NURS 3812: Exam 3
1/86
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
87 Terms
F&Es definitions
Process of regulating the extracellular fluid volume, body fluid osmolality, and plasma concentrations of electrolytes
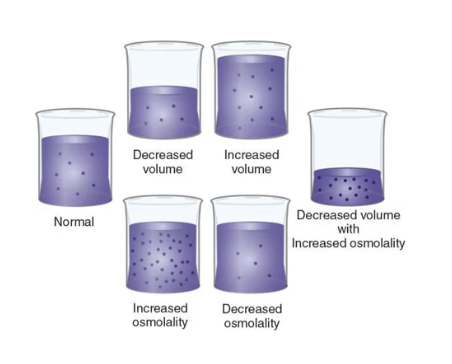
volume vs. osmolality
volume = amount of fluid , osmolality = degree of concentration
Intracellular fluid
fluid inside the cells
extracellular fluid
fluid outside the cells that includes intravascular, interstitial, and transcellular fluids
intravascular fluid
liquid part of the blood (plasma)
intersitial fluid
fluid between the cells and outside blood vessels
transcellular fluid
fluid in areas such as cerebrospinal, pleural, etc
intracellular vs. extracellular space
Most fluid is located in the intracellular space (inside cells)
The rest of fluid is in the extracellular space (interstitial, vascular space, and transcellular)
Vascular - liquid part of blood
Interstitial - fluid between cells and outside blood vessels
Transcellular - eg. cerebral spinal fluid, synovial fluid
Intake and absorption
oral, IV, NG
increased osmolality = thirst response (this is decreased in older adults)
Output
normal = urine, feces, skin/sweagt, lungs
abnormal = emesis, hemorrhage, wound drainage
regulated by kidneys - aldosterone (Na+ H20), ADH (H2O)
why do infants and children have a more vulnerable fluid balance?
unable to communicate thirst
larger ECF volume = faster fluid loss
higher rates of metabolism = use up more H20
higher percentage of body content water = need more H20 to maintain balance
higher BSA (body surface area) to volume = lose more H20 through skin (sweating)
immature kidneys = little reserve/need electrolytes
why do older adults have a more vulnerable fluid balance?
increased risk for ECV deficit and dehydration d/t:
lower percent of body weight as water, decreased thirst response, and decreased kidney function
chronic diseases and medications place them at risk for ECV imbalances
those with incontenence may restrict fluids placing them at risk for hypernatremia and ECV defecit
types of fluid imbalances
clinical dehydration
fluid volume deficit
fluid volume excess
clinical dehydration
loss of water
without loss of Na+ (high sodium)
decreased volume with increased osmolality (concentration)
extracellular volume deficit and hypernatremia (high sodium)
water shifts from inside of cell to the extracellular space = cell shrinkage
fluid volume deficit
loss of both water and electrolytes
decreased volume
fluid volume excess
too much isotonic fluid
increased volume
cues/causes of clinical dehydration
lack of water intake
gastrointestinal losses ( vomiting, diarrhea, NGT suction)
prolonged fever
excessive sweating (marathons, working outside in hot weather, tachypnea in infants, radiant warmer/phototherapy for newborns)
medications like benzodiazepines(decrease thirst sensation) and diuretics (excess urination)
poor thirst response (older adults)
unable to voice thirst (infants)
manifestations of clinical dehydration in adults
Manifestations
General: Postural hypotension, ↑HR, thready pulse, sudden weight loss, dry mucous membranes, poor skin turgor*, flat neck veins, dark yellow urine, ↓LOC (confusion, lethargy, coma), thirst, seizures with rapid change, ↑temp
Severe: restlessness, confusion, ↑HR & ↓BP, oliguria (UO<30mL/hr), cold clammy skin, seizures
Labs:
↑ Na+ >145mEq/L, ↑ serum osmolality > 295mOsm/kg, ↑hematocrit, ↑BUN (>20mg/dL) showing hemoconcentration, ↑urine specific gravity (>1.030)
manifestations of clinical dehydration in infants and children
Fewer wet diapers than usual
No tears when crying
Mucous membranes dry and sticky
Less playful, more tired, cranky
Lethargy
Very poor skin turgor
Increased respiraotry rate
Sunken fontanel
Sunken eyes with dark circles
Abnormal skin color/temperature

Isotonic IV fluid
No movement of water b/t ECF and ICF = expansion of ECF
0.9% NaCl, lactated ringer's
Hypotonic IV fluid
ECF has fewer solutes than fluid in cells = water moves from extracellular space into cells
0.45% NaCl
Hypertonic IV fluid
ECF has more solutes than within cells = water leaves cells and interstitial space into plasma
3% NaCl (cerebral edema and symptomatic hypernatremia), D5% in 0.45% NaCl (treat hypovolemia), D5 in 0.9% NaCl
A nurse caring for client who is experiencing dehydration. Which of the following findings should the nurse identify associated with this condition? Select all that apply.
