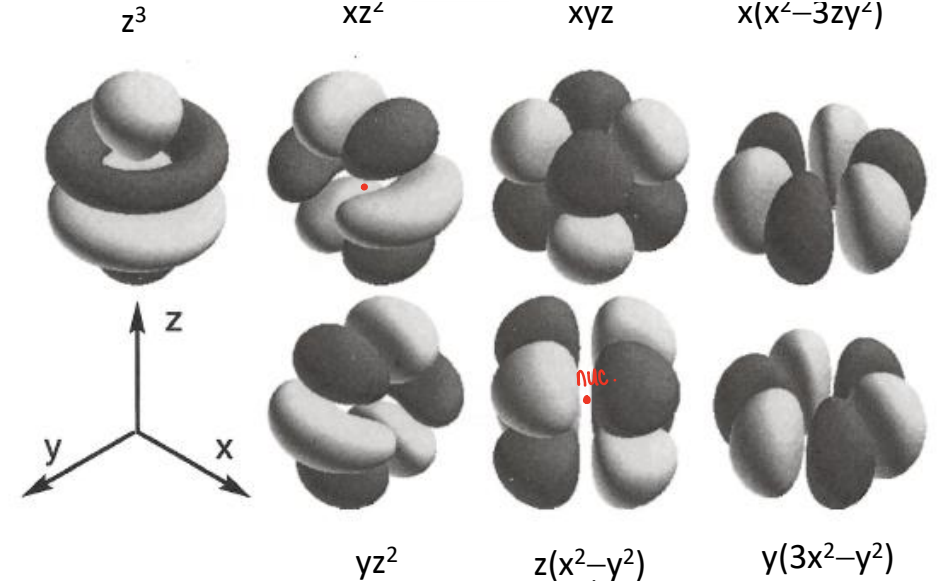
What are the properties of f orbitals?
They are like spherical ions, they are similar to d-orbitals but lumpier due to higher angular nodality. They have u-symmetry (not centrosymmetric), whereas d-orbitals are g.
This means that they have high coordination numbers and they behave like spherical atoms when bonding.
The wave equation gives solutions containing real and complex parts, the real parts are visualised in a general set and cubic set.
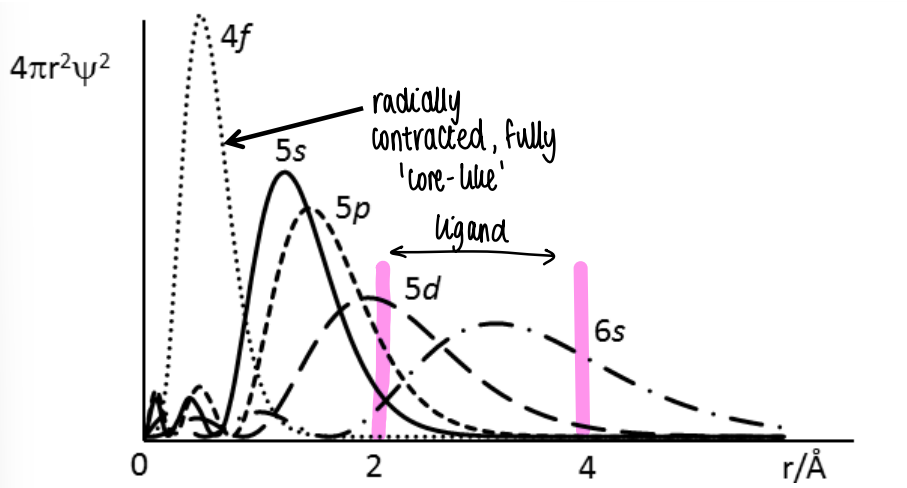
What does the radial distribution of the f orbitals show?
The 4f orbital has no radial node, so is core like and shrunk towards the nucleus.
The 5f is less core like and contains a node.
The typical L-M bond length is 2.5 A, therefore the overlap of the 4f orbitals with those of ligands is almost zero, so there is no appreciable covalent bonding in lanthanides, it is instead ionic.
There is a little covalent bonding character in actinides.
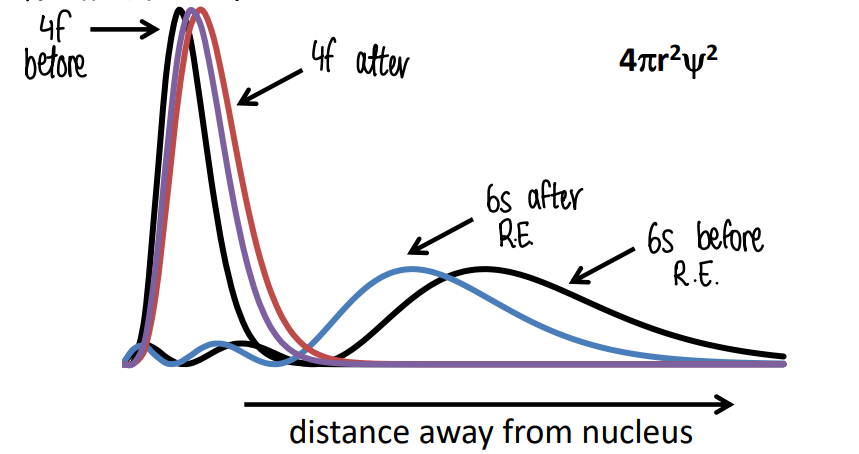
What is the effect of the relativistic effect on radial distribution of orbitals?
