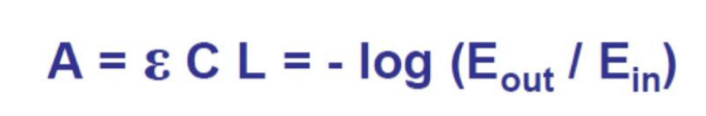1.1 HPLC
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
It is a technique which separates components in a mixture due to the differing time for each component to travel through a stationary phase when carried through it by a mobile phase.
Chromatography
An analytical technique used to separate, identify and quantify the individual components in any sample dissolved in liquid.
Liquid chromatography
This separation technique is suitable for non-volatile or thermally-unstable compounds.
Liquid Chromatography
These are the different types of chromatographic method.
Paper
Liquid
Gas
HPLC
It is is a chromatographic technique used to separate a mixture of compounds with the purpose of identifying, quantifying, or purifying the individual components of the mixture
HPLC
It is the is the moving phase in a chromatographic system, which can be liquid or gaseous.
Mobile phase (solvent)
It is the non-moving phase, consisting of a solid, liquid, or a solid/liquid mixture that is immobilized.
Stationary phase (column)
it is the component under study that moves with the mobile phase depending on its affinity to the solid phase
Solute
it refers to the solvent or mixture of solvents used in a chromatographic separation process.
Eluent
It is the resulting graph produced by the chromatograph instrument during a separation process
Chromatogram
An elution technique that maintains a constant eluent composition and is typically used for simple separations
Isocratic Mode
The principle is based on adsorption as well as partition chromatography depending on the nature of the stationary phase.
HPLC
In HPLC, if the stationary phase is solid, the principle is based on __.
adsorption chromatography
In HPLC, if the stationary phase is liquid, the principle is based on __.
partition chromatography
Examples of a moving phase.
Acetonitrile
Methanol
Water
Buffer
Examples of a stationary phase.
C18
C4
Amino
In the quantitative analysis in chromatography, the quantification is based on __.
Peak area/height
What are the different separation modes?
Normal-Phase
Reversed-Phase
Ion-Exchange
Size Exclusion
It refers to the localization of electrons within a molecule, which can cause molecules to have negative and/or positive poles.
Polarity
True or false: If a sample has a carboxyl group (-COOH), its hydrophobicity becomes stronger.
False. If a sample has a carboxyl group, its hydrophobicity becomes weak.
True or false: If a sample has an aromatic group, its hydrophobicity becomes stronger (aka it repels water more strongly and is highly non-polar).
True
Among the following molecules, which exhibits the strongest hydrophobicity?
Hexane (contains a 6-carbon −(CH2)n− chain)
Acetic acid (contains a −COOH group)
Ethanol (contains an −OH group)
Glycine (contains −NH2 and −COOH groups)
Hexane
If the sample contains these groups, its hydrophobicity becomes weak.
carboxyl
amino
hydroxyl
It separates polar analytes by partitioning onto a polar, bonded stationary phase.
Normal Phase
It separates non-polar analytes by partitioning onto a non-polar, bonded stationary phase.
Reversed Phase
It is the most common mode of separation.
Reversed Phase Chromatography
It separates moderately polar analytes, placing this technique in between Normal and Reversed Phase, by using a pure stationary phase, such as aluminum/silica.
Adsorption
It separates organic and inorganic ions by partitioning them onto ionic stationary phases bonded to a solid support.
Ion Chromatography
It separates large molecules based on the paths they take through the stationary phase.
Size Exclusion Chromatography
What are the two types of elution modes?
Isocratic Mode
Gradient Mode
This elution mode uses varying eluent compositions and is used for complex separations.
Gradient Mode
True or false: Reversed-phase chromatography utilizes a polar stationary phase and a non-polar mobile phase.
False. Reversed-phase chromatography uses a non-polar stationary phase and a polar mobile phase.
True or false: The primary goal of separation in HPLC is to have the best resolution possible between components.
True
True or false: A C18 (ODS) column is a common type of non-polar stationary phase used in reversed-phase chromatography.
True.
What are the common stationary phases used in normal phase chromatography?
Silica (SiO2)
Diol (-OH)
Aminopropyl (-NH2)
Cyanopropyl (-CN-)
Compare the stationary phases of Normal Phase and Reversed Phase Chromatography.
Normal Phase - Polar stationary phase
Reversed Phase - Non-polar stationary phase
Compare the mobile phases used in Normal Phase and Reversed Phase Chromatography.
Normal Phase - Non-polar mobile phase
Reversed Phase - Polar mobile phase
What is the difference between adsorption chromatography and partition chromatography in relation to the stationary phase?
Adsorption chromatography is the underlying principle when the stationary phase is a solid.
Partition chromatography is the principle when the stationary phase is a liquid.
What is the formula of the Lambert-Beer’s Law?