5.1.3 Acids, Bases and Buffers (incomplete)
1/138
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
139 Terms
Define ‘acid’
a proton donor
Define base
proton acceptor
What is a monobasic acid?
an acid which releases one proton per molecule when it dissociates in water
How many protons per molecule do monobasic acids release when in water?
1
Give an example of a monobasic acid
HCl
How many moles of OH- are required to neutralise a monobasic acid?
1
Define dibasic acid
an acid which releases 2 protons per molecule when it dissociates in water
How many protons per molecule do dibasic acids release when in water?
2
Give an example of a dibasic acid
H2SO4
Define tribasic acid
an acid which releases 2 protons per molecule when dissociated in water
How many protons per molecule do tribasic acids release when in water?
3
How many moles of OH- are required to neutralise a tribasic acid?
3
Give an example of a tribasic acid
H3PO4
Define salt
the compound formed when the H+ ion from an acid is replaced by a metal or other positive ion such as an ammonium ion
Ionic equation for a metal and acid
2H+ + Mg → Mg2+ + H2
Ionic equation for a carbonate and acid
CO32- + 2H+ → CO2 + H2O
Ionic equation for an alkali and acid
OH- + H+ → H2O
Ionic equation for an oxide and acid
O2- + 2H+ → H2O
Ionic equation for ammonia and acid
NH3 + H+ → NH4+
When does the release of a proton from an acid happen?
when an acid is added to water
Define strong acid
an acid which completely dissociates into its ions in solution
Define weak acid
an acid which partially dissociates into its ions in solution
Compare the symbols used for strong and weak acids
strong = irreversible arrow
weak = reversible arrow
What happens to the proton released from an acid in water?
it is accepted by water to form an oxonium ion
What happens when a proton from an acid is accepted by water
forma an oxonium ion (H3O + )
Draw an oxonium ion
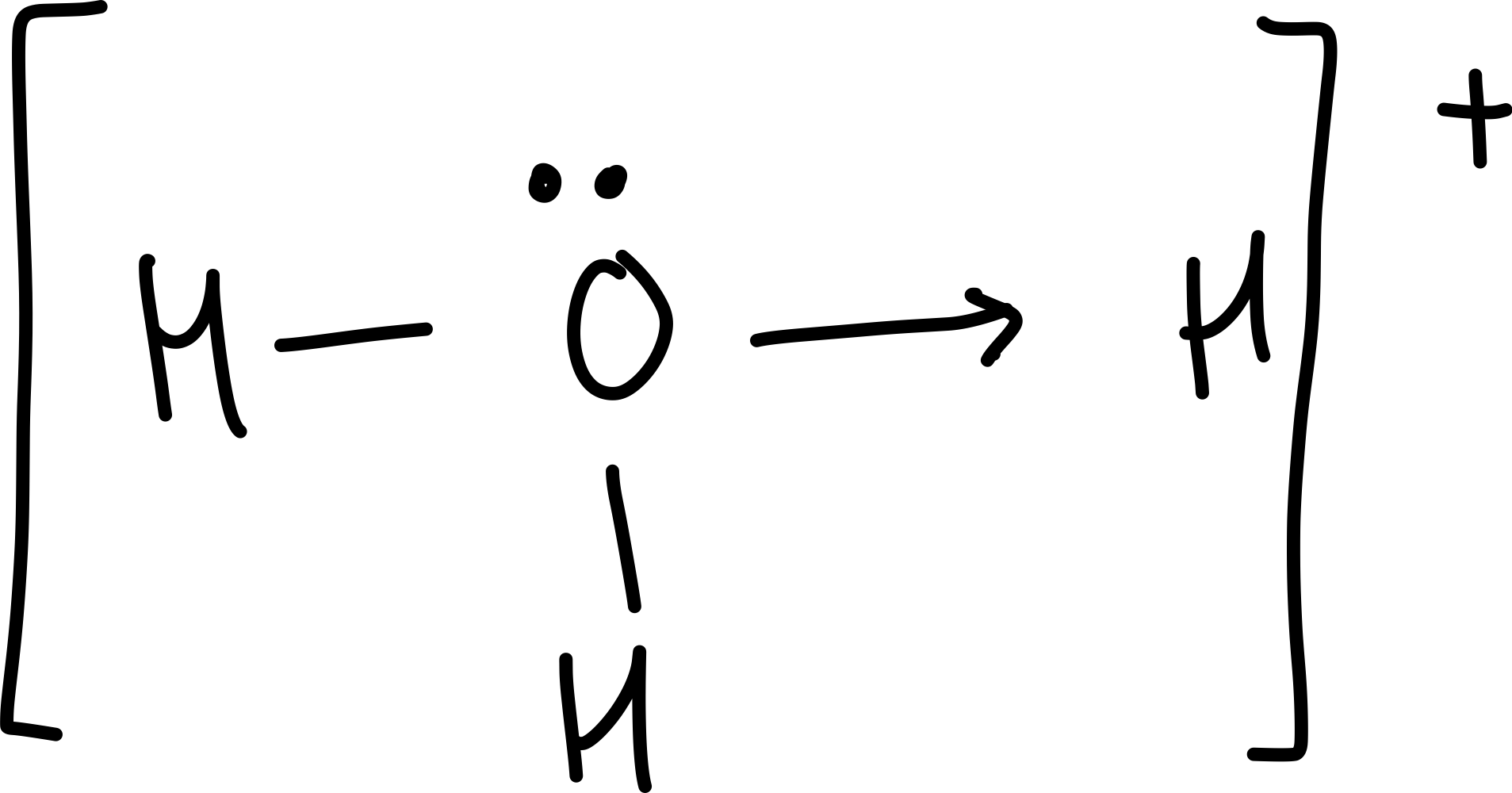
Why does water act like a base?
it accepts a proton
