CHEM 335 CA3
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms

what combination of items shown in the image below would be needed to set up a simple distillation in the lab?
A+D+E+F+G+I+J+K+L+M
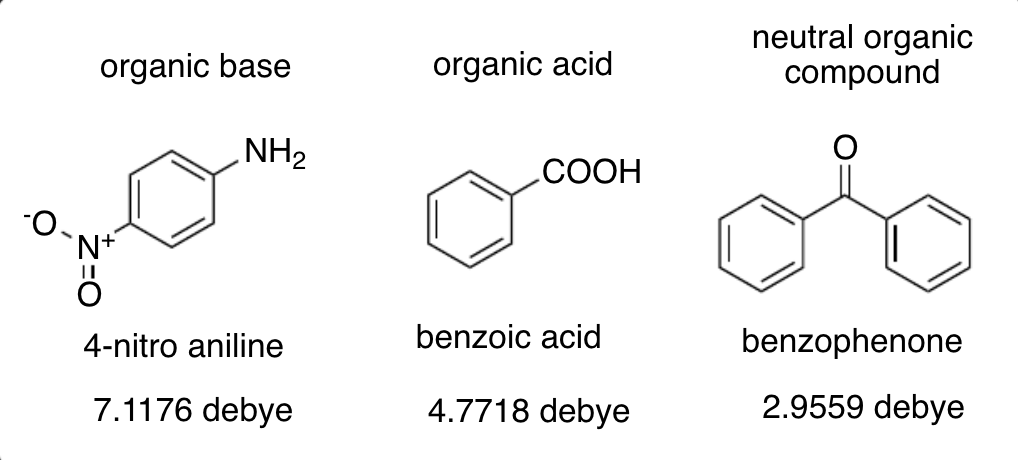
which compound will have the smallest Rf value on a developed TLC plate?
(suppose you start with a 1:1:1 weight mixture of the three compounds shown above)
4-nitro aniline
TLC plate Rf values and polarity relationship
the larger the polarity of the compound (larger Debeye) the smaller the Rf value, this is because the silica gel coating on the surface is polar (OH- groups) and thus polar compounds will stick to it and remain “stationary”
the main separation principle in thin layer chromatography (TLC) technique is through which principle?
adsorption
what is the purpose of boiling stones/chips?
to prevent uneven boiling in the liquid distilland
In IR spectroscopy, radiation absorption is accompanied by a change in what?
(*hint: think in units of debye)
a change dipole moment
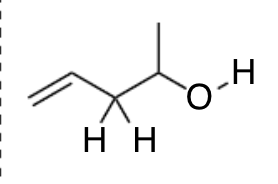
in this molecule, which bond would produce the highest intensity band? (peak with greatest IR absorbance?)
the O-H bond
what can happen to bonds of a molecule when they absorb IR radiation?
changes in molecular bond length and angles
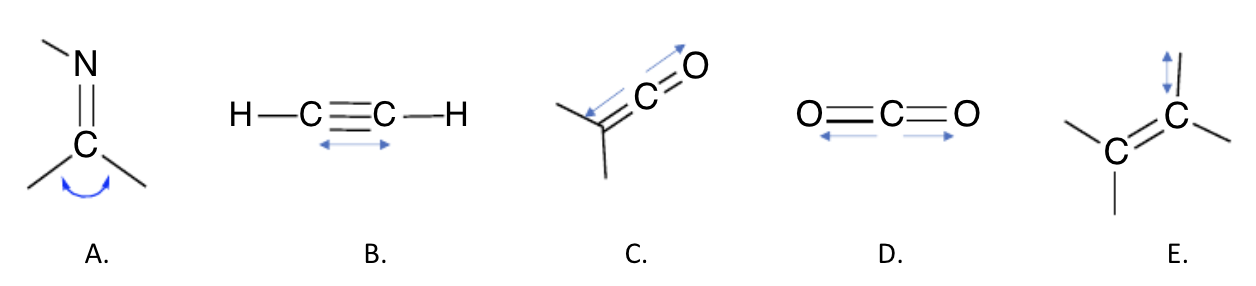
which of the vibrations won’t have a detectable IR absorption in the range usually associated with an absorption? (select all that apply)
B and D (bc net dipole is 0)
in Fischer esterification, an excess of one reagent is used to shift the equilibrium towards the product, which law is used to predict this effect?