Thready pulse
Dry mucous membranes
BUN 30 mg/dL
Urine output of 90 mL/hr
Blood pressure of 90/50
Thready pulse
Dry mucous membranes
BUN 30 mg/dL
Urine output of 90 mL/hr
Blood pressure of 90/50
Extracellular volume deficit/ Fluid volume deficit/ hypovolemia
decreased volume with normal osmolality
Extracellular space holds more Na+.
Output of isotonic fluid exceeds intake of sodium containing fluids.
Insufficient isotonic fluid in the extracellular.
risk factors for fluid volume deficit
blood loss
GI losses (diarrhea and vomiting)
severe burns
excessive sweating without water and salt intake
fever
medications (diuretics)
altered intake (impaired swallowing, prolonged NPO, confusion)
difference between clinical dehydration and fluid volume deficit
Clinical Dehydration = decreased fluid and too concentrated
fluid volume deficit/hypovolemia = decreased volume with normal osmolality

Cues of fluid volume deficit
Manifestations
General: thirst, dryness of mucosa, decreased skin turgor*, flat neck veins, dark urine, and decreased urine output, sudden weight loss, increased HR, thready pulse
Severe: extreme thirst, restlessness, confusion, increasing HR & worsening hypotension, oliguria (<30mL/hr), cold clammy skin
Labs
↑hematocrit (>52% males, >47% females) - unless related to bleeding then low, ↑BUN (>20mg/dL) showing hemoconcentration, ↑urine specific gravity (>1.030)

Fluid volume deficit interventions
Goal: treat underlying cause and restore fluid and electrolyte balance
Interventions:
Depending on severity
Mild: oral rehydration with electrolytes (pedialyte, sports drinks)
Peds: 1 cup (240 ml) for every 4.54 kg (10 lbs) - start with small sips 5 mL every 1-2 minutes
Moderate to severe: isotonic IV fluids: 0.9% normal saline or lactated ringers
Moderate to severe changes in vitals, symptomatic, unable to keep fluids down
Peds: IV bolus 20 mL/kg of 0.9% NS over 10-20 minutes
Treat underlying cause example:
If related to trauma/blood loss: packed red blood cells
Monitor: weight, I&Os, s/s of FVD and fluid overload with IVFs
Fall prevention
Evidence of successful rehydration: Pediatric
Moist mucous membranes
Sodium and potassium within normal limits
Voiding >1 mL/kg/h
Capillary refill of 2s or less
Skin turgor brisk (Fontanelle flat)
Fluid intake and output balanced
Vital signs within normal limits
Behavior normal (developmentally appropriate)
fluid volume excess/fluid volume overload
increased volume with normal osmolality
Too much isotonic fluid in the extracellular space
Intake of sodium-containing isotonic fluid exceeds output
Too much water and sodium
cues of fluid volume overload
Manifestations can vary based on cause:
General: sudden weight gain (1L fluid = 1kg or 2.2 lbs), edema (not a good indicator compared to weight), full neck veins, crackles in lungs, dyspnea, bounding pulse
Severe: confusion, pulmonary edema
Labs:
↓hematocrit <40% M, <36% F & ↓BUN (<10mg/dL - except not in kidney failure baseline will be high) hemodilution
risk factors for fluid volume overload
heart failure
kidney fialure
excessive Na+ containing IV fluids
cirrhosis
interventions for fluid volume overload
Goal: restore fluid balance - remove excess fluid
Interventions:
Impaired Gas Exchange -Fluid in lungs - elevate HOB, supplemental O2, possible IV diuretics (furosemide), dialysis (End stage kidney disease)
Fluid Imbalance -Daily weight: 1kg (2.2lb) in on day = 1L of fluid gained, monitor Intake and output, Edema in legs - elevate, medications, fluid and Na restrictions
Manage the cause:
Fluid and sodium restriction
Medication Regimen
Dialysis if missed sessions
Fluid moves between blood vessels and interstitial fluid by filtration; water moves between ECF and ICF by osmosis; both processes maintain fluid and electrolyte balance.