Give another name for oxonium ion
hydrated hydrogen ion
Write the equation for the reaction between HCl and water
HCl = H2O → H3O+ + Cl-
Define conjugate acid-base pair
a pair of two species that transform into each other by the gain or loss of a proton
Write the reaction for ethanoic acid and water
CH3COOH + H2O → H3O+ + CH3COO-
Draw a diagram for the acid-base conjugate pairs in the dissociation of ethanoic acid

Why does CH3COOH act as an acid?
it donates a proton
Why does H2O act as a base?
it accepts a proton
Why does Ch3COO- act as a base?
it accepts a proton
Why does H3O+ act as an acid?
it donates a proton
What are the acid-base conjugate pairs in the dissociation of ethanoic acid?
CH3COOH and CH3COO-
H2O and H3O+
How do conjugate acids and bases differ?
only by a proton
Write 2 equations for the dissociation of H2SO4
H2SO4 → H+ + HSO4-
HSO4- (reversible) H+ + SO42-
Write 3 equations for the dissociation of H3PO4
H3PO4 → H+ + H2PO4-
H2PO4- (reversible) H+ + HPO42-
HPO42- (reversible) H+ + PO43-
Describe the dissociation of weak and strong acids
strong = dissociation is complete as only the forward rection takes place
weak = dissociation is partial
Describe the concentration of protons in strong acids
high
Describe the concentration of protons in weak acids
low
Describe the equilibrium of a weak acid (homo/heterogenous)?
homogeneous
What is the equation for KA value?
Ka = [H+][A-] / [HA]
What are the units for KA value?
moldm-3
What does KA value indicate?
the extent of acid dissociation
Is KA is a large value, where does equilibrium lie?
far to the right, indicating a strong acid
Describe dissociation is Ka is large?
a lot of the HA is dissociated into its ions
When Ka is small where does equilibrium lie?
far to the left
Describe dissociation is Ka is low
a lot of the HA is not dissociated into its ions
How does Ka change as an acid gets stronger?
it increases
Why are pKa values used instead of Ka values?
they are more manageable
How do you calculate pKa from Ka?
