Chem unit 12
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
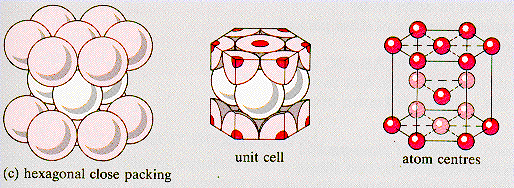
hexagonal close-packed (hcp)
crystal lattice in which the layers of atoms or ions in hexagonal unit cells have an ababab stacking patter (as tightly packed as poss)
unit cell
repeating unit of the arrangement of atoms, ions, or molecules in a crystal lattice (minimum repeating pattern)
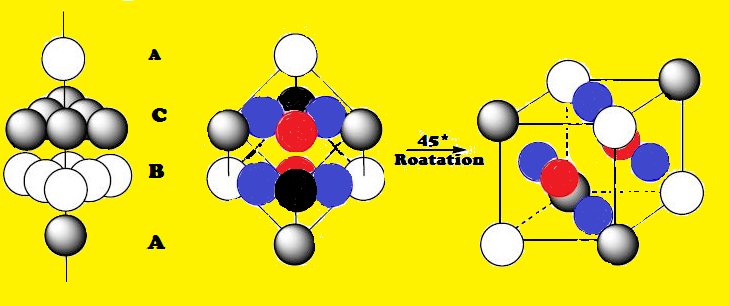
cubic close-packed (ccp)
a crystal lattice in which the layers of atoms or ions in face-centered cubic unit cells have an abcabc stacking pattern
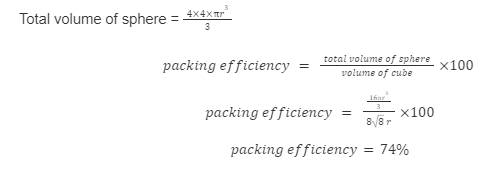
packing efficiency
the percentage of the total volume of a unit cell occupied by the atoms, ions, or molecules
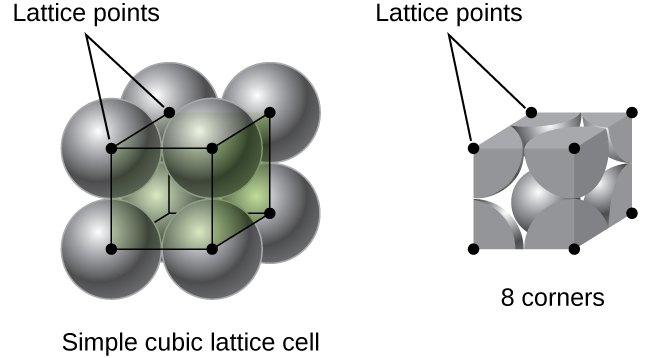
simple cubic unit cell
an array of atoms or molecules with one particle at the eight corners of a cube (square packing, aaa pattern)
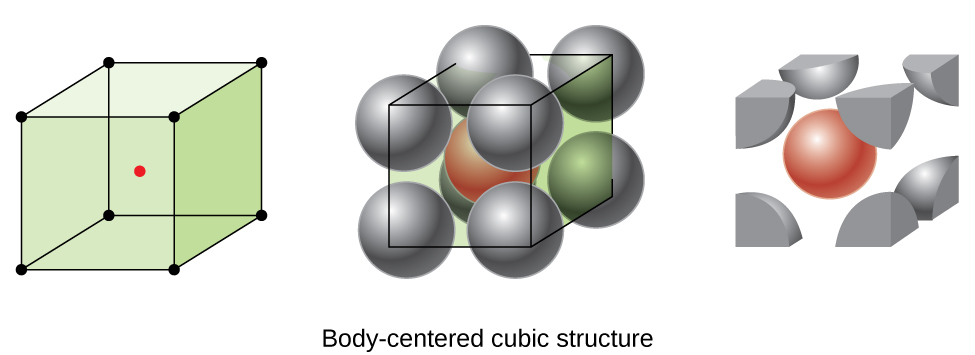
body-centered cubic unit cell
an array of atoms or molecules with one particle at each of the eight corners of a cube and one at the center of the cell (all group 1 metals and many transition metals)
formula for edge length
l = 2r
formula for sc
r = l/2 (1 eq atom)
formula for bcc
r= (l*sqrt 3)/4 (2 eq atoms)
formula for fcc
r = (l*sqrt 2)/4 (4 eq atoms)
coordination number (N)
number of atoms which it has close contact with (hcp: 12, ccp/fcc: 12, bcc: 8, sc: 6)
substitutional alloy
nonhost metal replaces host
interstitial alloy
added element occupies space between atoms of host
low carbon content
0.05-0.19%; malleable/ductile
medium carbon content
0.20-0.49%; high strength
high carbon content
0.5-3.0%; hard but brittle
Ionic crystals
lattice points are cations and anions, electrostatic attraction, hard/brittle/high melting point, poor conductor bc ions are held in place
Covalent crystals/network solids
lattice points are atoms, covalent bonds, hard/high melting point, poor conductor
Molecular crystals
lattice points are molecules, IMF, soft/low melting point, poor conductor bc neutral species
Metallic Crystal
lattice points are metallic bonds, soft to hard/low to high melting point, good conductors bc of mobile e-
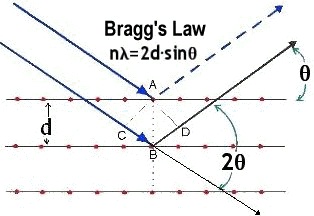
Bragg equation
mlambda=2dsintheta