true
Electrolytes play a critical role in:
Balancing body fluids (Na+)
Cerebral function (Na+)
Regulating heart rhythm
Supporting neuromuscular function
(K+, Ca+, Mg+)
Electrolyte homeostasis involves 3 processes:
intake and absorption
distribution within the body
output and loss
electrolyte distribution in the body
Na+ high concentration in the ECF
K+ inside cels
Mg2+ inside cells and bones
Ca2+ in bones (Calcitonin moves Ca2+ into bone & PTH shift Ca+ from bone to ECF)
clients at greatest risk for electrolyte imbalances
Infants and children, older adults, clients with cognitive impairment, clients with chronic illnesses
Acute illness or trauma
Burns, hemorrhage, head injuries
Chronic illness
Renal disease, heart failure, cancer
Medications
Diuretics, laxatives
Electrolyte Imbalance Causes
Output greater than intake and absorption
Examples: prolonged anorexia, lack foods rich in electrolytes, etc.
Output less than intake and absorption
IV infusions, oliguria
Distribution altered
Shifting of electrolytes out of their normal location (K+, Mg+, Ca+ moving into ECF)
Homeostasis:
the ability of the body to maintain internal stability while adjusting to changing conditions
Acid-base balance
process of regulating the pH, bicarbonate concentration (HCO3), and partial pressure of carbon dioxide (CO2) of body fluids
pH definition
degree of acidity or alkalinity
Expected range for human blood is very narrow and regulated by homeostatic processes
pH ranges
Low pH < 7.35 = acidotic (too much acid)
High pH >7.45 = basic (alkaline) (too little acid)
Cells and Tissues cannot function optimally or at all if the pH goes too far out of range
What are some contributors towards our bodies’ Acidic content?
Carbon Dioxide (CO2)
Stomach acid
Lactic Acid
Ketoacidosis
What is the main contributor for Alkaline content?
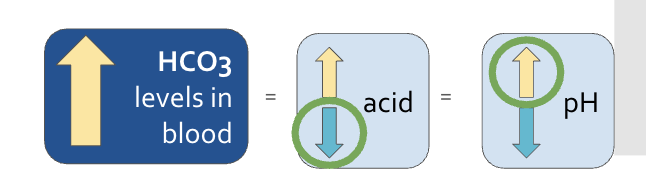
Bicarbonate (HCO3-)
What body system primarily regulates bicarbonate (HCO3)?
kidneys
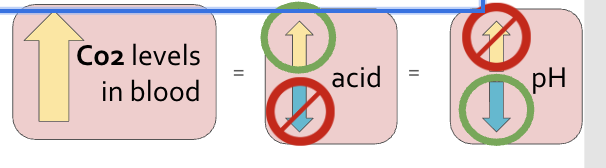
What body system regulates Carbon Dioxide (CO2)?
lungs
Hyperventilation (Increasing respiratory rate) -CO2 is breathed off
CO2 is breathed off
↑CO2 in blood = ↑ resp. rate and depth = to ↓ CO2 level
Hypoventilating (decreasing respiratory rate)
-CO2 is retained
↓CO2 = ↓ resp. rate and depth = to ↑ CO2 level
respiratory response in lungs
When the body senses an acidic or alkaline pH, this can trigger a response in the lungs to alter respiration rate
Measured by PaCO2
FAST response (minutes/hours)
METABOLIC (Renal) Response: Kidneys
When the body senses an acidic or alkaline pH, this can trigger a response in the kidneys to alter bicarbonate excretion or absorption
The kidneys filter acidic byproducts and HCO3– and either excrete them in the urine or reabsorb them back into the bloodstream
Measured by HCO3-
SLOW response (24-48 hours to completely respond)
difference between respiratory and metabolic response to pH shift
respiratory = FAST response (minutes/hours)
metabolic = SLOW response (24-48 hours to completely respond)
What are some potential causes of respiratory acidosis?
Respiratory depression (sedatives, opioid use)
Inadequate chest expansion (trauma or weakened muscles)
Airway obstruction (sleep apnea)
Reduced alveolar-capilary diffusion (pneumonia, emphysema/COPD, acute respiratory distress, pulmonary edema, PE)
hint: what would cause the lungs to not be able to remove enough CO2 from the body
what are some potential causes of metabolic acidosis?
Overproduction of hydrogen ions (lactic acidosis: shock, diabetic ketoacidosis, starvation)
Underelimination of hydrogen ions (kidney failure)
Underproduction of HCO3 (kidney failure, liver failure, pancreatitis)
Overelimination of HCO3 (diarrhea)
what would cause the body to lose HCO3? What is the most commonly known cause of metabolic acidosis?
what are some potential causes of respiratory alkalosis?
Any condition that results in hyperventilation (pain, anxiety, fever, sepsis, fever (esp. infants)), trauma
When hyperventilation occurs, the body exhales too much CO2 and RESPIRATORY ALKALOSIS can occur —> loss of CO2
Hint: what would cause hyperventilation?
what are some potential causes of metabolic alkalosis?
