organic synthesis
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
what do you need to be able to do as part of a PAG?
prepare a sample of an organic liquid
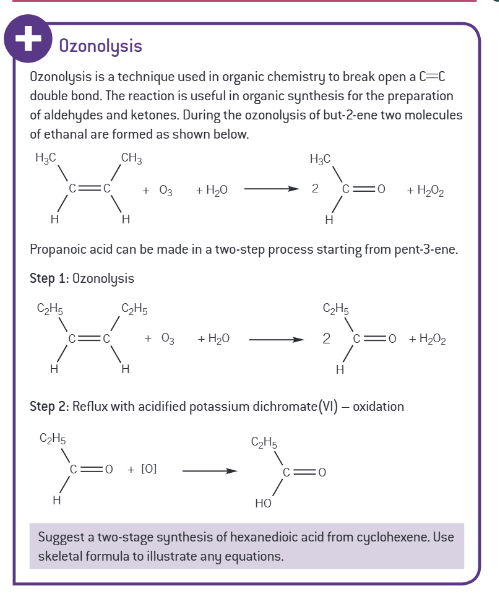
what is in a standard set of quickfit apparatus (necessary for organic preparation)?
round bottomed/ pear shaped flask
receiver
screw tap adaptor
condenser
still head
how do many organic reactions occur?
slowly at room temp
what is common in organic reactions?
for them to be heated to overcome the activation energy and increase the rate of reaction
whats a common procedure to prepare an organic liquid without boiling off the solvent, reactants, or products?
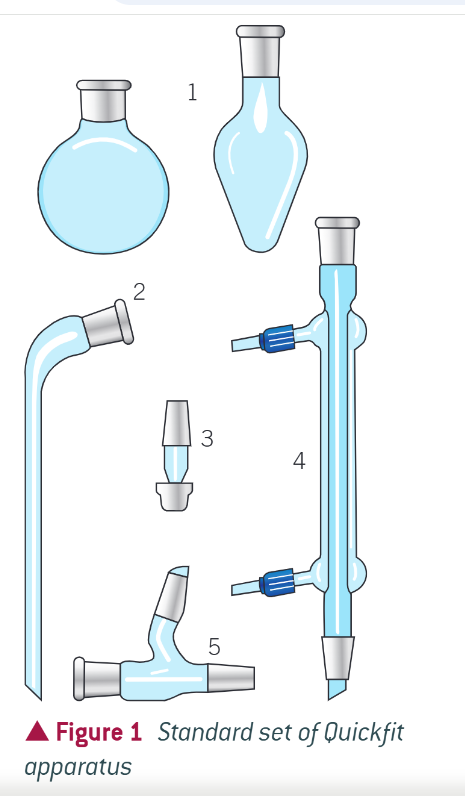
heating under reflux
what pieces of apparatus do you need for heating under reflux?
round bottom flask
condenser
rubber tubing
stand and clamp
heat source (bunsen burner, tripod, heating mantle, water bath if carried out under 100 degrees))
experimental setup for heating under reflux:
what heat source should you use if you are heating flammable liquids?
a heating mantle so that theres no naked flame present, for added safety in case apparatus breaks
what should you do before fitting the condenser to the flask?
add the reaction mixture and add anti bumping granules
why are anti bumping granules added?
so the contents boil smoothly. if they arent added, large bubbles form at the bottom of the liquid and make the glassware vibrate and jump violently
what else do you add before joining apparatus?
apply a thin layer of grease to the ground glass joint, then join the apparatus carefully and rotate back and forth to provide a good seal and ensure the apparatus comes apart at the end of the experiment
how is the condenser kept during reflux?
in the upright position
what is important things with the condenser?
only clamp condenser loosely as they are fragile and easily broken
never put a stopper in the top, otherwise you would have a closed system and pressure would build as heat air expands (breaks apparatus)
what is rubber tubing used for?
to connect the inlet of the condenser to the tap and the outlet to the sink
how does water always move in the system?
always enters at the bottom and leaves at the top to ensure that the outer jacket is full
what does heating under reflux enable?
enables a liquid to be constantly boiled whilst the reaction takes place, preventing volatile components from escaping and the flask from boiling dry

what happens to the vapour?
rises up the inner tube of the condenser until it meets the outer jacket containing cold water
the vapour then condenses and drips back into flask
what may happen with chemical reactions?