s orbital electrons are able to penetrate the nucleus so will get much faster due to the relativistic effect. This causes the 6s electrons to experience a large direct relativistic contraction.
f orbitals cannot penetrate the nucleus, therefore experience a relativistic expansion due to the increased shielding caused by the s orbital that penetrates the nucleus. 5f electrons experience this more significantly, causing the 5f orbitals to become less core-like and so participate in covalent overlap with ligands. The 4f remains core-like.
What is the trend in ionic radius of M3+?
The actinides are bigger than the lanthanides, therefore bond lengths from An to L will be long (2.5-3.0 A).
There is a contraction in both Ln and Ans due to the relativistic effect and increasing zeff.
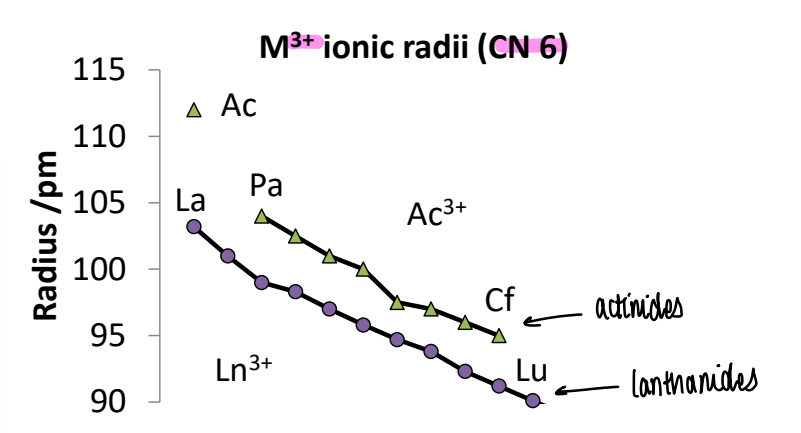
Why are 4d and 5d elements difficult to separate?
The 5d elements are smaller than expected as they experience no contraction like 5f elements. This means 4d and 5d elements have similar sizes and so have similar chemistry.
Ti (3d): 140pm, Zr (4d): 155pm, Hf (5d) 155pm
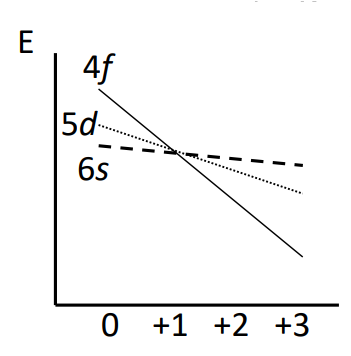
What does the Afbau principle tell us about ionisation?
Tells us that the 1st electron removed on ionisation should be the f electrons as they sit at a higher energy. However, this would result in 6s and 5d valence electrons, which is not true.
How do lanthanides form ions with a 4f valence electrons?
Upon oxidation, the 4f (and 5f) levels drop in energy significantly. By the time we reach a 3+ oxidation state, they become the lowest energy levels. This means for any Ln3+ ion the valence electrons are all in the 4f levels (this process is limited at +3 O.S.).
What is the most common oxidation state of lanthanide elements?
+3 for majority of lanthanides.
Eu and Yb are unusual as they can maintain longer, weaker bonds and exhibit stability in the +2 O.S
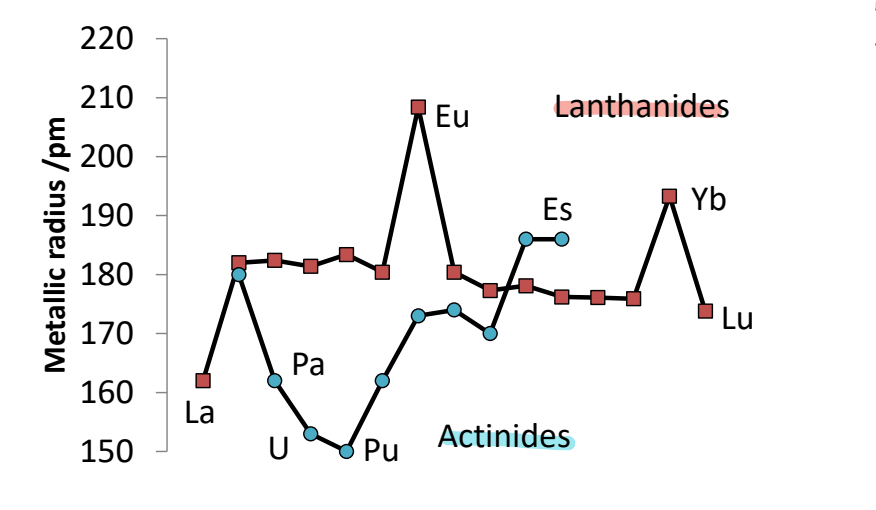
What is the trend in metallic radii of the lanthanides and actinides?