Decrease of acids:
Prolonged vomiting
Gastric suctioning
Excessive diuretic use
Increase in base (HCO3):
Excessive antacid use
Renal impairment
hint: what would cause the body to lose acid?
Causes of Hypocalcemia (↓Ca+)
Lack Ca+ foods, poor absorption (chronic diarrhea, lack vit D), hypoparathyroidism
Findings of Hypocalcemia (↓Ca+)
Increased neuromuscular excitability - +chvostek and trousseau, twitching, hyper reflexes, seizures, laryngospasm, cardiac dysrhythmias
Causes and Findings of Hypercalcemia (↑Ca+)
Causes:
Vit D or Ca+ overdose, thiazide diuretics, hyperparathyroidism, bone cancer
Findings:
Decreased neuromuscular excitability - constipation, muscle weakness, dec reflexes, decreased LOC, cardiac dysrthythmias, bone pain
Causes and findings of Hypomagnesemia (↓ Mg+)
imbalances occur when normal homeostasis is disrupted and compensatory mechanisms fail (impaired function of organ systems) OR compensatory mechanisms are overwhelmed (external stressors are too severe or significant)
true
Arterial Blood Gases (ABGs)
lab draw used to interpret acid-base balance in the body
acid-base status
underlying cause of imbalance
body’s ability to regulate pH
overall oxygen status
sample drawn from an artery rather than a vein
pH, PaCo2, HCO3, PaO2, O2 sats
Acidosis
= too much CO2 or not enough HCO3-
Alkalosis
= too much HCO3- or not enough CO2
ROME
Respiratory
Opposite
Metabolic
Equal
What are signs and symptoms of respiratory acidosis?
Dyspnea
Anxiety
Confusion
Fatigue, lethargy and sleepiness
Flushed skin and sweating
Tachycardia
Treatment for respiratory acidosis
treat the underlying cause
supplemental oxygen
CDB, IS ?
medications such as bronchodilators and corticosteroids
mechanical ventilation if needed
Signs and symptoms of metabolic acidosis?
long, deep breaths - Kussmaul respirations
confusion, headache
tachycardia
loss of appetite
nausea, vomiting
hyperkalemia
treatment for metabolic acidosis
treat the underlying cause
remove additional acid
IV fluids and electrolytes
sodium bicarbonate (pH < 7.2)
LOW and SLOW
Signs and symptoms of respiratory alkalosis?
hyperventilation!
lightheadedness, dizziness
confusion
chest pain
numbness in hands and feet
treatment for respiratory alkalosis
decrease respiratory rate!
talk them down
pain, fever management
rebreathing expired air
fall precautions d/t neurologic and musculoskeletal impacts
trat underlying cause
signs and symptoms of metabolic alkalosis?
hypocalcemia (occurs with metabolic alkalosis)
muscle twitching or spasms
tremors
tingling in face or lower extremities/feet
nausea and vomiting
lightheadness
headache
treatment for metabolic alkalosis
treat underlying cause
correct acid loss
correct electrolyte imbalance
correct fluid loss/imbalance
fall precautions d/t neuromuscular effects
role of RN when caring for a client with an acid-base imbalance?
assess for signs and symptoms of acid-base imbalance
consider potential underlying causes and communicate with care team
analyze lab values including ABGs
monitor vital signs, RR and effort, neuromuscular status, I&O
place the client on fall precautions if indicated
administer medications and treatments as prescribed
treatment for sodium electrolyte imbalance
monitor neuro!!