-log(Ka)
How do you calculate Ka from pKa?
10^(-pKa)
What does Ka change with?
temperature
Does Ka change with concentration of acid?
no
Describe the extent of the ionisation of water?
very small
How many water molecules dissociate into their ions?
1 in 500 million
What is the ionisation of water known as (2)
self-dissociation
self-ionisation
Is the ionisation of water equilibrium homogeneous or heterogeneous
heterogeneous
What is Kw?
ionic product of water
What is the equation for Kw?
Kw = [H+][OH-]
What are the units for Kw?
mol2dm-6
What is Kw at 25*C
1.00 × 10^-14
Is the self-dissociation of water endothermic or exothermic?
endothermic
Equation for pH?
pH = -log[H+]
What does a low pH indicate?
high [H+]
What does a high pH indicate?
low [H+]
Describe the pH scale
logarithmic
How to calculate H+ from pH
10^-pH
How do you find the pH of a strong monobasic acid?
[H+] = [HA]
find H+ using HA
pH = -log[H+]
How do you find the concentration of a strong monobasic acid by pH?
[H+] = 10^-ph
[HA] = [H+]
How do you find the pH of a weak monobasic acid?
Ka = [H+][A-] / [HA]
Ka = [H+]²/[HA]
[H+]² = Ka * [HA]
square root
pH = -log[H+]
What assumptions are made when calculating pH of a weak monobasic acid?
[HA] dissociated = [HA] undissociated
ionisation of water is minimal
What are the limitations of Ka in calculating the pH of a monobasic acid?
