Unit 4 Exam-Styled Questions
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
B
Which of the following coordination complexes is expected to be the most inert toward ligand substitution?
a. [Ni(Cl)6]4-
b. [Ni(CN)6]3-
c. [Ni(NH3)6]2+
d. [Ni(CN)6]4-
e. [Ni(Br)6]4
D
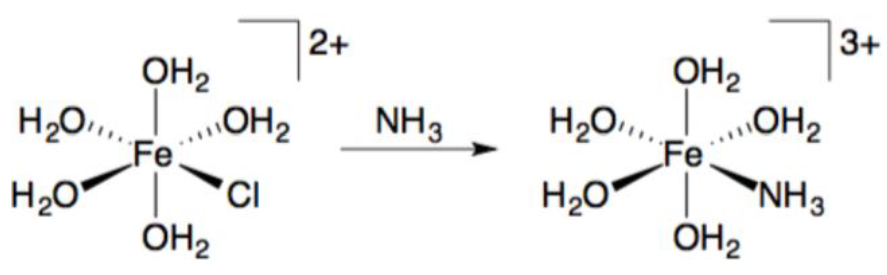
By replacing the H2O ligands in the complex shown below with P(CH3)3, the ligand exchange kinetics significantly increases in speed. What type of mechanism does this support?
a. Associative
b. Outer sphere electron transfer
c. Alkyl migration
d. Dissociative
e. CO insertion
C
Which complex below would you expect to have the highest thermodynamic stability
based on the chelate effect?
a. [Cu(H2O)6]2+
b. [Cu(NH3)4]2+
c. [Cu(en)3]2+
d. [Cu(Cl)4]2-
e. [Cu(en)(NH3)4]2+
C
Which statement best describes the inner sphere mechanism?
a. Electron transfer occurs without any direct interaction between the metal centers.
b. A very fast electron transfer reaction where no ligand is exchanged.
c. Typically requires a ligand to bridge the two metal centers, facilitating electron transfer.
d. Occurs only in square planar complexes.
e. None of the statements are correct.
D
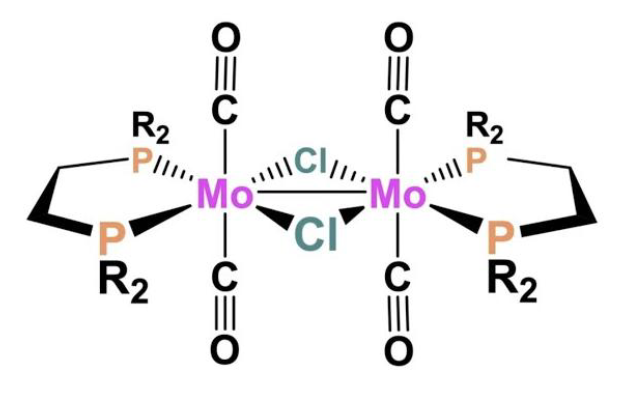
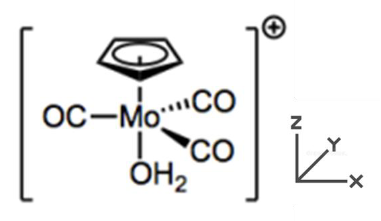
What is the total electron count of the coordination complex shown below and what is the oxidation state of Mo?
a. 16e- total, Mo2+
b. 18e- total, Mo2+
c. 12e- total, Mo3+
d. 18e- total, Mo1+
e. 16e- total, Mo1+
E
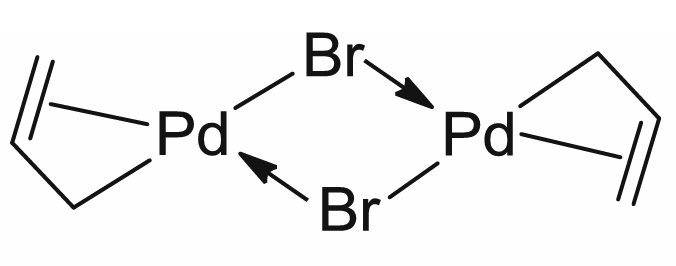
What is the total electron count of the coordination complex shown below and what is the oxidation state of Pd?
a. 18e- total, Pd0
b. 18e- total, Pd2+
c. 16e- total, Pd0
d. 14e- total, Pd0
e. 16e- total, Pd2+
B
What is the total electron count of the coordination complex shown below and what is the oxidation state of Ni?
a. 14e- total, Ni0
b. 16e- total, Ni2+
c. 16e- total, Ni0
d. 18e- total, Ni2+
e. 18e- total, Ni3+
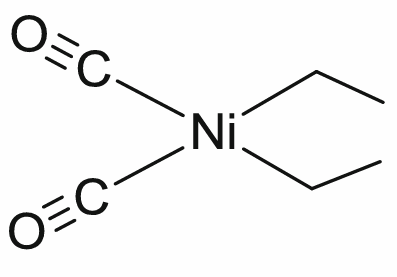
D
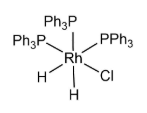
What is the total electron count of the coordination complex shown below and what is the oxidation state of Mo?