For the lanthanides the metallic radius is quite constant.
Eu and Yb have a larger radius, showing longer weaker bonding occurs.
Actinides have no real trend, reflecting their complexity.
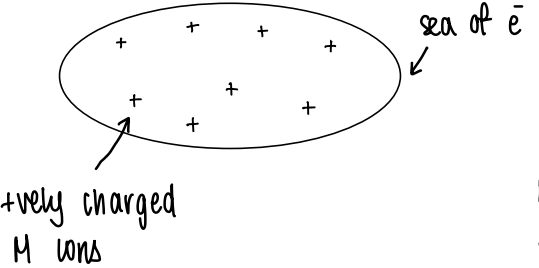
How does bonding occur in metallic solids?
There is an attraction between a sea of electrons and the metal ions, holding the metal solid together.
If the charge on the ion is constant, e.g. 3+, the bonding strength is constant. This causes the mostly constant radii of the lanthanides as they share a common O.S.
Why is Eu2+ stable but other Ln2+ ions are not?
The ionisation of Ln2+ to Ln3+ is mostly energetically uphill, however it is particularly endothermic for Eu and Yb. This shows the 2+ O.S is chemically stable.
The Eu2+ ion has a f7 and Yb has f14, these are half-filled and filled sublevels, so are particularly stable.
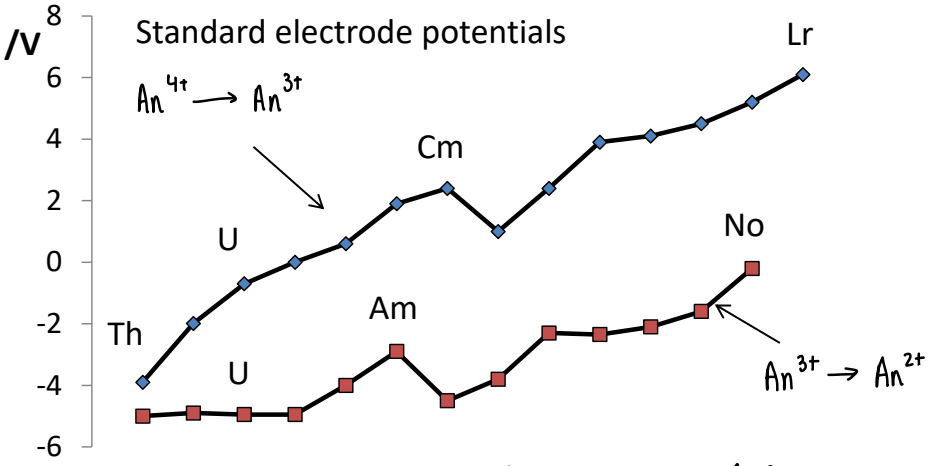
What are the standard electrode potentials of the actinides?
The electrode potentials show that actinides form a variety of O.S, this is due to the radial node in 5f orbitals. They are not core-like.
What are the oxidation states of the actinides?
They exist in a variety of oxidation states, with some reaching +7.
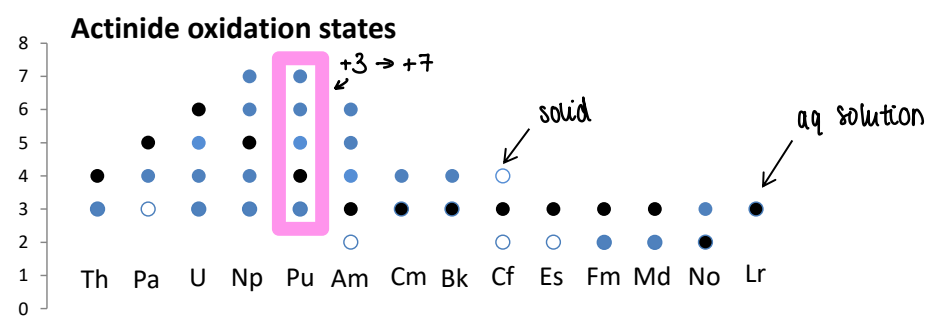
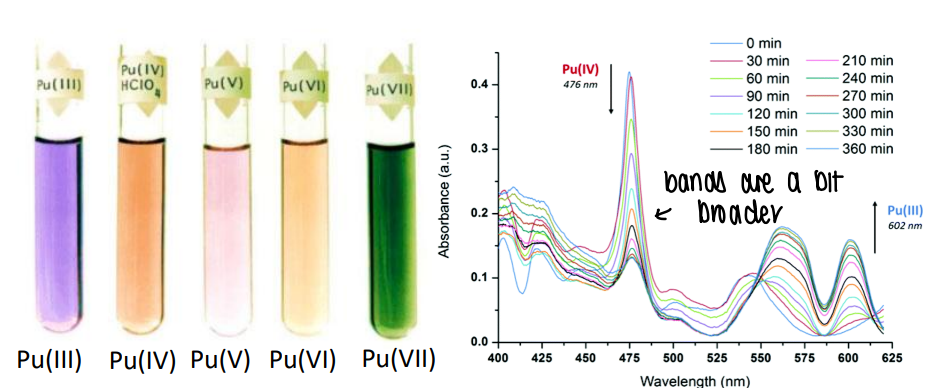
How is Pu in radioactive waste oxidised?