they may not go to completion or may produce by-products as well as the desired product
what has to happen once reflux is complete?
the crude oil/ solid product present in the flask needs to be purified to remove any by products and remaining reactants
what is distillation?
a common method used to separate a pure liquid from its impurities
what apparatus do you need for distillation?
round bottomed flask
condenser
rubber tubing
heat source
stand and clamp
screw cap adaptor
receiver adaptor
still head
thermometer
set up for distillation:
flask clamped by its neck
still head connected to flask
still head adaptor is T shaped and has 2 ground glass joints
one joint fits to screw cap adaptor, other fits to condenser
grease joints so they come apart after
2nd clamp placed round the receiver at the point at which it is attached to condenser - removes the need to clamp the condenser

what is also used in distillation similarly to reflux?
rubber tubing to connect the inlet (top) of the condenser to the tap and the outlet (bottom) to the sink
what is a flask used to do?
collect the distillate so that the distillation apparatus isnt airtight
what happens once the apparatus is set up?
the flask is heated and the mixture in the flask will start to boil. the different liquids in the mixture will have different bp, so the liquid with the lowest bp will boil first
what happens to the vapour in the experiment?
moves out of the flask up to other parts of apparatus, leaving the other liquids
when vapour reaches the cold condenser, it condenses and becomes a liquid
this liquid drips to the collecting flask
what may happen when preparing samples of organic liquids?
water may be obtained with the product, and you will see 2 layers in the collection flask
is it easy to separate the 2 mixtures?
yes, but you have to know which layer is organic so you dont throw it away
how do you identify this?
add some water to the mixture. the layer that gets bigger is the aqueous layer
how do you then separate the mixtures?
using a separating funnel

process of doing this:
ensure that the separating funnel tap is closed
pour the mixture of liquids into the separating funnel, place a stopper in the top of the funnel, and invert to mix the contents
allow the layers to settle
add some water to see which layer increases in volume: aq layer
place a conical flask under the separating funnel, remove the stopper and open the tap until the whole of the lower layer has left the funnel
place a second conical flask under the separating funnel to collect the other layer
label flasks so you dont muddle aq with organic
what may be the case in preparing using acids?
your impure product may contain acid impurities
how can these impurities be removed?
add aqueous sodium carbonate and shake the mixture in the separating funnel
what will then happen to any acid present?
it will react with the sodium carbonate, releasing CO2 gas. the tap needs to be slowly opened, holding the stoppered separating funnel upside down to release any gas pressure that may build up. the aq sodium carbonate layer can then be removed and the organic layer can be washed with water before running both layers into separate flasks
what may still remain in the organic product?
water
how is the water removed?
by adding a drying agent to the organic liquid
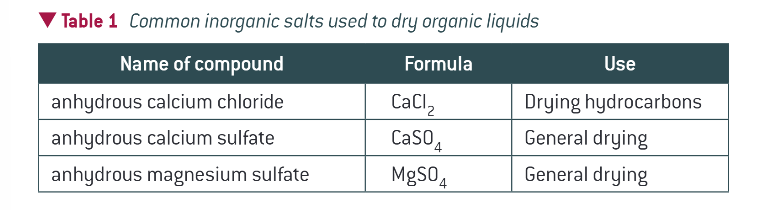
what is a drying agent
an anhydrous inorganic salt that readily takes up water to become hydrated
most common drying agents:
procedure for drying an organic liquid:
add the organic liquid to a conical flask
use a spatula and add some drying agent to the liquid and gently swirl the contents to mix together
place a stopper on the flask to prevent your product from evapourating away. leave for 10 mins
if solid has stuck in a lump, there is still water present. add more until solid is dispersed in the solution as a fine powder
decant the liquid from the solid into another flask. if liquid is dry it should be clear
what is sometimes the case?
organic liquids have boiling points that are relatively close together, so your prepared sample may still contain some organic impurities. the distillation apparatus is cleaned and dried and set up again so that a second distillation can be carried out
what should you do with this second distillation?
only collect the product with the boiling point of the compound youre trying to make
the narrow the boiling range…
the purer the product
what is organic synthesis?
the preparation of complex molecules from simple starting materials. with it, chemists can make new structures that can be investigated for their uses
what are some uses?
manufacture of modern medicines targeted at diseases
what are many chemists employed by to do?