stronger acids have a larger Ka value and greater degree of dissociation so the assumption that [HA] undissociated = [HA] dissociated may no longer be valid
weaker acids have a low [H+] meaning that the dissociation of water will be significant and the assumption [H+] = [A-] may no longer be valid
How do you calculate the concentration of a weak monobasic acid from pH?
[H+] = 10^-pH
Ka = [H+]²/[HA]
[HA] = [H+]²/Ka
How do you find the pH of a strong base?
find [OH-] from [base]
Kw = [OH-][H+]
[H+] = Kw/[OH-]
pH = -log[H+]
How do you find the concentration of a strong base from pH?
[H+] = 10^-pH
Kw = [OH-][H+]
[OH-] = Kw/[H+]
find concentration of base
What is an indicator used for in a titration?
determine the equivalence point
What is the equivalence point?
the point in a titration at which the volume of one solution has reacted exactly with the volume of the second solution. H+ concentration is equal to OH- concentration
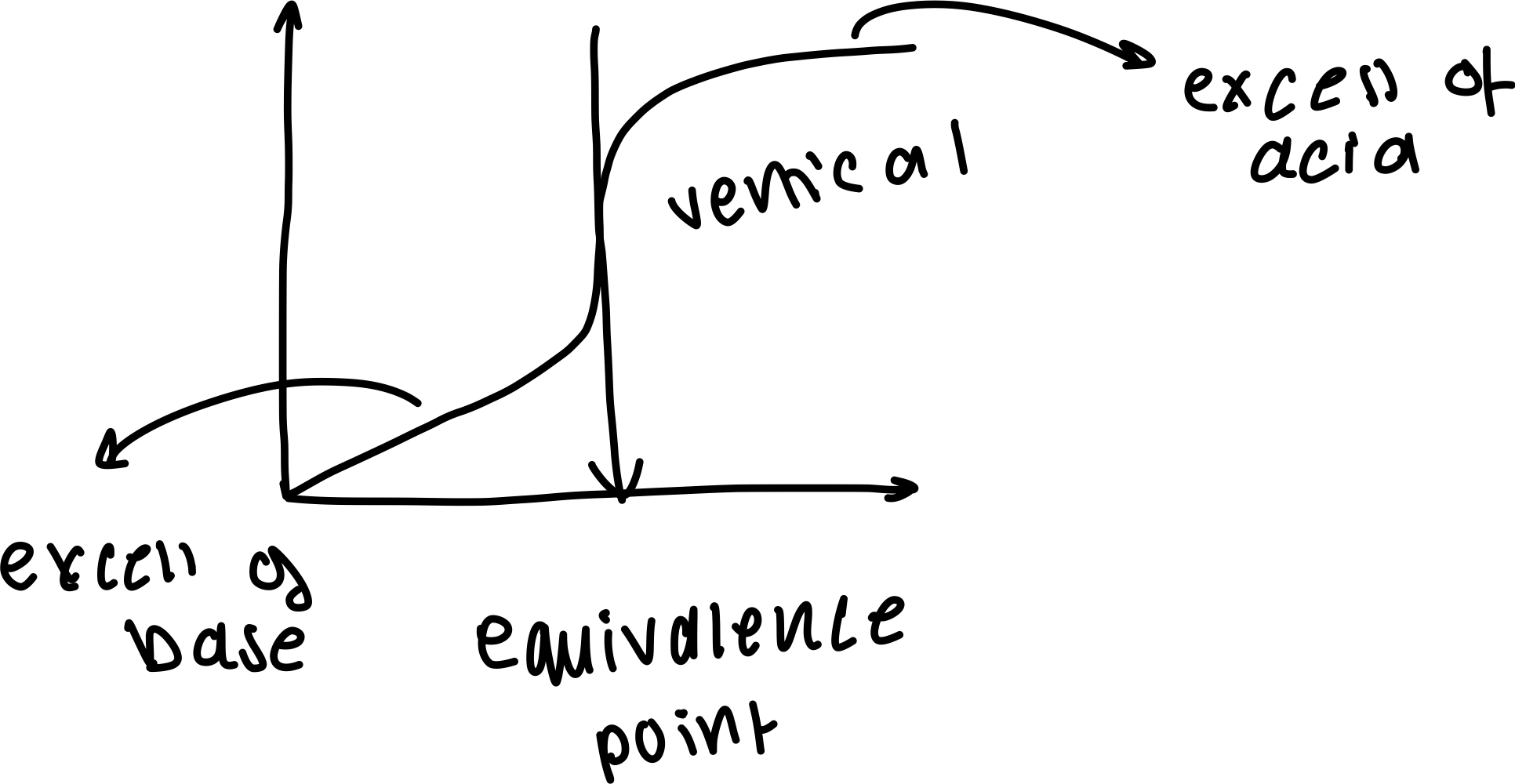
What does a pH curve show?
how the pH of a solution changes during an acid-base titration
How can pH be measured during a titration?
using a pH probe
What is a pH meter?
an electrode that is dipped into a solution to read the pH
How many decimal places does a pH meter read to?
2
Give an example method of a pH titration?
using a pipette, add 25cm³ of 0.100 moldm^-3 HCl to a conical flask
