Lab Exam CHEM 3460
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
TA’s first name
Mehdi
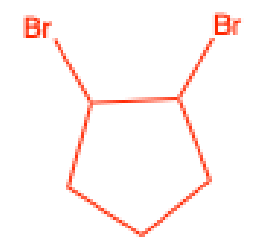
What does Br test for
Unsaturation
Positive Br test
immediate disappearance of the redish-brown color
What does the Lucas test test for
tertiary and secondary alcohols
Positive Lucas test
turning cloudy or forming an oily layer
Purpose of DNP test
take melting point
What does the Iodoform test test for
methyl ketones and secondary methyl alcohols
Positive Iodoform test
produces yellow precipitate
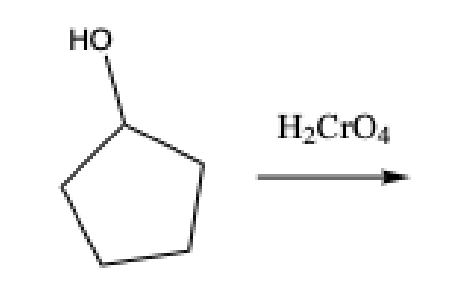
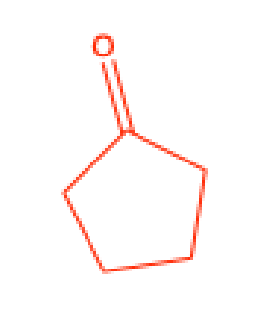
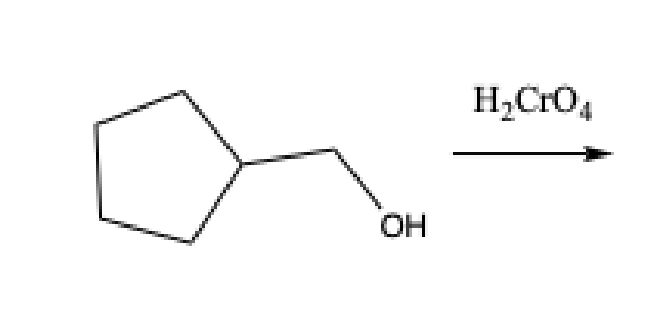
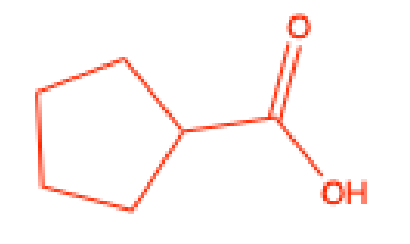
What does the Chromic Acid test test for
aldehydes, primary alcohols, and secondary alcohols
Positive Chromic Acid test
formation of a green precipitate
What does the Tollens test test for
aldehydes
Positive Tollens test
formation of a silver mirror
What is the purpose of recrystallization
to remove impurities
Characteristics of a good recrystallization solvent
dissolves impurities at all temps, but only dissolves solute at high temps
Does polar or nonpolar travel farther in TLC
Nonpolar
Does a more polar or more nonpolar solvent result in higher rf values
more polar
What gas is produced in Diels Alder lab and does it recondense
SO2 and yes
Why don’t we put a stopper on the drying tube
Some gas needs to escape so the system doesn’t explode
Why is CaCl removed from drying tubes
old CaCl is hard to get out of the drying tubes
Why are cyclic dienes more likely to dimerize
because they are locked into s-cis formation
Will a polar or nonpolar molecule travel faster through the chromatography column
nonpolar
Why don’t we let the level of solvent go below the top of the adsorbent
to avoid cracking, air bubbles, and uneven flow
What charge do EWG like
negative
What charge do EDG like
positive
Examples of EWG
NO2, CN, C=O, CA, SO3H
Examples of EDG
R groups, OR, OH, NH2, NR2, OCH3
Examples of Ortho / Para Directors
NH2, NHR, NR2, OH, OR, X, OCH3, R groups
Examples of Meta Directors
NO2, CN, CA, COOR,
Why do we use brine
to remove water from ether layer
Why are ketones more reactive than esters in a grignard reaction
the OR group in an ester makes the C=O less positive, which makes it less reactive
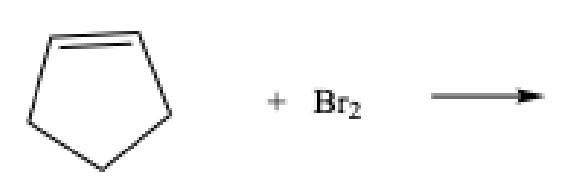
OH Stretch (alcohols and phenols)
3700 - 3200
OH Stretch (Carboxylic Acid)
3300 - 2500
C=O Stretch
1820 - 1630
C-H Stretch
3300 - 2700
Aldehyde C-H Stretch
2720
Carbon isotopes and ratio
12 and 13 (99:1)
Chlorine isotopes and ratio
35 and 37 (3:1)
Bromine isotopes and ratio
79 and 81 (1:1)
Nitrogen rule
If the molecular mass is an odd number, there are an odd number of nitrogens in the molecule