Pu3+(OH)3 is insoluble and stored in radioactive waste. However, when oxidised it forms [PuO4]-, which contains Pu 7+ and is soluble.
How are the energy levels of the f electrons split?
The energy levels of the valence f electrons are split due to different interactions.
A large splitting is caused by electron-electron repulsion and effects of the ligand field.
A smaller splitting is caused by spin-orbit coupling
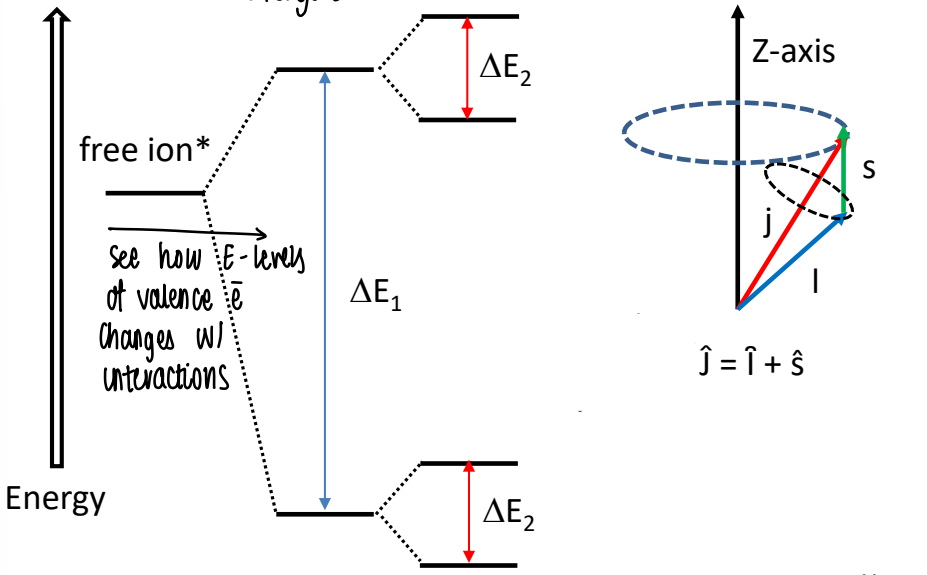
How does the splitting of f valence electrons compare to d valence electrons?
TMs with d orbital valence electrons tend to have splitting mostly determined by the ligands and electron-electron repulsion.
Lanthanides (which behave like gas phase ions) and actinides are mostly determined by electron-electron repulsion and spin-orbit coupling.
Why do lanthanides act like gas phase ions?
Molecules containing lanthanides behave as if the lanthanide is barely bonded at all, therefore similar to a gas phase ion.
How does spin-orbit coupling affect the energies of the valence electrons?
Each electron in the f orbital has its own spin (S) and angular momentum (L), these can all individually couple together
How does Russel and Saunders simplify J-J coupling?
Can simply say spin-orbit coupling becomes J=S+L
S = S1+S2…
L = L1 +L2
How do you find the term symbol?
Where J takes a series of values.
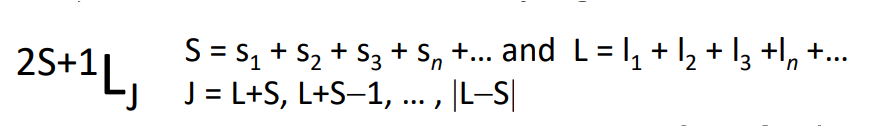
What are Hund’s rules?
The ground state is the one with maximum spin multiplicity (largest S).
When we have different states with the same S, then the lowest of these states is that with the largest L.
When we have different values of J for greater than half filled shells, then the state with the max value of J is the ground state.
How do you determine the ground state term?
Fill the orbitals with electrons, maximising S and L values.
The value of L determines the term symbol.
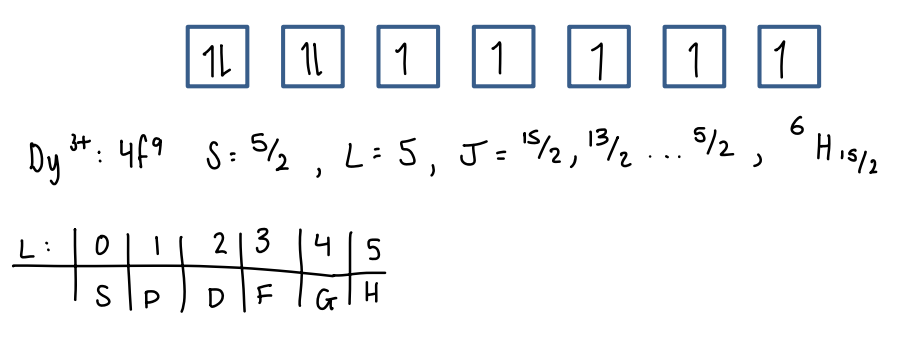
How do you use the ground state term symbol to find the magnetic properties?