a. 16e- total, Rh2+
b. 18e- total, Rh2+
c. 20e- total, Rh3+
d. 18e- total, Rh3+
e. 16e- total, Rh0
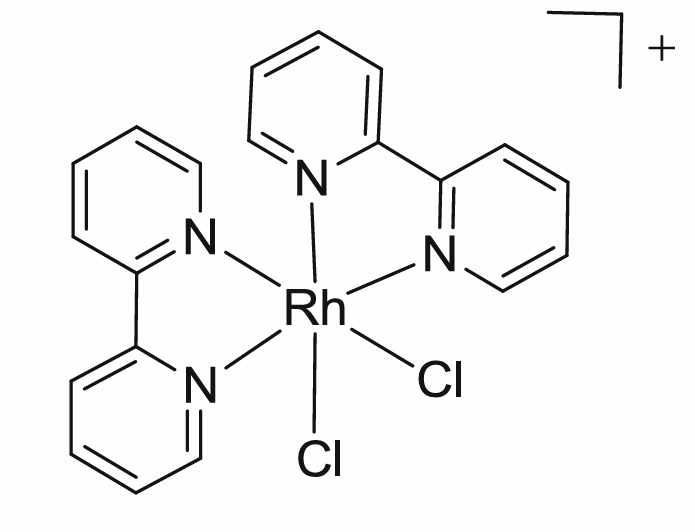
C
Which of the coordination complexes below will most likely have a square planar geometry?
a. [ZnCl4]2-
b. [Hg(CN)4]2-
c. [Ag(CN)4]1-
d. [Cd(CN)4]2-
e. [Ag(CN)6]3
E
Which orbital from the metal Mo center in the complex below will likely overlap with the pz orbitals in the Cyclopentadienyl ligand?
a. dyx
b. px
c. dx2-y2
d. py
e. dxz
A
What type of reaction is shown below?
a. Oxidative addition
b. Reductive elimination
c. Metathesis
d. Dissociation
e. Addition
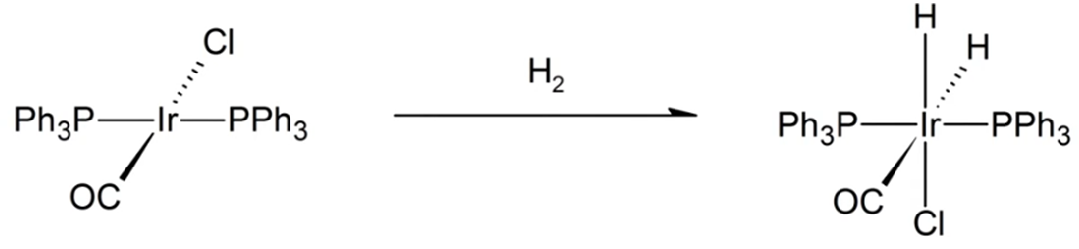
B
What type of reaction occurs in Step 1 and Step 2 as shown below?
a. Dissociation, oxidative addition
b. Reductive elimination, oxidative addition
c. β – elimination, oxidative addition
d. Dissociation, insertion reaction
e. Addition, reductive elimination
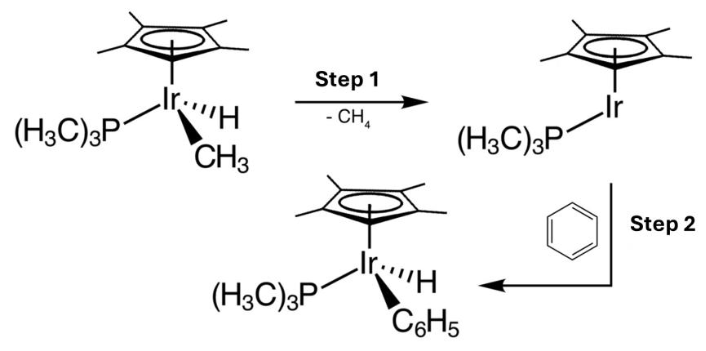
C
Which complex below is expected to have the longest C – O bond length?
a. [Mn(CO)6]+
b. Cr(CO)6
c. [Ti(CO)6]2-
d. [Fe(CO)6]2+
e. [V(CO)6]-
C
Which statement below correctly describes the trans effect?
a. The trans effect is the preference of strong σ-donor ligands to weaken the bond to the ligand cis to them, increasing its substitution rate.
b. The trans directing ligand strengthens the metal – ligand bond trans to it.
c. The trans effect is the ability of certain ligands, especially strong σ-donors or good π- acceptors, to increase the rate of substitution of the ligand positioned trans to them.
d. The trans effect is the preference of strong π -donor ligands to strengthen the bond to the ligand trans to them, increasing its substitution rate.
e. None of these statements are correct
E
Which complex below is most likely to undergo a metathesis reaction?