hyponatremia: oral or IV replacement - SLOW to prevent rapid fluid shifts/seizures, fluid restrictions
Hypernatremia: fluid replacement (D5W), Na+ restriction
treatment for low potassium imbalance (hypokalemia)
oral (meds or food) or IV replacement
IMPORTANT: IV must be diluted, do not exceed 40meq/L, rate 10 mEq/hr (NEVER push), continuous ECG monitoring, need adequate kidney function
Oral meds - take with food - GI distress, never crush ER
Monitor UO - need adequate renal function
treatment for high potassium imbalance (hyperkalemia)
restrict K+ rich foods
renal failure = dialysis
patiromer/sodium polystyrene sulfunate (K+ excreted through stool), IV insulin and dextrose (pushing K+ into cells) - monitor for hypogylcemia
Monitor cardiac
treatment for magnesium electrolyte imbalance
Hypomagnesemia: oral medication; IV for severe cases: monitor respiratory depression and renal function
Monitor neuromuscular and cardiac
Hypermagnesemia: stop meds with Mg, IV calcium gluconate if severe (cardiac dysrhythmias)
Monitor neuromuscular, respiratory, & cardiac
treatment for calcium electrolyte imbalance
Hypocalcemia: calcium and vit D supplements, dietary changes
Seizure precautions, monitor airway, cardiac and neuromusculare
Hypercalcemia: oral phosphate, IV saline bolus + loop diuretic
Monitor neuromuscular, cardiac
Lab value changes associated with dehydration
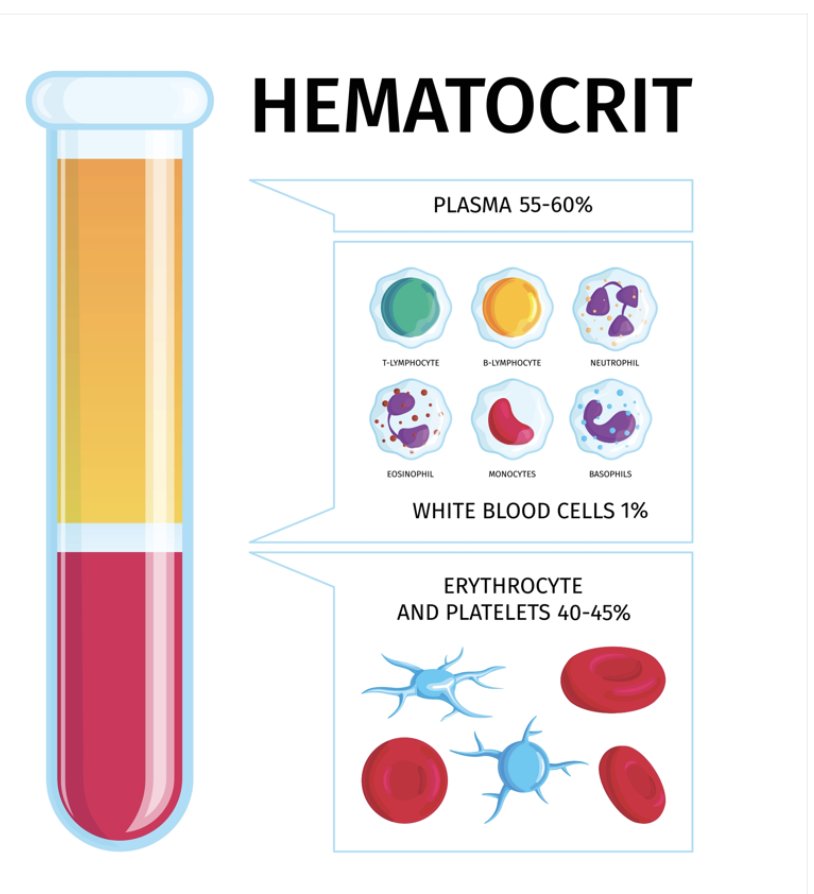
concentrated hematocrit (Hct)
plasma becomes more concentrated with red blood cells
increased serum sodium
dehydration resulting from lack of fluid intake vs. fluid loss
sodium is maintained, but fluid volume is deficient
hypernatremia can occur
hematocrit
= the proportion of red blood cells in the blood
Patient with febrile illness with diarrhea x 3 days will have these lab value changes:
Lab value changes associated with GI loss:
Hypokalemia
Fluid volume deficit
Lab value changes associated with febrile illness or infection:
Elevated WBC count
Patient with Heart failure exacerbation with fluid overload, admitted for diuresis will have these lab value changes:
Lab value changes associated with fluid overload:
Dilution of Hematocrit
Decreased serum sodium concentration
Lab value change associated with diuretics:
Loss of Potassium
Normalization of fluid balance and correction of dilutional hematocrit
Patient Postoperative day 1 status post Thyroidectomy will have these lab value changes:
Lab value changes associated with endocrine disruptions caused by thyroid or parathyroid surgery:
Hypocalcemia
Calcium is regulated by the thyroid and parathyroid
Possible blood loss during surgery
Possible dilutional Hct due to fluids received during surgery
what electrolyte does the thyroid and parathyroid regulate?
calcium
Calcium homeostasis is regulated primarily by two hormones:
Calcitonin
Parathyroid Hormone
when is each type of saline used?
3% NS - is hypertonic for clients experiencing hyponatremia
0.9%NS - is isotonic and used for hydration from vomiting diarrhea, hemorrhage, and shock
0.45% - is hypotonic and is used to treat hypernatremia and diabetic ketoacidosis.
Dextrose 10% in water is hypertonic and used to treat hypoglycemia