The term symbol can be used to calculate the expecting magnetic moment, assuming the electrons behave as if they are in a gas-phase ion.
The Lande formula is used.
These calculated values are very close to the experimental values, therefore showing the electrons do behave similar to gas-phase ions.
Some deviation around Eu due to the unpure ground state (low lying other states interfere).
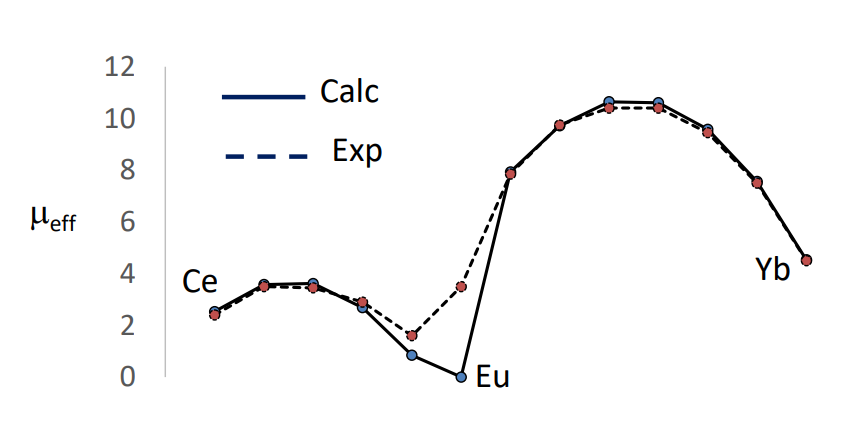
What are the magnetic properties of actinides?
The Russel-Saunders scheme starts to breakdown, J-J coupling is the best way to describe electron-electron repulsion.
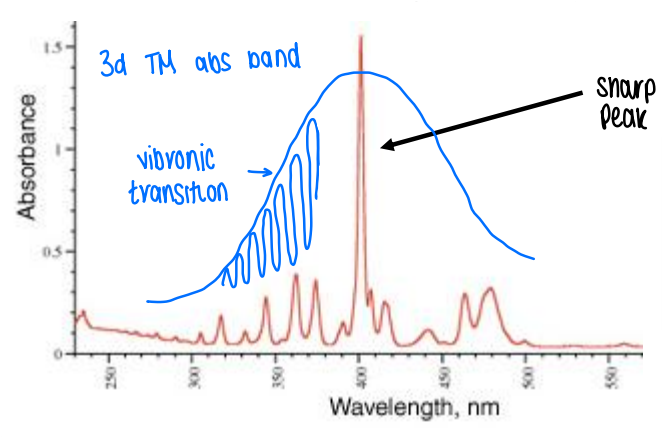
What does the electronic absorption spectra of 4f-orbitals show?
As the energy of the electrons in 4f orbitals are well defined by the Russel-Saunders scheme, any transitions between 4f energy states will also be well defined.
f-f transitions are not broadened by interactions with vibrating ligands, therefore a sharp peak is seen in the spectrum.
Why are lanthanides not brilliantly coloured as expected?
Due to the selection rules, the intensity of the light absorbed is very low, so lanthanides are essentially colourless.
Spin selection rule- cannot change spin between states, there are many Russel-Saunders states of the same spin multiplicity so this rule is followed.
Symmetry selection rule- for an intense transition, it must be g → u or u → g, all f orbitals are u symmetry so f-f transitions will be weak.
What does an actinide electronic absorption spectra look like?
As the 5f orbitals are more spatially extended, they can interact with ligands and cause some breakdown of the symmetry selection rule.
Actinides will have broader and more intense absorption than lanthanides, becoming more similar to 3d TMs.
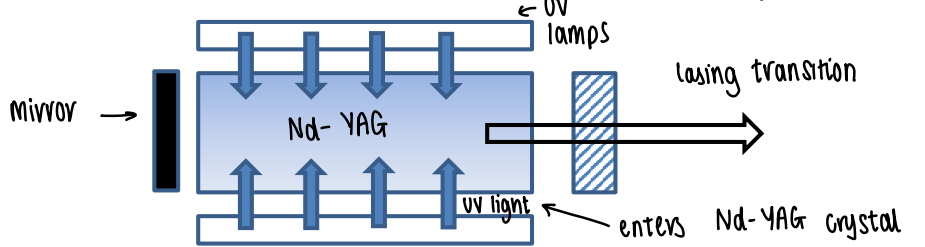
How are lanthanides used in lasers?
Nd can be used in a Nd YAG solid state laser.
UV light excites the electrons to an allowed excited state (not f-f, so fully allowed).
Relaxation occurs to an excited f state via isc.
Build up of the excited state occurs (population inversion).
