acid base week 1 sec 3
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
acid
proton donor (H+ ion) and forms its conjugate base
base
proton acceptor and forms its conjugate acid
bronsted-lowry acid/base must contain
H in its formula/lone pair of electrons to bond to H+
each side of acid/base reaction has
an acid and a base
acid-base reaction transfers
protons
arrhenius theory
acids dissociate in water to form H30+, bases dissociate in water to form OH-
acid ionization
proton exchange reaction between an acid (donating proton) and water
base ionization
proton exchange reaction between a base (accepts proton) and water
water is amphiprotic, meaning
can react as either an acid or a base
conjugate acid/base pairs differ by
an H+
autoionization of water
reaction where water is gaining and losing a proton by itself, with associated equilibrium constant Kw
Kw =
[H3o+][OH-]
if we disturb equilibria by adding base or acid
system responds i.e. increase in [H3O+] results in decrease of [OH]- and vice versa
acidic solution
[H3O+] > [OH-] and pH < 7
neutral solution
[H3O+] = [OH-] and pH = 7
basic solution
[H3O+] < [OH-] and pH >7
whether an aqueous solution is acidic or basic depends on
relative concentrations of the ions (both are present)
if equilibrium constant K for acid-base reaction is » 1
forward reaction favoring the products s.t. most of the acid HA will ionize to form A- and most of the base B will ionize to form HB+
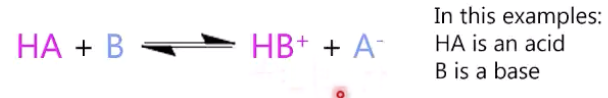
we compare acids and bases’s strength with
eq constant of the proton-exchange reaction of the acid/base with water
acidic strength
if eq constant is very large, stronger than water and vice versa
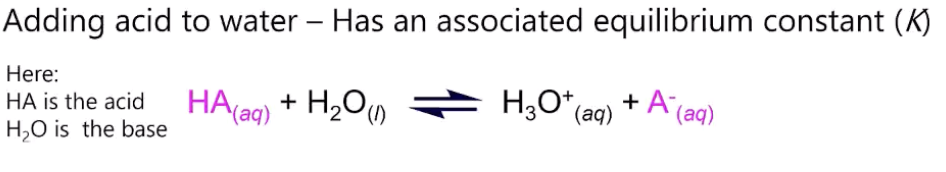
acid dissociation constant Ka
products/reactants i.e. [H3O+][A-]/[HA]
strong acid
Ka is irrelevant, no equilibria s.t. there is no HA remaining, complete dissociation into atoms
all strong acids are
equally strong in water
strong acids
HClO4, HCl, HBr, HI, HNO3, H2SO4
perchloric acid
HClO4
hydrochloric acid
HCl
hydrobromic acid
HBr
hydroiodic acid
HI
nitric acid
HNO3
sulfuric acid
H2SO4
weak acids leave
a measurable amount of HA at equilibrium, with the value of Ka indicating acid strength
low pKa corresponds to
high Ka and a stronger acid
basic strength
if eq constant is very large, stronger than water and vice versa
Kb, base ionization constant
[BH+][OH-]/[B]
strong base
completely ionizes to produce OH-
lithium hydroxide
LiOH
sodium hydroxide
NaOH
potassium hydroxide
KOH
calcium hydroxide
Ca(OH)2
strontium hydroxide
Sr(OH)2
barium hydroxide
Ba(OH)2
Ka x Kb =
kw
the stronger the acid is
the weaker its conjugate base
dilute strong acid
concentration is less than 10-6 M, so we also have to account for the water donating [OH-] or [H3O+]
polyprotic acid
has more than one ionizable proton s.t. in solution, each dissociation step has a different Ka value
for nonmetal hydrides, acid strength depends on
electronegativity of the central nonmetal and the bond strength
across a period or down a group, acid strength
increases (due to e-negativity increase and bond strength decrease, resp.)
oxo acids
oxygen between the nonmetal and H, their acid strength depends on e negativity and number of O atoms around central nonmetal
more electronegative atom bound to oxygen
higher acidity
oxoacids with same central atom comparison
more oxygen means higher acid strength
group 1 cations have
no acidic/basic properties
for concentrated solution of weak acid, [AH] » ka
[H3O+] = √[HA]initialKa
for concentrated solution of weak base, [B] » kb
[OH-] = √[BOH]initialKb