Polymers
1/45
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
Name three types of condensation polymers.
Polyamides.
Polyesters.
Polypeptides.
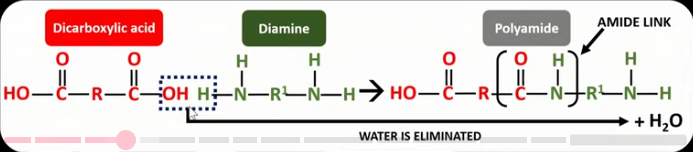
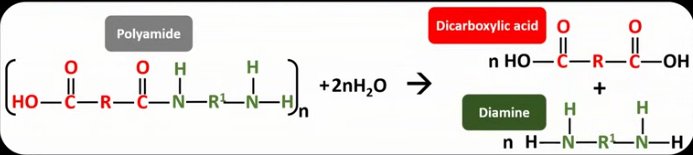
What is condensation polymerisation?
When two monomers with at least two functional groups react together, forming a link and eliminating a molecule of water.
Which monomers must you react together to form a polypeptide? What are the two functional groups on each of these monomers?
Amino acids. They each have one amine functional group and one carboxylic acid functional group.
Which monomers must react together to form a polyamide?
Diamines and dicarboxylic acids.
Which monomers must react together to form a polyester?
Dicarboxylic acids and diols.
What is the name of the link found in polyamides?
Amide link.
Why is it essential for monomers to be difunctional in polymerisation?
It means that there is always a free group to react and therefore long chains can form (the reaction wont stop after one reaction).
Draw the displayed formula for the reaction between the two monomers that form a polyamide. Label the amide link in the product. Label the atoms in the reactants used to make a water molecule.
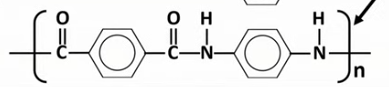
What type of condensation polymer is Kevlar?
A polyamide.
Give three examples of where Kevlar is used.
Bulletproof vests.
Sports equipment.
Car tyres.
In general, why is Kevlar useful?
It is lightweight but strong.
Draw the displayed formulae and name the two monomers that make up Kevlar.

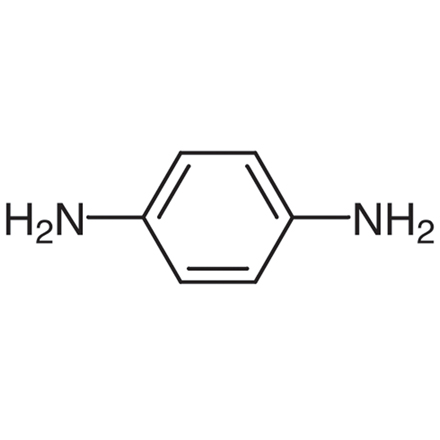
(Avoid using Kekule structure in exam) 1 = benzene-1,4-dicarboxylic acid, 2 = 1,4-diaminobenzene.
What type of condensation polymer is nylon-6,6?
A polyamide.
Draw the displayed formula of the repeat unit of Kevlar.
Give for examples of where nylon-6,6 is used in real life.
Parachute fabric.
Ropes.
Carpets.
Clothing fabric.
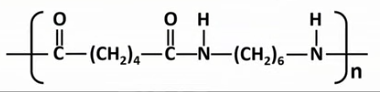
Draw the displayed formulae and name the two monomers that make up nylon-6,6.
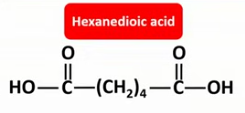
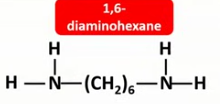
Draw the displayed formula of the repeat unit in nylon-6,6.
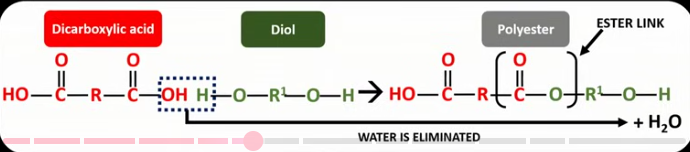
Draw the displayed formula for the general reaction between the two monomers that make polyesters. Label the ester link in the product. Label the atoms from the reactants that make up the eliminated water molecule.