An intense transition occurs of one f state to another with a fixed energy difference (almost perfectly monochromatic).
Relaxation to the ground state occurs.
How are lanthanides used in magnets?
Due to the large number of unpaired electrons in lanthanides, we can get some very high static magnetic fields.
E.g. Nd2Fe14B.
How do lanthanide complexes exhibit luminescence?
It is possible to create lanthanide complexes where there is an intense absorption (usually based on the ligand) and the excitation of this can be transferred via isc to the lanthanide excited state.
A MLCT occurs, usually with UV light (fully allowed, intense).
Isc occurs.
Excitation goes to an f-state on the metal, the efficiency of this transfer depends on how close the states A and B are.
Emission occurs.
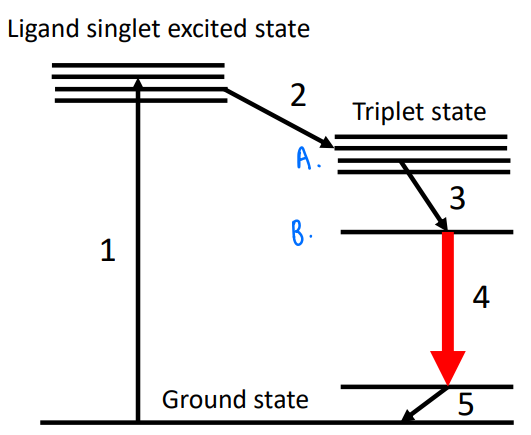
How can lanthanide complexes be used as sensors?
If we can tune the energy difference between the states A and B, then we can use this as a sensor.
If the f-f transition intensity changes due to binding of a substrate, this adjusts the energy of the A state and so the emission changes.
How are lanthanides used in screen phosphors?
Due to the sharp transitions of lanthanides, this can be used in displays to emit certain colours.
Red comes from Eu3+ embedded in Y2O2S
Green from Tb3+ in La2O2S
Different Ln ions can occupy or replace other Ln ions in structures.
How are lanthanides used in MRI contrast agents?
As lanthanides have high spin magnetism, we can use complexes of lanthanides to relax spins in other molecules.
Gd3+ can rapidly relax the spin of nearby nuclei in water due to the magnetic field from its spin centre.
Gd3+ has 7 unpaired electrons and so is very magnetic.
Gd DPTA used to image stroke effects.
How many and what kind of ligands can lanthanides bind?
Due to their core-like nature, the M-L interaction is mostly ionic. This means the ligand must be charged.
Lanthanides bind well with oxygen based ligands.
The large size of lanthanides means they can have unusually large coordination numbers.
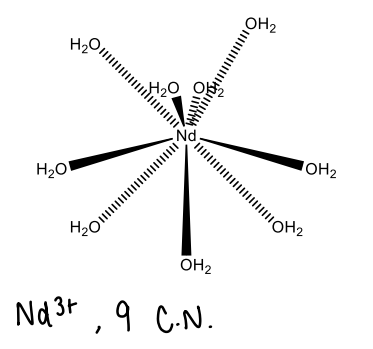
Why do lanthanides accept ligands with small bite angles?
Because they have little directional symmetry.
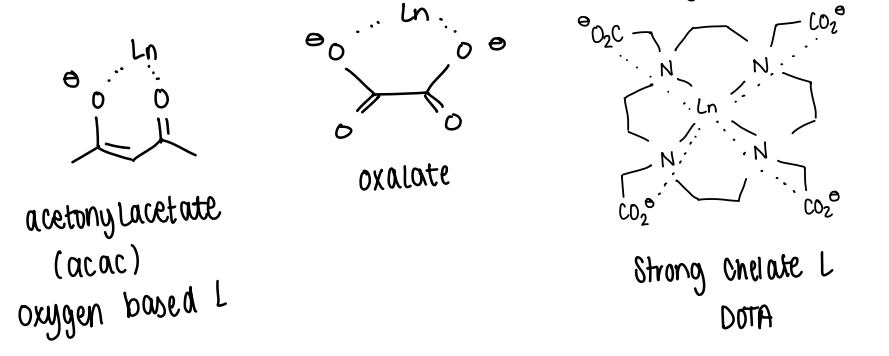
How is the chelate effect used in lanthanide complexes?
As the lanthanide ionic interaction with ligands tends to be weak, the complexes can be thermodynamically unstable.
The chelate effect is used to coordinate strongly to the Ln3+ ion.
The displacement of multiple smaller ligands by a larger chelating ligand increases the K value by many magnitudes.
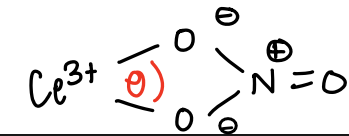
How do ligands much use of the oxophilic nature of lanthanides?
The oxygen lone pair can chelate to the lanthanide/actinide.
E.g. TBP, used to extract uranium in nuclear reprocessing.
