E.3 Radiactive decay
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
What is the mass defect of a nucleus?
The difference between the mass of the nucleus and the sum of the masses of its individual constituent nucleons.
What is the definition of nuclear binding energy?
The minimum energy required to completely separate the nucleons of a nucleus to infinity, or the energy released when a nucleus is formed from its nucleons.
How do you calculate energy released in a nuclear reaction using mass-energy equivalence?
Energy equals the change in mass (mass defect) times the speed of light squared.
What are the main characteristics of the strong nuclear force?
It is a very short-range, highly attractive force that acts between all nucleons (protons and neutrons) to overcome electrostatic repulsion.
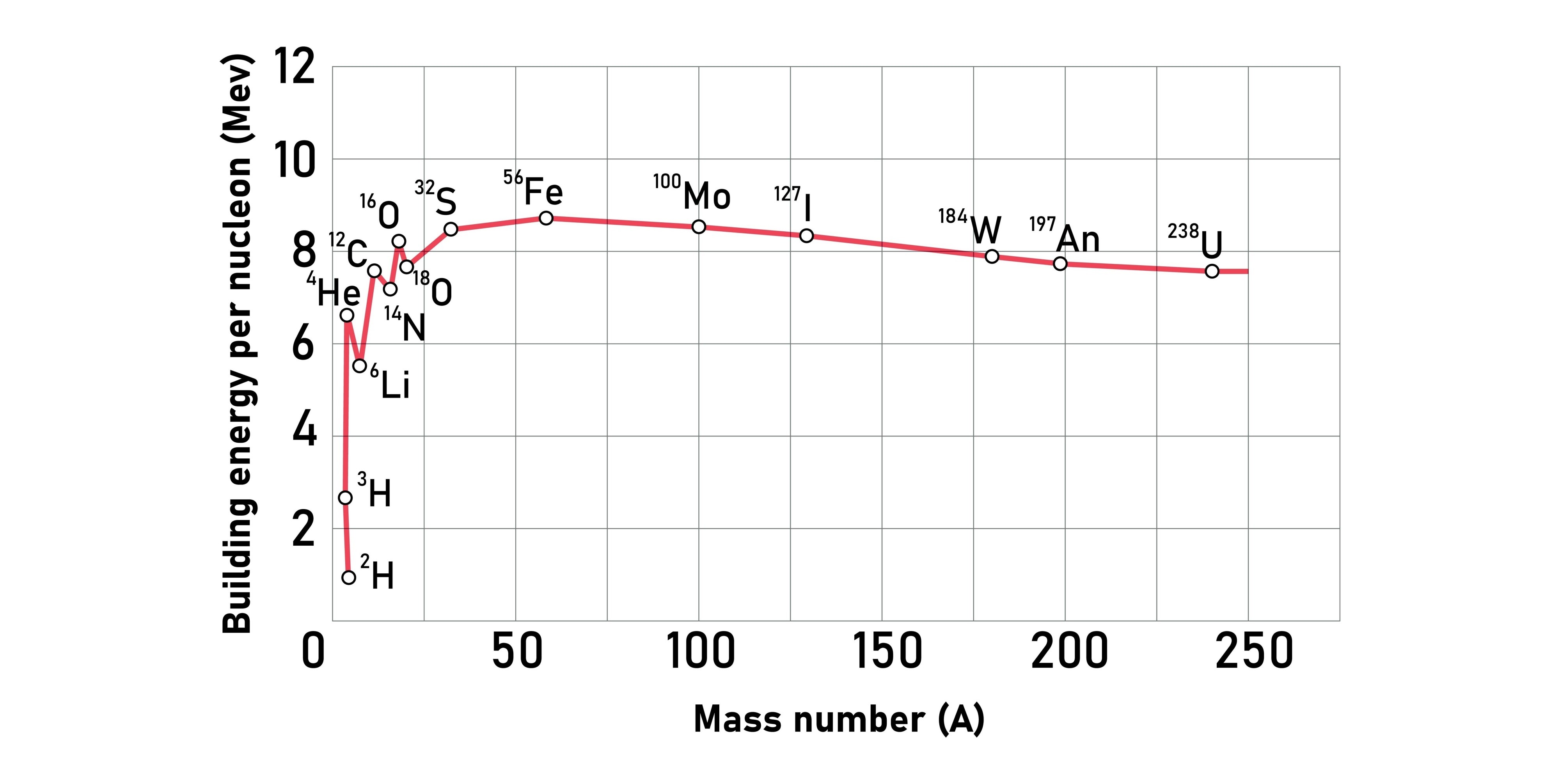
Describe the binding energy per nucleon curve.
It rises sharply for light nuclei, peaks at Iron-56 (the most stable nucleus), and then decreases slowly for heavier nuclei.
What are the products of alpha decay?
An alpha particle (two protons and two neutrons) is emitted, reducing the nucleon number by 4 and the proton number by 2.
What occurs during beta-minus decay?
A neutron decays into a proton, an electron, and an antineutrino; the proton number increases by 1 while the nucleon number stays the same.
Contrast the ionizing ability and penetration of alpha, beta, and gamma radiation.
Alpha has high ionizing power but low penetration; Gamma has low ionizing power but high penetration; Beta falls in the middle for both.
What is the definition of the half-life of a radioactive isotope?
The time taken for half of the radioactive nuclei in a sample to decay, or the time taken for the activity of the sample to decrease by half.
How does the neutron-to-proton ratio affect nuclear stability?
Small nuclei are stable with a 1:1 ratio, but larger nuclei require more neutrons than protons to provide enough strong nuclear force to counteract electrostatic repulsion.
What evidence do alpha and gamma spectra provide about the nucleus?
Because these radiations are emitted at discrete energy levels, they provide evidence that the nucleus itself has discrete energy levels.
How did the continuous energy spectrum of beta decay lead to the discovery of the neutrino?
Since beta particles are emitted with a range of energies rather than a single value, a third particle (the neutrino) must carry away the "missing" energy to conserve total energy.
What is the Radioactive Decay Law in words?
The number of remaining nuclei equals the initial number of nuclei times "e" raised to the power of negative decay constant times time.
How is the decay constant ($\lambda$) related to the half-life?
The half-life equals the natural logarithm of 2 (approximately 0.693) divided by the decay constant.
What is the definition of Activity (A) and its formula?
Activity is the rate of decay (decays per second), calculated as the decay constant times the number of undecayed nuclei.
Why must background radiation be considered when measuring the count rate of a sample?
Background radiation must be subtracted from the total measured count rate to find the "corrected" count rate of the specific radioactive source.