a. Ni(H2O)₄
b. Mn(CO)₃Cp
c. Pd(PPh₃)₄
d. Fe(CO)₅
e. TiCl₂Cp₂
E
The variable temperature 13C NMR of Ru(C6H6)2 shows only a single peak at room temperature and at lower temperatures. Which chemical formula best describes the hapticity of this complex based on the NMR spectrum?
a. Ru(η1-C6H6)2
b. Ru(η2-C6H6)2
c. Ru(η3-C6H6)2
d. Ru(η4-C6H6)2
e. Ru(η6-C6H6)2
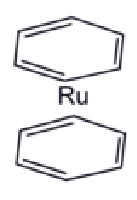
A
Suppose you have [Pt(Cl)4]2- as your starting material. What addition of reagents would result in having cis-[Pt(NH3)(NO2)2] as your product?
A. First, add NH3, then add [NO2]-
B. First, add 2NH3, then add [NO2]-
C. First, add [NO2]-, then add NH3
D. First, add [NO2]-, then add 2NH3
E. None of the above
C
Suppose you have [Pt(Cl)4]2- as your starting material. What addition of reagents would result in having trans-[Pt(NH3)(NO2)2] as your product?
A. First, add NH3, then add [NO2]-
B. First, add 2NH3, then add [NO2]-
C. First, add [NO2]-, then add NH3
D. First, add [NO2]-, then add 2NH3
E. None of the above
A
What reaction is shown in the picture?
A. reductive elimination
B. 1,1-insertion
C. oxidative addition
D. β-hydrogen elimination
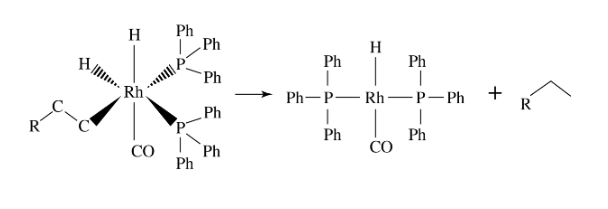
C
What reaction is shown in the picture?
A. reductive elimination
B. 1,1-insertion
C. oxidative addition
D. β-hydrogen elimination
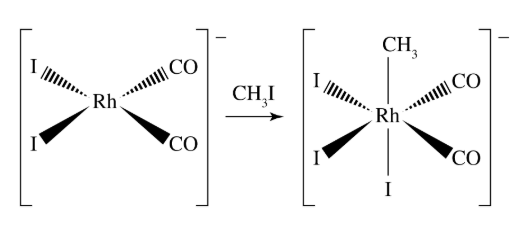
D
What reaction is shown in the picture?
A. reductive elimination
B. 1,1-insertion
C. oxidative addition
D. β-hydrogen elimination
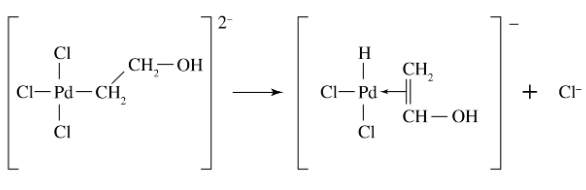
B
How would the rate change of the dissociative substitution reaction with the following scenario: the leaving group on [RhCl(OH2)5]2+ changes from Cl- to Br-?
A. The rate would increase
B. The rate would decrease
C. The rate would be unchanged
D. None of the above
C
How would the rate change of the dissociative substitution reaction with the following scenario: the entering group on [RhCl(OH2)5]2+ changes from Cl- to F-?
A. The rate would increase
B. The rate would decrease
C. The rate would be unchanged
D. None of the above
A
Consider the substitution reaction: [Pd(PMe3)4]2+ + 2 NH3) → ?
Select the product of the substitution reaction. Note that the rreaction will not be balanced.
A. cis-[Pd(PMe3)2(NH3)2]2+
B. trans-[Pd(PMe3)2(NH3)2]2+
C. [Pd(PMe3)4]2+
D. [Pd(NH3)4]2+
C
What is the electron count for the organometallic complex (η5-C5H5)2?
A. 12
B. 14
C. 16
D. 18
E. 20
A
[Co(CN)5]3- + [Co(NH3)4OH]2+ → [Co(CN)5OH]3- + [Co(NH3)5]2+
Determine what type of reaction is occurring in the scheme above.
A. inner-sphere mechanism
B. outer-sphere mechanism
C. metathesis
D. β-hydrogen elimination
B
[Ir6]2- + [Fe(CN)6]4- → [Ir6]3- + [Fe(CN)6]3-
Determine what type of reaction is occurring in the scheme above.
A. inner-sphere mechanism
B. outer-sphere mechanism
C. metathesis
D. β-hydrogen elimination
D
What is the dn count for the following complex?
Mo(CO)52-
A. 2
B. 4
C. 6
D. 8
E. 10
E
What is the dn count for the following complex?
Pt(PPh3)4
A. 2
B. 4
C. 6
D. 8
E. 10
C
What is the dn count for the following complex?
Mo(CO)52-
A. 2
B. 4
C. 6
D. 8
E. 10