Chemical equilibria, Le Chatelier's principle, and Kc
1/25
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
Sketch a graph showing the concentration of reactants and products over time in a forwards reaction.
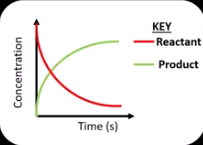
What is dynamic equilibrium?
The point in a reversible reaction where the forwards and backwards reaction are going at the same rate and the concentration of each substance remains constant.
True or false? Dynamic equilibrium can only occur in closed systems.
True.
State Le Chatelier’s principle.
If a reaction at equilibrium faces any change in pressure, temperature, or concentration, the position of equilibrium will move to oppose that change.
What happens to the yield of reactants and the yield of products in a reversible reaction if equilibrium shifts left?
Yield of reactants increases.
Yield of products decreases.
What happens to the yield of reactants and the yield of products in a reversible reaction when equilibrium shifts right?
Yield of reactants decreases.
Yield of products increases.
How will equilibrium shift if the concentration of a reactant is increased?
Equilibrium will shift right to decrease the concentration of reactant.
How will equilibrium shift if the concentration of a reactant is decreases?
Equilibrium will shift left to increase the concentration of the reactant.
How will equilibrium shift if the pressure is increased?
Equilibrium will shift to favour the reaction that produces the substances with less moles (less gas particles).
How will equilibrium shift if the pressure is decreased?
Equilibrium will shift to favour the reaction that produces the substances with the more moles (more gas particles).
How will equilibrium shift if there is an increase in temperature?
Equilibrium will shift to favour the endothermic reaction.
How will equilibrium shift if there is a decrease in temperature?
Equilibrium will shift to favour the exothermic reaction.
True or false? Catalysts have no effect on the position of equilibrium, they will increase the rate of both the forwards and backwards reaction equally. They will increase the rate at which equilibrium is reached but have no effect on yield.
True.
If a reaction in an industrial process is exothermic, decreasing the temperature will increase the yield of product. Why must a compromise temperature be reached?
If the temperature if too low the rate of reaction will be lower. A compromise temperature takes both product yield and rate of reaction into account.
What is the compromise temperature in the industrial production of ethanol?
300 degrees Celsius.
Why might a compromise pressure have to be chosen in an industrial reaction?
Higher pressure may give higher yields of product and increase the rate of reaction however high pressure environments are expensive (due to need for thicker vessels).
What is Kc?
The equilibrium constant.
Which values do you need to be able to work out Kc?
Molar concentrations of all the reactants and products in a reaction.
Reaction: 2A + B →← 2C + D
Write the Kc expression for this reaction.
Kc = ([C]2 [D]) / ([A]2 [B])
What does ICE stand for?
Initial moles, change in moles, equilibrium moles.
How do you work out the change in moles for an ICE table?
Use the mole ratios from the balanced symbol equation and apply those to the change in moles (- for forwards reaction, + for backwards reaction).
True or false? Changing concentrations changes the equilibrium constant.
False. THE ONLY THING THAT CHANGES THE EQUILIBRIUM CONSTANT IS TEMPERATURE.
What can you assume about the concentrations of two products at equilibrium if they have a 1:1 mole ratio?
The two products have the same concentration.
True or false? Kc is only valid for one temperature.
True.
How will Kc change if the temperature change causes equilibrium to shift right?
Kc will increase.
How will Kc change if the temperature change causes equilibrium to shift left?
Kc will decrease.