Nuclear Fuel Cycle Front End
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Fuel, cladding, moderator and coolant of PWR
Fuel: enriched UO2
Cladding: zircaloy
Moderator: light water
Coolant: light water
Fuel, cladding, moderator and coolant of BWR
Fuel: enriched UO2
Cladding: zircaloy
Moderator: light water
Coolant: light water
Fuel, cladding, moderator and coolant of CANDU
Fuel: natural UO2
Cladding: zircaloy
Moderator: heavy water
Coolant: heavy water
Fuel, cladding, moderator and coolant of AGR
Fuel: enriched UO2
Cladding: stainless steel
Moderator: graphite
Coolant: CO2
What is the benefit of the two loop system in a PWR?
Second loop protected from cladding failure.
In the two loop system, pressurised water carries heat from core to boiling water circuit.
How many loops in a BWR?
One loop - fuel in core directly boils water
Basic concept of how a nuclear reactor generates electricity
Controlled nuclear chain reaction generates heat
Heat boils water
Boiled water produces steam
Steam drives a steam turbine to generate electricity
Why do we choose UO2 as fuel?
U-235 is the only naturally occurring fissile material
(Fissile = undergoes fission with thermal neutrons)
High melting point
Dense pellets obtained through sintering
What are the processes in the front end of the nuclear fuel cycle? What is the AIM?
Mining uranium ore
Milling ore to produced yellowcake
Conversion to uranium hexafluoride gas
Enrichment to increase concentration of U-235
Deconversion to UO2
Fuel fabrication to make pellets
AIM: To produce a pure UO2 pellet enriched in U-235 for fission
What are the conventional mining approaches, and how do they differ?
Open pit - uranium ore located near surface; uses drilling and explosives; benches to prevent collapse; destructive with high risks of contamination
Underground mining - uranium ore located deeper underground; less waste rock removed hence less environmental impact; exposure to radon gas
What is the milling process? What is the product of the milling process?
Milled to separate uranium from other constituents. Ore ground into powder and goes through a series of leaching and filtering processes to remove unwanted elements.
Converted into uranium concentrate U3O8 aka yellowcake.
What are the conventional milling approaches, and how do they differ?
Heap leaching - crushed ore laid on a gradient on thick plastic; sulphuric acid sprayed on ore for 30-90 days as the uranium is extracted as a liquid and treated; slower but cheaper than traditional milling
In-situ leaching - leaching solutions pumped into drill holes to dissolve ore minerals; uranium-rich fluid pumped to surface and uranium compounds extracted; minimal surface environmental disturbance; no waste rock; must avoid contaminating groundwater
Why do we enrich uranium fuel?
Mined natural uranium contains -0.7% U-235 (fissile isotope)
Enrichment increases this to 3-5%
Fission occurs when U-235 absorbs a neutron. Can make this more likely by slowing the neutrons down, and enriching the uranium.
What are the two routes for conversion? What do they produce?
Wet process and dry process.
Make UF6.
What is the conventional enrichment process?
Centrifuge
UF6 gas rotated at high speed
Slightly lighter U-235 stays in the middle
Heavier U-238 pushed by centrifugal force to the edge of the centrifuge.