place the electrode of the pH meter into the conical flask
fill a burette with 0.100 moldm^-3 NaOH
add the sodium hydroxide too the conical flask 1cm³ at a time
after each addition swirl the contemt
Draw a labelled diagram of a general pH curve
Draw a pH curve where the base is in the conical flask and the acid is added in the burette

Draw a labelled diagram of the pH curve with a strong acid and strong base
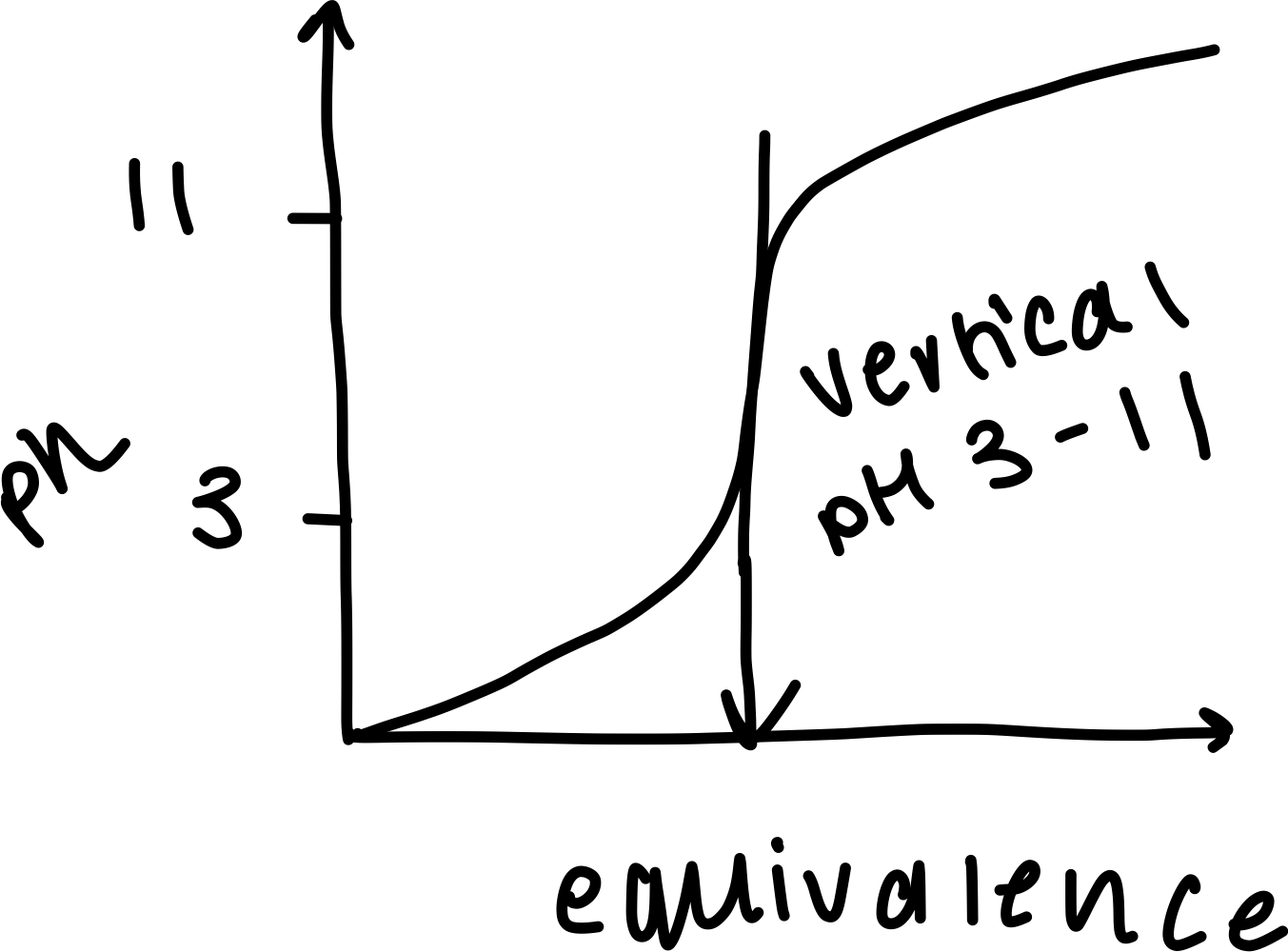
Draw a labelled diagram of a pH curve with a strong acid acid and a weak base
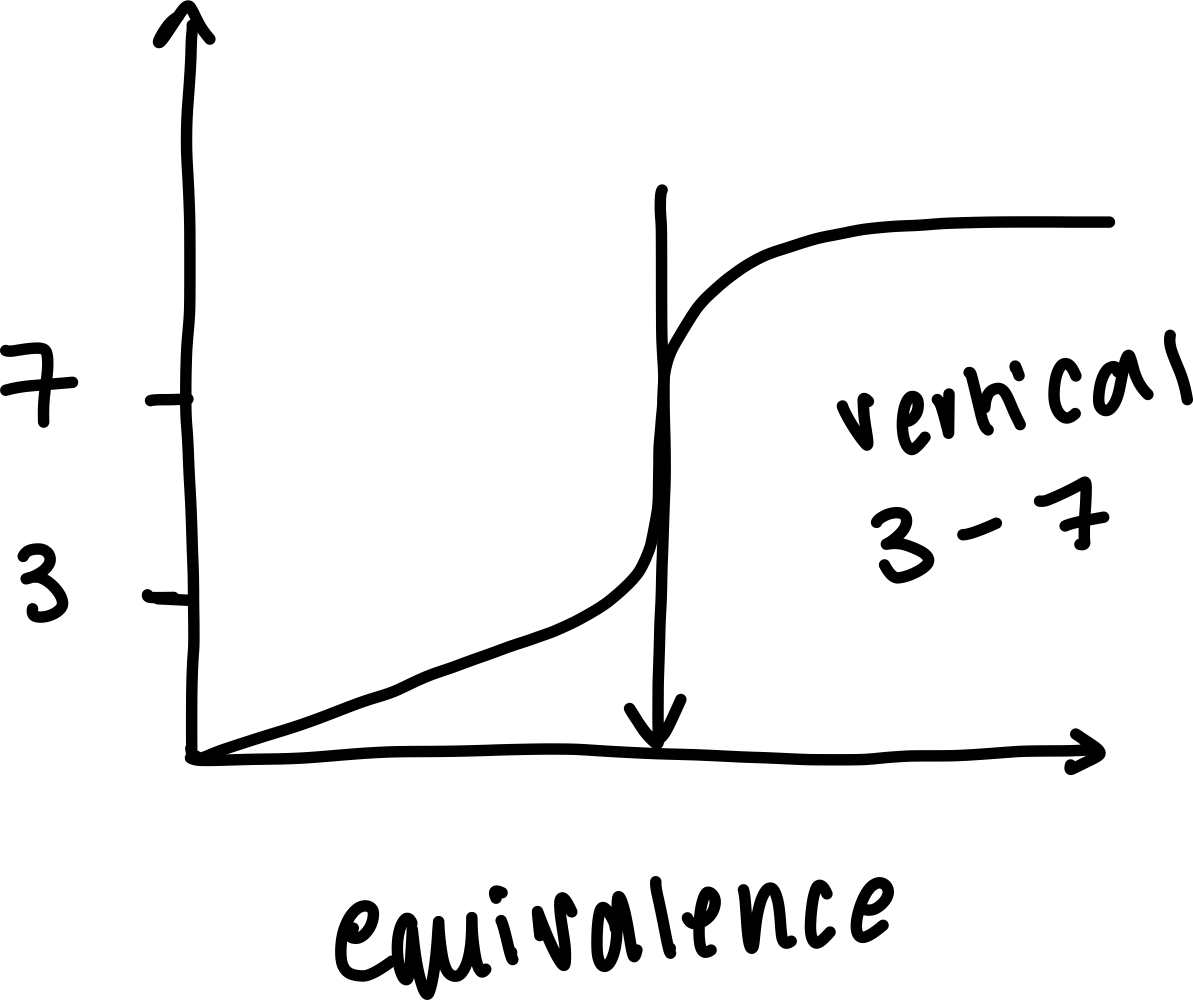
Draw a labelled diagram of a pH curve of a weak acid and strong base
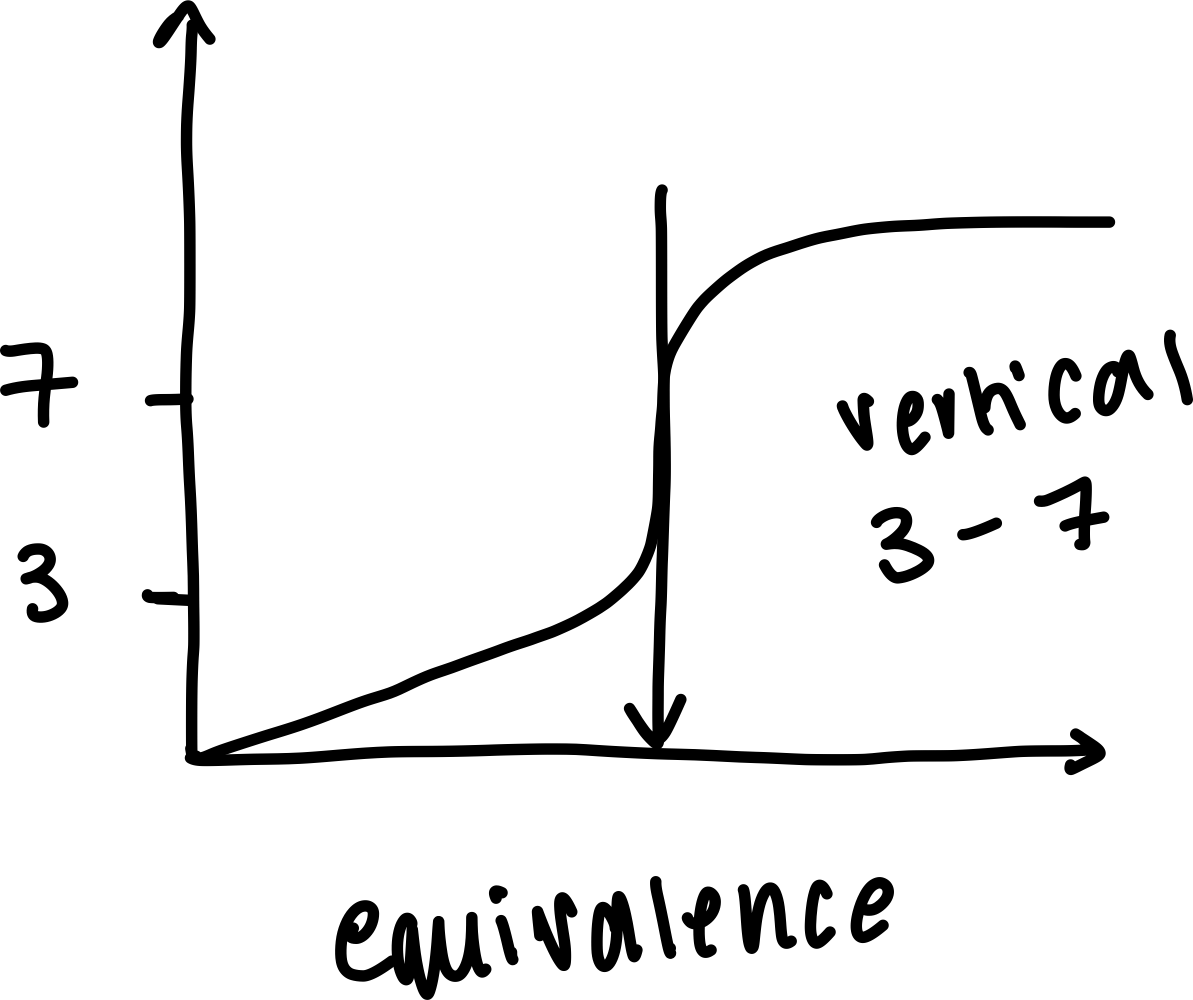