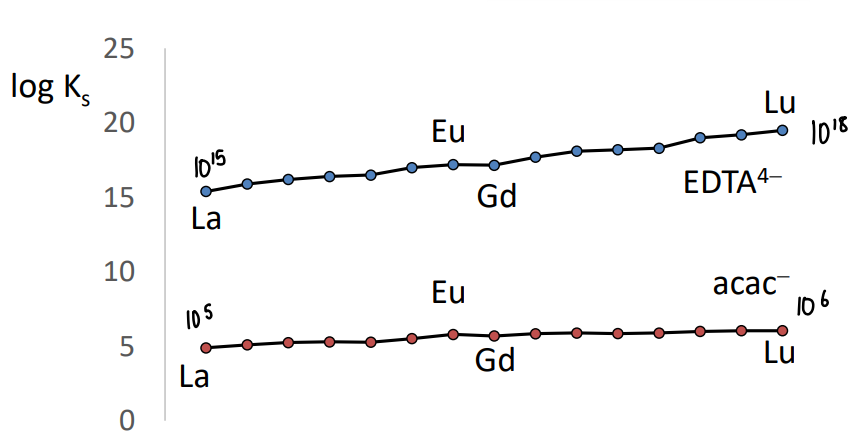
What is the trend in stability constants of lanthanide complexes?
Complexes of Ln2+ ions get slightly more stable towards the right hand side of the series due to the increasing zeff.
Only a small difference in relative stability.
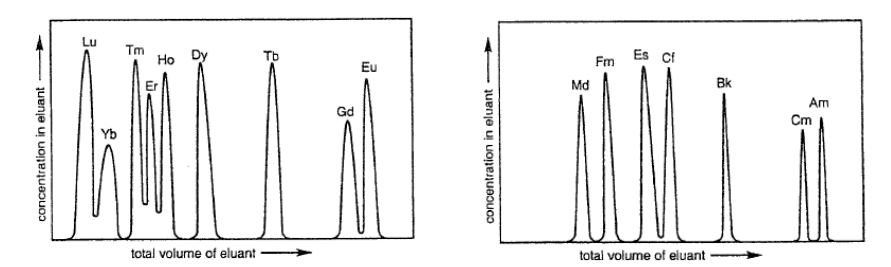
How are lanthanide complexes separated?
Using the chelate effect, we can use the small difference in stability as a basis for separation.
Using an essentially immobilised ligand on a resin, where different lanthanides stick to the resin to different extents.
What is ion-exchange chromatography?
We can separate lanthanides by using a ligand immobilised on a resin.
An aqueous solution is thrown over the resin to selectively separate the lanthanides.
How capable are lanthanides as catalysts?
Lanthanide complexes are known to act as catalysts but mostly redox-inactive Lewis acids. Their 3+ oxidation state gives them strong Lewis acidity.
The LA polarises the substrate and makes them more reactive.
Why do lanthanide catalysts have high rates of reaction?
Lanthanide catalysts benefit from high ligand exchange rates, giving a high rate of reaction.
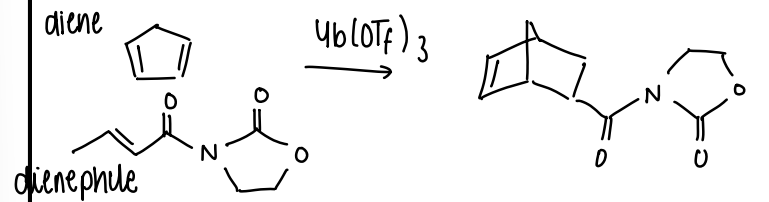
How does Yb(OTF)3 catalyse reactions?
It is able to catalyse the formation of one stereoisomer.
How does SmI2 act as a catalyst?
It is a redox active catalyst as Sm is in the +2 oxidation state, which is unusual. The Sm is expected to donate an electron to become Sm 3+.
Used in a pinacol coupling reaction
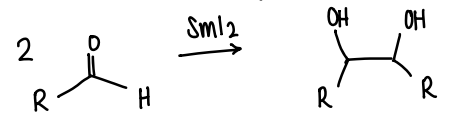
How is Ce(IV) used as an oxidant?
Ce is able to form stable oxidation states other than +3, therefore is able to oxidise other reagents and so gains an electron itself.
How are lanthanide organometallic complexes formed?
Although we expect lanthanides to form larger ionic bonds with ligands, organometallic complexes with covalent bonds can be formed.
This forms a lanthanide complex with a +2 O.S.