Require a cascade of multiple centrifuges to gradually enrich the UF6 in U-235
What are the two steps in converting enriched UF6 back to UO2? (deconversion)
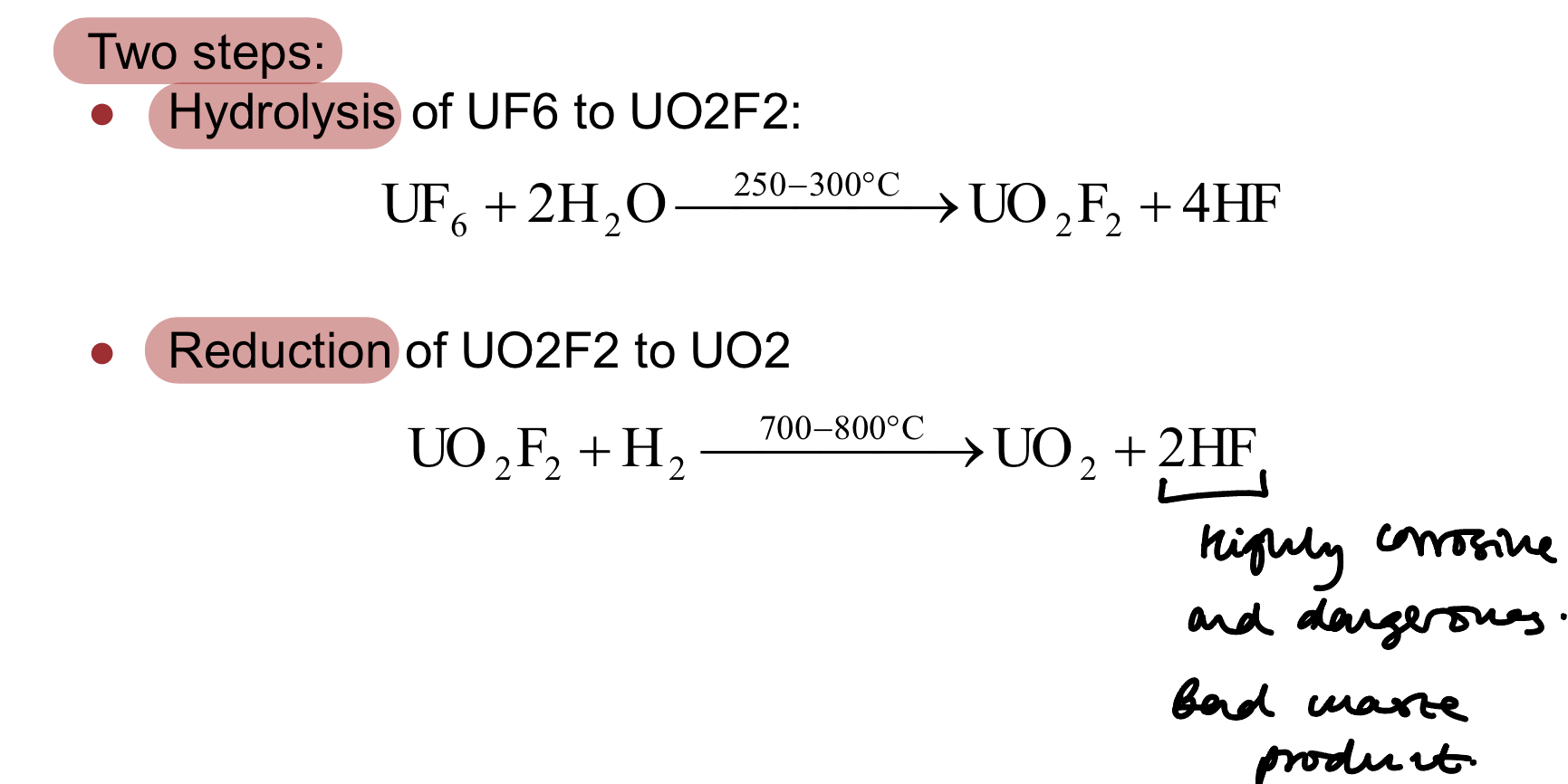
Describe the process of fabricating uranium dioxide pellets.
Pressed into green pellets
Sintered to remove pores
Rectified (to ensure same size and shape)
Want pellets as dense as possible: more fuel and safer
Define burnup
Amount of energy produced by tonne of fuel
Describe current burnup trends and implications
Average burn for PWRs in the range 40-50 GWd/tHM
BWRs in range 35-45 GWd/tHM
New plant designs aim to reach 60 GWd/tHM
GWd/tHM = gigawatt-day per metric tonne of heavy metal
Advantages of high burnup
Less fuel needed, less waste produced, operate for longer between refuelling.
Disadvantages of high burnup
Higher enrichment levels required, higher irradiation, zirconium cladding becomes brittle, more radioactive products, more decay heat.
What is the purpose of fuel cladding?
Acts as barrier between radioactive fission and activation products in the fuel and the coolant
Fuel must NOT make direct contact with coolant
Cladding prevents corrosion of fuel and accelerated release of fission products
Prevents potential for radioactivity to be released into the environment
Also acts as structural component of fuel assembly
Key criteria for fuel cladding materials
Low neutron absorption cross section
High melting point
Low thermal expansion coefficient
No dangerous activation products
Safe waste material
What is the most common fuel cladding material?
Zirconium alloy (zircaloy)
Key processes in zirconium production
Use chlorine chemistry to separate zirconium from hafnium.
Kroll process to get Zr metal from Zr ore
What is corrosion?
Deterioration of a material due to its environment.
Balance between thermodynamics and kinetics.
Thermodynamically, corrosion is inevitable (metals oxidise to return to a lower energy state).
Longevity of a material depends on kinetics (rates) of corrosion reactions.
Describe electrochemical corrosion as a redox reaction.
Oxidation = removing electrons from metal
Reduction = generating negative ions
Results in anodic and cathodic regions, with a potential difference between them due to the transfer of electrons.
What is the redox potential?
Measure of the tendency of a chemical species to acquire/lose electrons.
The more negative a materials potential, the more energetically favourable it is to oxidise (corrode).
Name the four requirements for corrosion
Anode
Cathode
Electrical connection
Ionic connection
Describe corrosion protection approaches
Corrosion inhibitor
Protective coatings
Cathodic protection
Name and distinguish between key corrosion phenomena
General - uniform loss of material over the entire surface at an approximately uniform rate
Galvanic -
Crevice - intensive, localised corrosion, occurring within shielded areas
Pitting - intensive, localised corrosion that results in holes/pits
Intergranular attack - localised corrosion that occurs when the microstructure of the grain boundary is more susceptible to attack, so corrosion spreads along the edges of the grain
Stress corrosion cracking - progressive fracturing that occurs in metals as a result of the combined influence of tensile stress and a corrosive environment
Describe corrosion control parameters implemented in nuclear power plants.