What type of condensation polymer is terylene (PET)?
A polyester.
Give three examples of terylene (PET) being used in real life.
Plastic bottles.
Sheeting.
Clothes.
Draw the displayed formulae and name the monomers that make up terylene (PET).
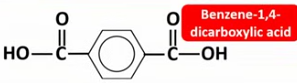
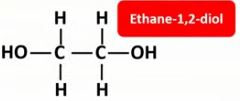
Draw the displayed formula of the repeat unit of terylene (PET).
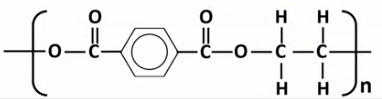
How many water molecules are eliminated per formation of a repeat unit of terylene (PET)?
Two
How many water molecules are eliminated per formation of Kevlar repeat unit?
Two.
How many water molecules are eliminated per formation of a repeat unit of nylon-6,6?
Two.
How can you produce the monomers that make up a polymer with the polymer?
Hydrolyse the polymer.
What is hydrolysis?
Splitting a molecule (breaking bonds) by adding water.
Draw the displayed formula for the hydrolysis reaction of a polyamide.
True or false? Polyamides and polyesters contain polar bonds.
True.
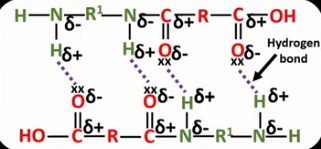
Why are condensation polymers often more rigid and stronger than addition polymers?
Condensation polymers contain polar bonds meaning they have intermolecular forces (hydrogen bonds, dipole-dipole, Van der Waals) between chains, addition polymers don’t.
Draw and label the intermolecular forces between two polyamide chains.
How does the polarity of condensation polymers make them more biodegradable?
As condensation polymers contain many polar bonds they can be easily dissolved (biodegraded) by polar solvents (such as water).
Give three general uses of synthetic polymers.
Plastics.
Digital technology.
Non-stick pan coating.
What type of species are condensation polymers susceptible to attack from and why?
As condensation polymers are polar they are susceptible to attack from nucleophiles.
Explain why polyalkenes are not biodegradable.
Polyalkenes are saturated molecules and normally non-polar, so they are chemically inert and unreactive.
Why are polyalkenes ok to use near food?
They are unreactive so don’t react with the food.
Give four examples of when landfill is used to dispose of plastics.
The plastic is not biodegradable.
The plastic is too difficult to recycle.
The plastic is too difficult to separate from other materials.
There is not enough plastic to extract to make it economically worth it.
Give two risks of using landfill to dispose of plastics.
When waste decomposes in landfill it produces methane, a greenhouse gas that contributes to global warming.
Waste can leach into water and contaminate it.
Give a way you can dispose of plastics while generating electricity.
Incineration (burning waste).
When could incineration be used to dispose of plastics?
If the plastics cannot be recycled.
How is electricity generated from the incineration of plastics?
When the plastics are burned they release energy which can be used to generate electricity.
What is a disadvantage of incinerating plastics?
They release toxic gases.
Give an example of a toxic gas released when a plastic is incinerated.
PVC releases HCl gas.
Give four advantages of recycling plastics.
Cheaper to recycle plastics than to make them from scratch.
Reduces our need to use (non-renewable) crude oil to make plastics.
Less carbon dioxide is produced recycling plastics than incinerating them.
Reduces reliance on landfill.
What are the two ways in which plastics can be recycled?
Cracked into monomers that are organic feedstock for making plastics and other substances.
Melted down and remoulded into something different.
Give four disadvantages of recycling plastics.
Plastics can be contaminated with other materials when being recycled.
Wide variety of different plastics can make them difficult to recycle.
Difficult to re-make original plastic from recycled material.
More expensive and time-consuming (sorting) than incineration.