employed by large pharmaceutical companies to synthesise and test new medicines, eg. those to target the growth of cancer cells in the human body
what do you need to do before attempting organic synthesis?
know the different functional groups and be able o identify them in unfamiliar molecules containing several functional groups
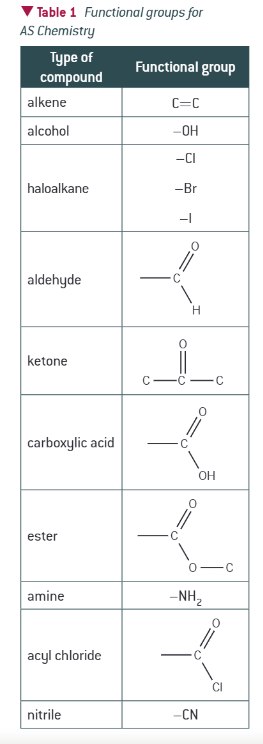
functional groups for AS chemistry:
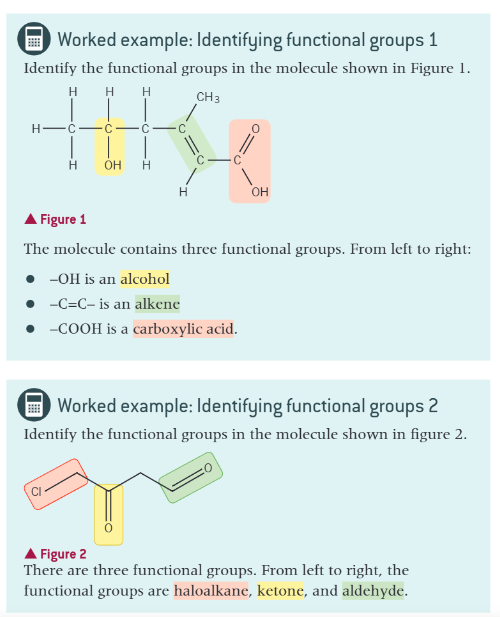
worked examples: identifying functional groups 1 and 2:
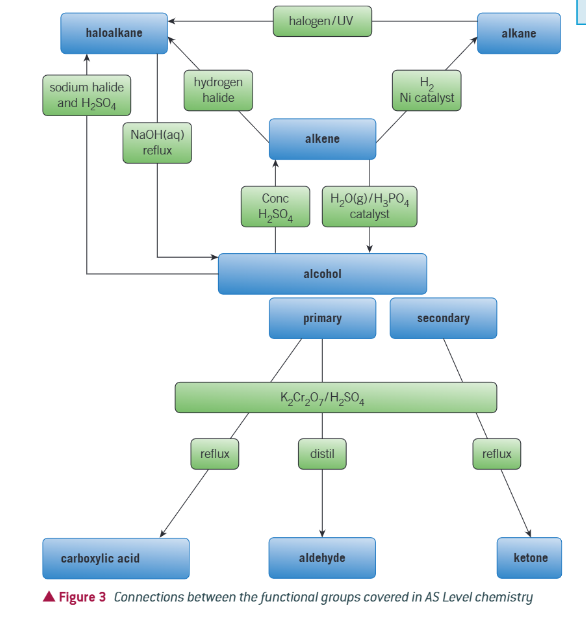
flowchart to link together all the functional groups (useful for predicting reaction and solving synthesis problems):
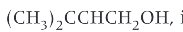
what is prenol?
a natural compound found in many fruits and widely used in organic synthesis
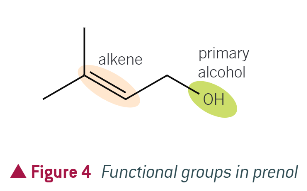
functional groups of prenol relating to its properties and reactions:
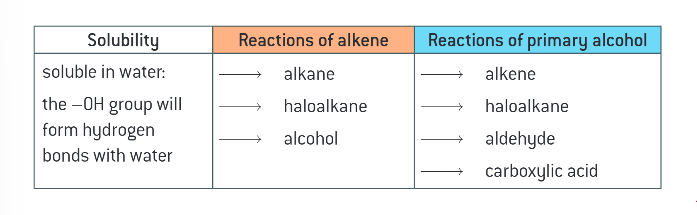
what is target molecule used to describe?
