acids and bases
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
If you add electrons to an element
what happens to the oxidation state?
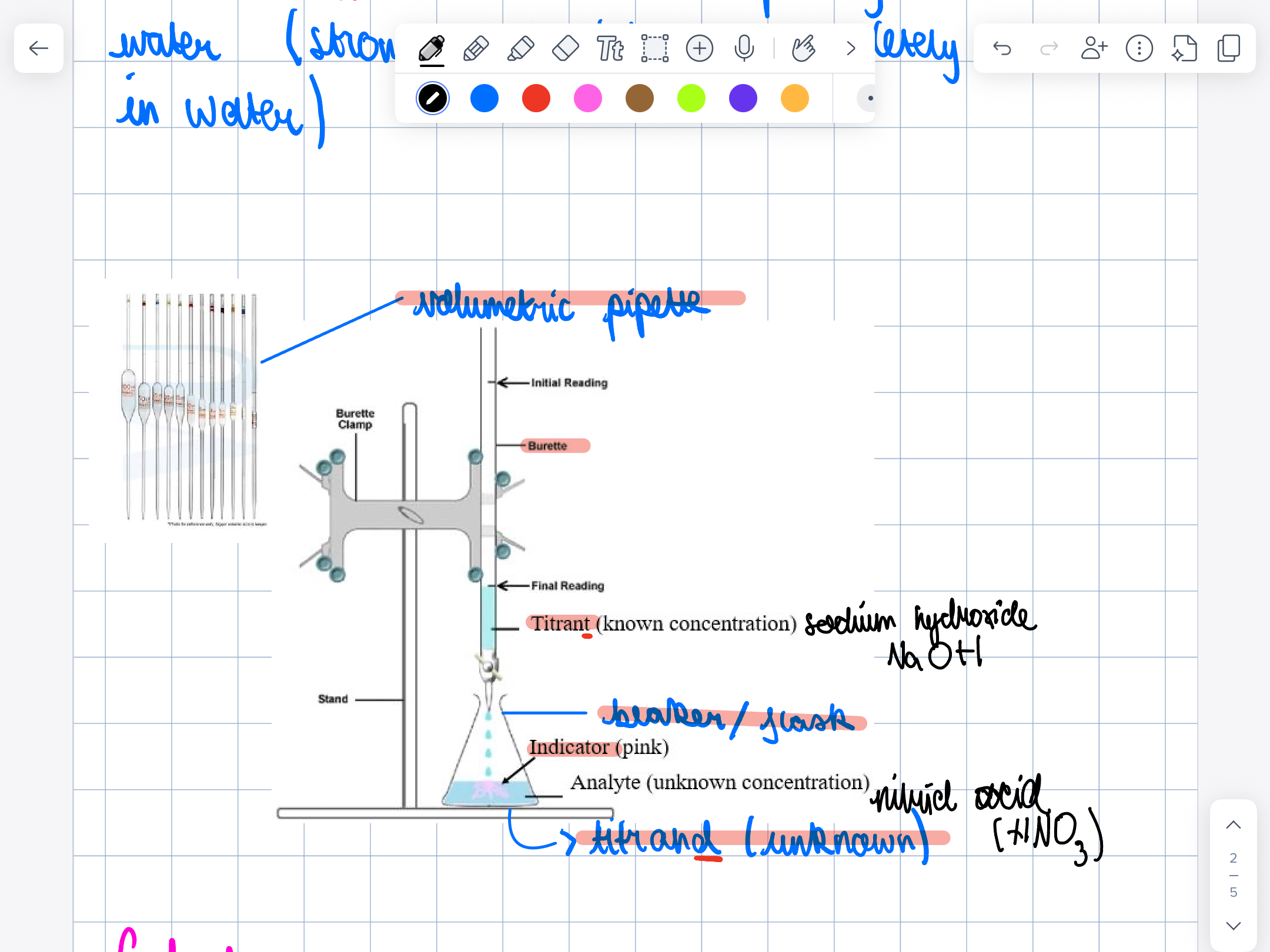
How do you calculate titration
n
—
C v
n:mol
c:mol/dm3
V= dm3 (:1000)
find moles of titrant n=cv
Moles have a 1:1 ratio
Use values of titrands to calculate concentration
What happens to the oxidation states when you subtract electrons to an element
Oxidation state will increase (oxidation)
Give an example of an ion in a redox reaction
V2+ the ion is 2+
V2+ -> V3+ +e- explain
-Each side is balanced 2 -> 3 + (-1)
What is an oxidation state?
the charge an atom would have if it were an ion in a compound (02- ox state is -2)
Oxidation
loss of electrons
reduction
gaining electrons
What is the oxidation state of a single element? (Cu
O2)
What's the oxidation state state in Poly atomic Ions
Oxidation state is the sum of all ions
What do the oxidation states in a neutral element add up to
0
What is an acid?
proton donor (H+)
Disassociation
Substances splitting into ions in water(strong acid/basis completely disassociate in water)
Explain increases in the pH scale
Change of one is equal to times 10 in H + on the scale
What is a base?
proton acceptor H+
An example of a strong acid
hydrochloric acid
An example of an acidic compound
Hydrochloric acid HCl
Given an example of a basic compound
NH3 ammonia
Give a example of a neutral compound
Water
What is a proton?
positively charged particle
Describe the pH scale and what it measures.
The pH scale measures the acidity or alkalinity of a solution. -0-14 0-7 acidic
Acidic pH
less than 7
Basic pH
greater than 7
Neutral pH
7
What is a conjugate base?
the particle that remains when an acid has donated a hydrogen ion
What is a conjugate acid?
the particle formed when a base gains a hydrogen ion
What’s in what’s indicator would be best to identify the equivalent points of a reaction between sodium hydroxide and nitric acid
Pink and white, shows the biggest contrast between colors, changes colors, exactly at equivalent point of pH 7
Label this