Le Chatlier’s principle
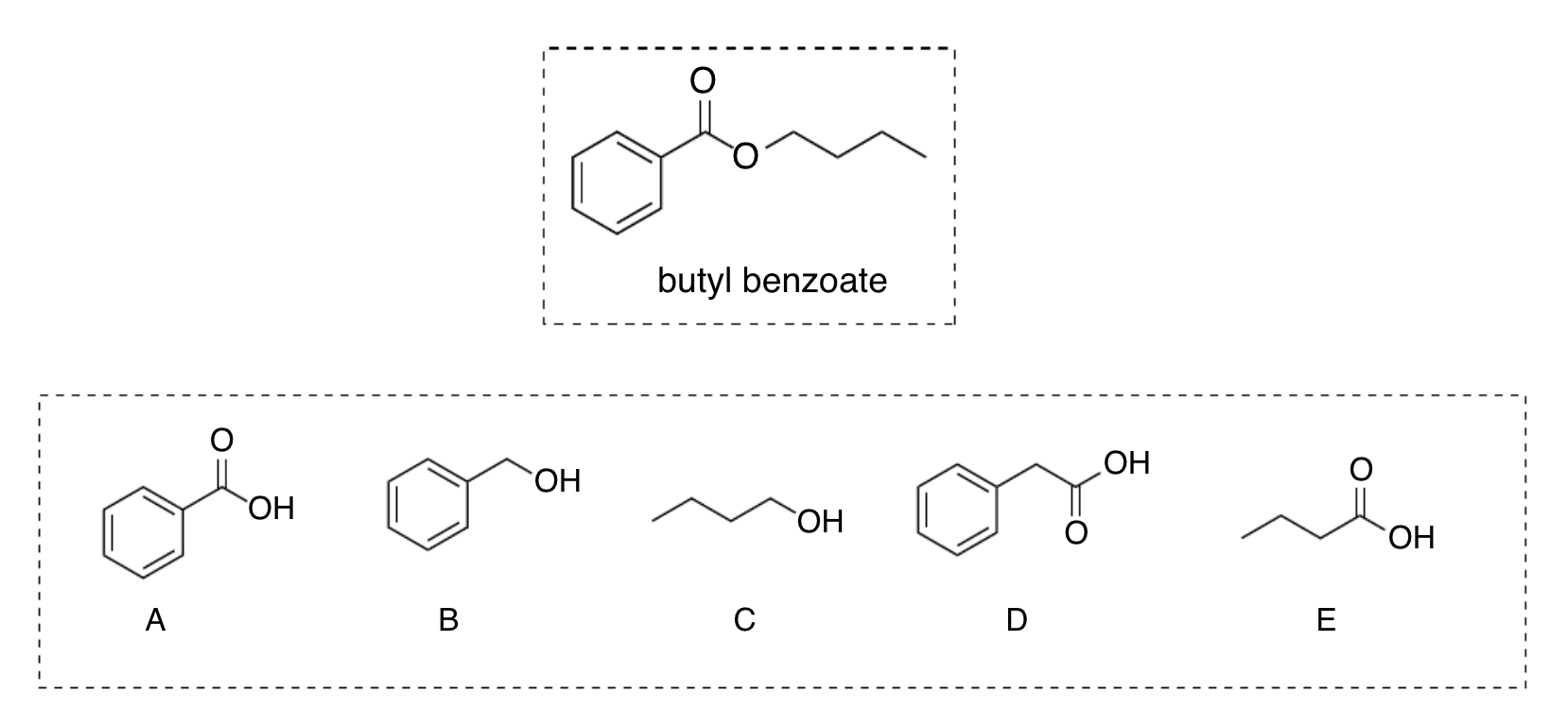
which reagents should be used to synthesize butyl benzoate by Fischer esterification method?
A and C (carboxylic acid + alkyl alcohol)
is the laboratory synthesis of isoamyl acetate, the reaction mixture is heat up under reflux for ~45 mins and the reaction is then terminated with a workup process that involves extraction of the crude reaction mixture with 10% bicarbonate solution. what is the purpose of sodium bicarbonate in the workup process?
to remove the unreacted acetic acid from the crude reaction mixture