the compound that a chemist is attepting to prepare by organic synthesis
how can a target molecule be obtained in simple synthesis?
by reacting a readily available starting material with a readily available reagent in a 1 step reaction
what ay some organic synthesis routes require?
any steps to change the functional groups/ add carbon atoms to the chain length to obtain the target molecule
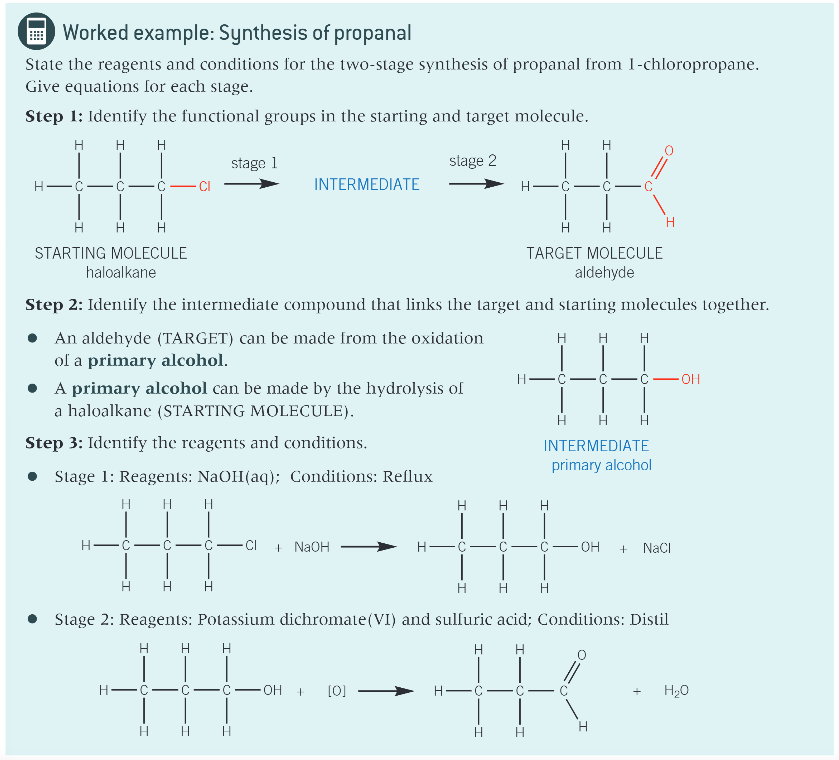
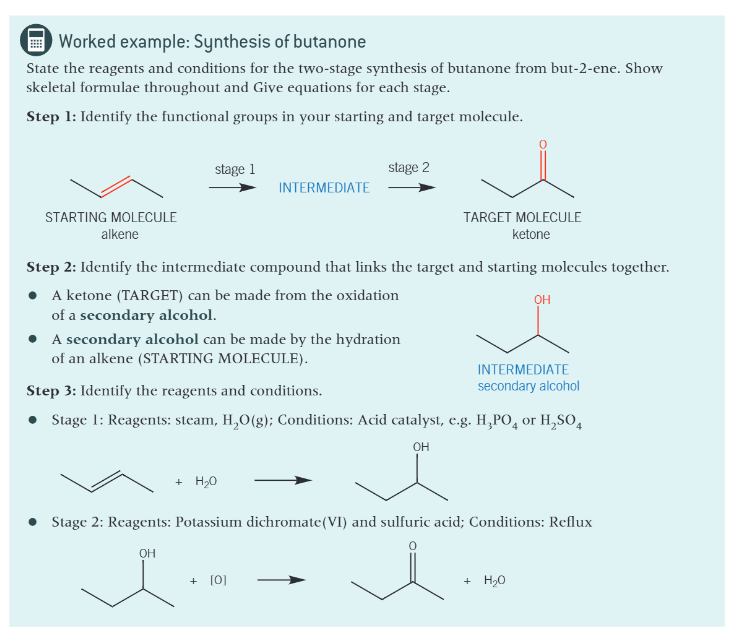
how do you convert a starting molecule into the target molecule?
identify the functional groups in your starting and target molecules
identify the intermediate that links the starting and target molecules
state the reagents and conditions for each step
worked example: synthesis of propanal:
worked example: synthesis of butanone:
worked example: predicting reactions:

extra info: does nature really provide a cure for everything?

extra info: ozonolysis