2.4. Saponification Number
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
The chemical breakdown (hydrolysis) of a triacylglycerol in the presence of Potassium Hydroxide (KOH) + heat.
Triacylglycerol + KOH + Heat → Fatty Acids + Glycerol
saponification
Reaction for saponification
Triacylglycerol + KOH + Heat → Fatty Acids + Glycerol
It refers to the amount of base (mg KOH) required to hydrolyze 1 gram of oil.
saponification number
What is the unit measurement of saponification number?
mg KOH/g oil
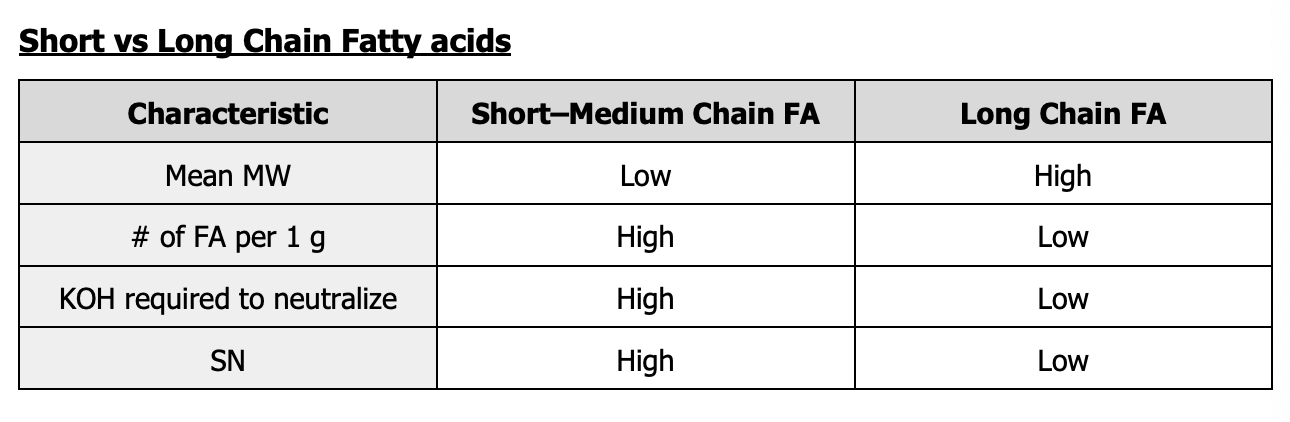
It serves as an indicator of the mean molecular weight of the fatty acids present in the sample.
Saponification number
What does a low or high SN indicate about the oil?
High - Short to medium chain FA
Low - Long chain FA
Saponification number formula
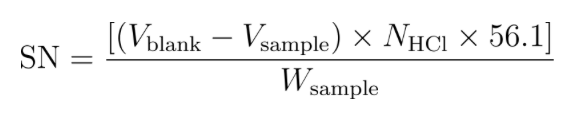
Differentiate between short-medium fatty acids and long chain fatty acids.
In the determination of saponification number, what is the purpose of making the KOH alcoholic?
Since fats are insoluble in water but soluble in alcohol, using alcohol as a solvent allows the KOH and the oil to mix, facilitating the hydrolysis of ester bonds. Its lower boiling point also allows faster boiling and evaporation.
In the determination of saponification number, what is the purpose of refluxing the KOH + oil mixture for 30 minutes?
Refluxing is the process of heating the oil + alcoholic KOH mixture while the flask is attached to a condenser. The heating allows for the saponification process to happen as it requires heat, the condenser prevents the loss of the volatile KOH and alcohol by condensing them back into the flask as a liquid, and doing this process for 30 minutes allows for the completion of hydrolysis of the ester bonds.
In the determination of saponification number, what is the purpose of using a condenser during the reflux period?
Boiling vaporizes the alcohol and volatile KOH. The condenser cools these vapors and returns them to the flask as a liquid, preventing solvent loss and keeping the reaction volume constant.