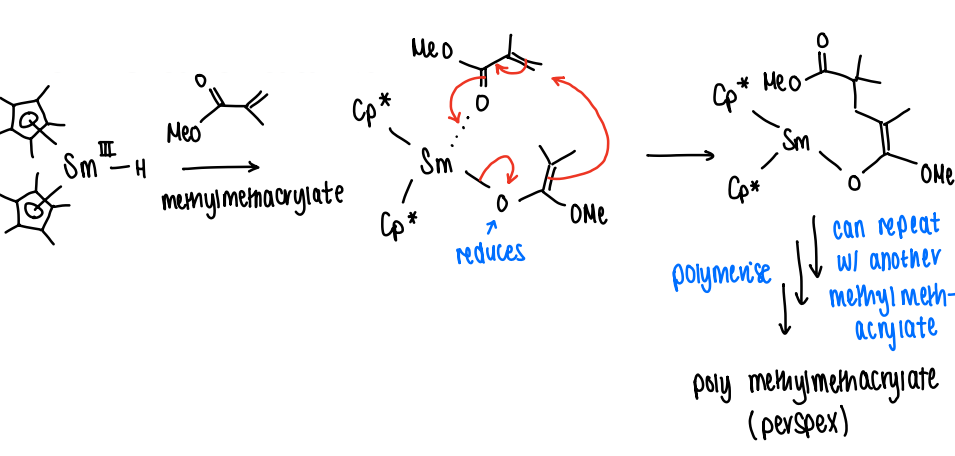
How is Sm(III) used as a polymerisation catalyst?
Sm 3+ acts as a Lewis acid, reducing methylmethacrylate and polymerising it into perspex.
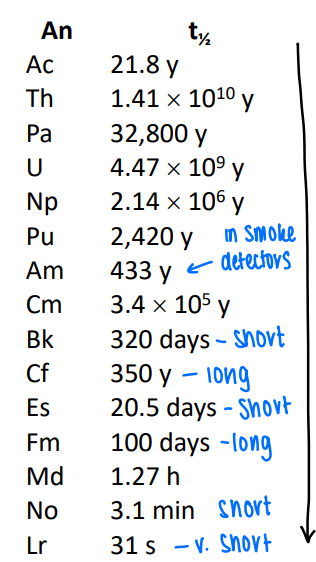
What is the trend in actinide nuclear decay?
As we get heavier the half lives get shorter.
An up down pattern of stability can be seen, this is due to the stable number of nuclei.
What is the liquid drop model?
A way of describing the stability of a nucleus as the mass number increases.
As the mass number increases, the binding force and strong nuclear attraction energy becomes overpowered by the repulsive p-p coulomb interaction, causing a decrease in nuclear stability.
What is the Coulomb barrier?
There is a coulombic repulsion which increases as a proton approaches the nucleus.
The height of the coulombic barrier is a function of the charge and nuclear mass. The bigger the nuclear mass (A), the smaller the barrier.
The proton is able to tunnel through the barrier.
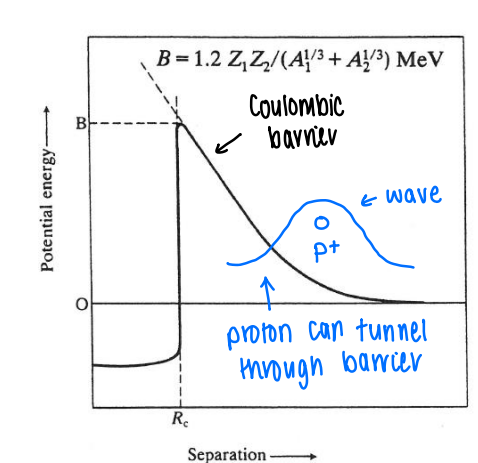
How are nuclear reactions feasible if the Coulombic barrier is so high?
The barrier is in mega-eV, which is a huge energy, therefore these reactions should not occur at room temp.
The protons are able to tunnel through the barrier. The probability of tunnelling is relating the the velocity of the emitted particle. the higher the charge, the higher the probability of tunnelling.
Why are actinides dangerous?
As they are heavy nuclei, they are alpha-emitters. This alpha radiation is very dangerous.
What is the Segre plot?
If we plot the number of protons against the number of neutrons in a nuclei, we find that as the atomic number increases the relative ratio of neutrons to protons increases for stable nuclei.
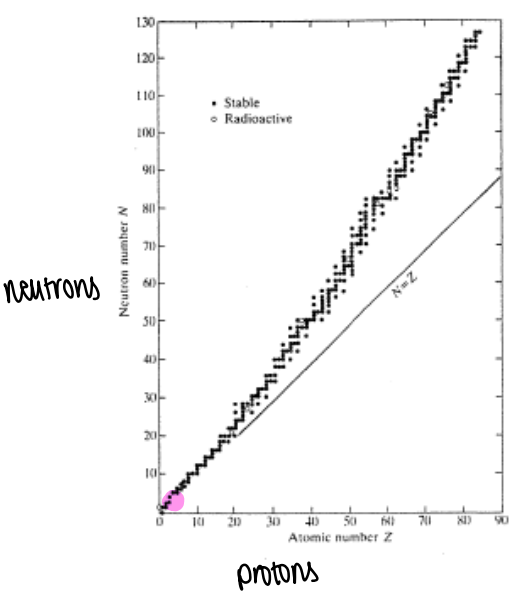
Why does the relative number of neutrons to protons start to increase?
As the Coulombic repulsion between protons becomes much greater, more neutrons are needed per proton to reduce the effects of coulombic repulsion.
What happens in unstable nuclei?
Unstable nuclei have too many protons, causing a proton to split.
The proton splits into a neutron, a neutrino and a positron (positively charged electron).