Draw a labelled diagram of a pH curve of a weak acid and weak base
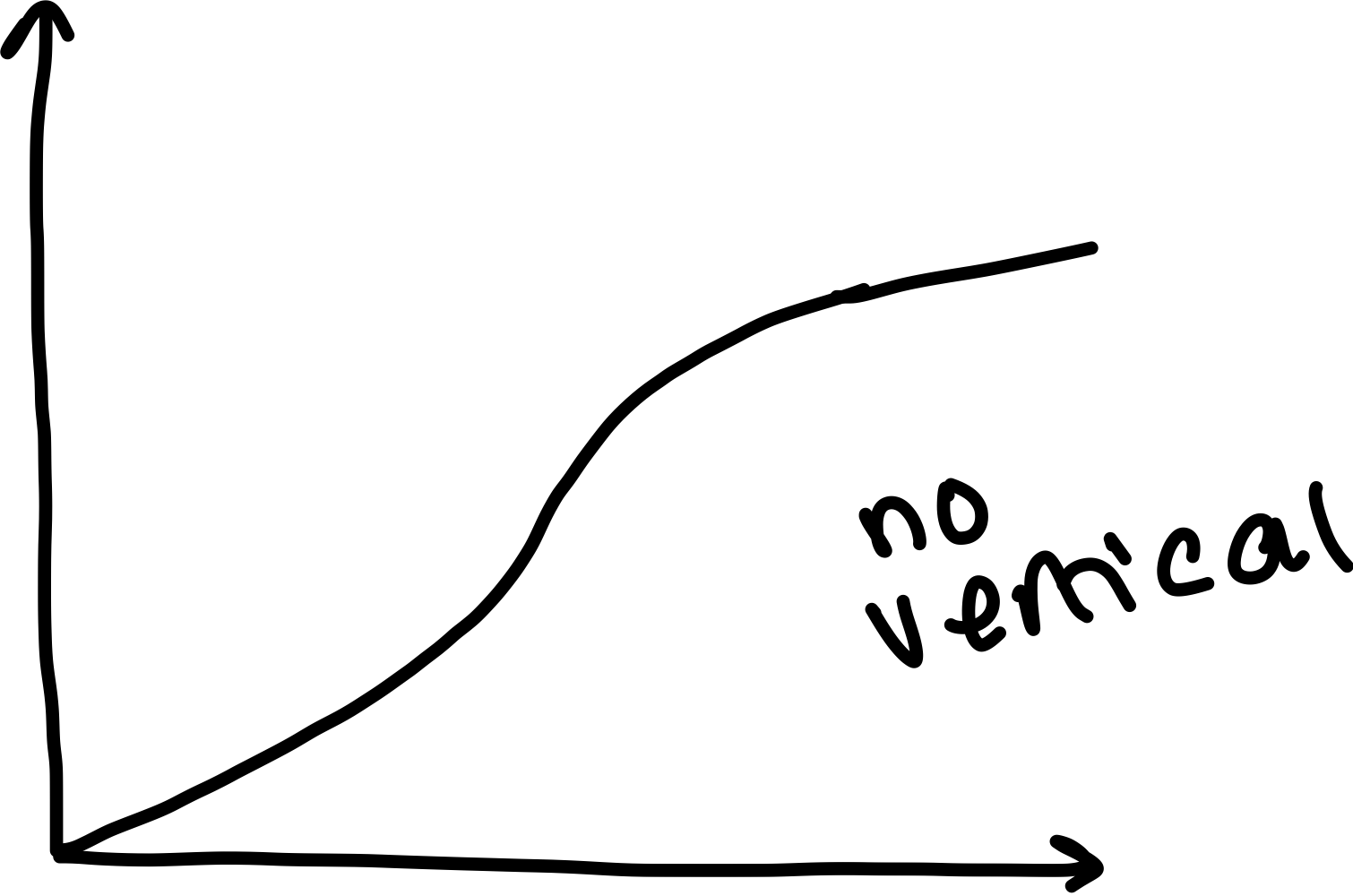
What are indicators?
chemical dyes that have a different colour in acid and alkali
Give 2 examples of indicators
methyl orange
phenolphthalein
What colour is methyl orange in acid?
red
What colour is methyl orange in alkali?
yellow
What are indicators usually?
weak acids where HA and A- are different colours
For methyl orange, what colour is the undissociated acid?
red
In methyl orange, what colour is the conjugate base?
yellow
What does the addition of an acid do to an indicator?
increase in concentration of H+
equilibrium shifts left to minimise increase
H+ reacts with A- to form more HA