What is the titrant used in the determination of saponifican number?
0.25 N HCl
What is the indicator used in the determination of saponifican number?
phenolphthalein
In the determination of saponification number, what is the purpose of using HCl?
It acts as the titrant, which neutralizes the excess, unreacted KOH remaining in the flask after the lipid has been fully saponified.
In the determination of saponification number, what is the purpose of doing a blank titration?
The blank titration serves as a control that shows the total amount of KOH available, so the calculation reflects only the KOH used to saponify the lipid sample.
How does an insufficient reflux time (<30 minutes) affect the sample volume and SN of the oil sample?
Sample: Increase
SN: Decrease
Saponification does not go to completion. Fewer ester bonds are broken, so less KOH is consumed by the fat. The unreacted KOH remains artificially high, requiring more HCl to neutralize it.
How does using aqueous KOH instead of alcoholic KOH affect the sample volume and SN of the oil sample?
Sample: Increase
SN: Decrease
Without the alcohol to act as a co-solvent, the lipid matrix will not properly dissolve, preventing the KOH from reaching and breaking the ester bonds. Unreacted KOH remains high.
How does overtitration with HCl affect the sample vol and SN of the oil sample?
Sample: Increase
SN: Decrease
This increases the recorded sample volume. A larger sample volume reduces the difference between the blank, leading to a lower SN.
How does weighing by addition affect the sample volume and SN of the oil sample?
It fails to account for the oil that remains on the weighing boat, increasing the sample volume since there was less oil to consume the KOH. The difference between the blank and the sample decreases, which decreases the calculated SN.
How does using an expired HCl (<0.25 N) affect the sample volume and SN of the oil sample?
Increase both
Expired HCl becomes weaker, so you need more acid to neutralize KOH. The blank uses more acid than the sample, so its volume increases more and the gap (VB − VS) becomes larger.
How does using 0.1 N HCl (but accidentally leaving N=0.25 in the formula) affect the sample volume and SN of the oil sample?
Increase both
If you use the weaker 0.1 N acid (which makes the volume gap bigger) but still use the 0.25 multiplier in the formula, that larger gap will be multiplied and falsely increase the SN.
How does refluxing without a functional condenser affect the sample volume and SN of the oil sample?
Sample: Decrease
SN: Increase
Refluxing without a condenser leads to a falsely low titrant volume (S) due to the evaporation of the volatile solvent (alcohol) and the reagent (KOH), resulting in a falsely highSaponification Number.
What is the reagent used in the determination of saponifican number?
alcoholic KOH
How does prolonged exposure to open air during cooling affect the sample volume and SN of the oil sample?
Sample: Decrease
SN: Increase
Exposure to air neutralizes KOH via CO2 absorption, leading to a falsely low titrant volume (S) and an overestimation (falsely high) of the SN
How does failing to swirl the flask continuous during titration affect the sample volume and SN of the oil sample?
Failing to swirl causes over-titration (S increases), which shrinks the (B−S) gap and results in a falsely low SN.
How does refluxing for more than 30 minutes affect the sample volume and SN of the oil sample?
No effect
Additional heating with a condenser causes no further chemical reaction because the lipid is already fully consumed.
How does adding extra distilled water to wash down flask walls before titration affect the sample vol and SN of the oil sample?
No effect
Adding neutral water only dilutes the solution and does not change the total moles of unreacted KOH in the flask. Therefore, the same volume of HCl is required to reach neutrality.
How does adding extra drops of phenolphalein (> 3 drops) affect the sample vol and SN of the oil sample?
Phenolphthalein is a weak acid, but the extra micromoles added are mathematically negligible against a strong 0.25 N HCl titration.
How does handling the flask with bare hands affect sample vol and SN of the oil sample?
No effect
Titration is a volumetric analysis, not gravimetric. Adding physical mass (fingerprints, oils) to the exterior glass does not interact with the internal chemical stoichiometry.