Reaction Kinetics
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
Reaction Kinetics:
Rate at which reactants is converted into products
Rate at which reactant is
USED UP
Rate at which product is
FORMED
Rate Experimentations examples
1) Color intensity using a colorimeter: Br2, NO2 -> both decolorize with alkene
2) Conc. of reactant/product by titration
Quenching
slowing down/stopping a reaction to analyse
by:
1) rxn mixture to large V of cold solvent -> dec in K.E -> reduc in collision
2) rxn mixture + substance -> t recorded after quenching takes place
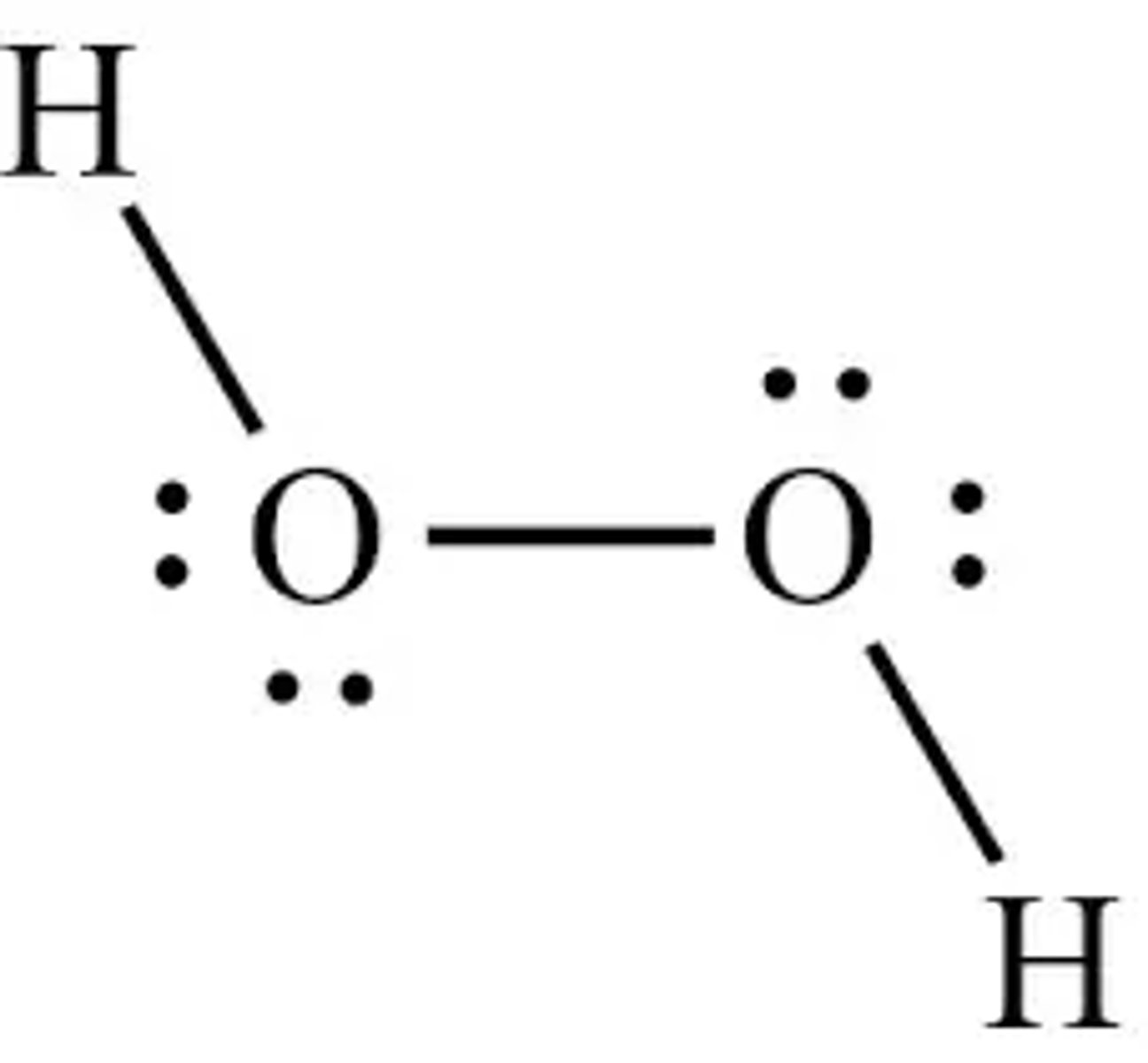
2H2O2 --> 2H2O + O2
Catalytic Decomposition
- Catalyst: MnO2
H2O2 STRUCTURE
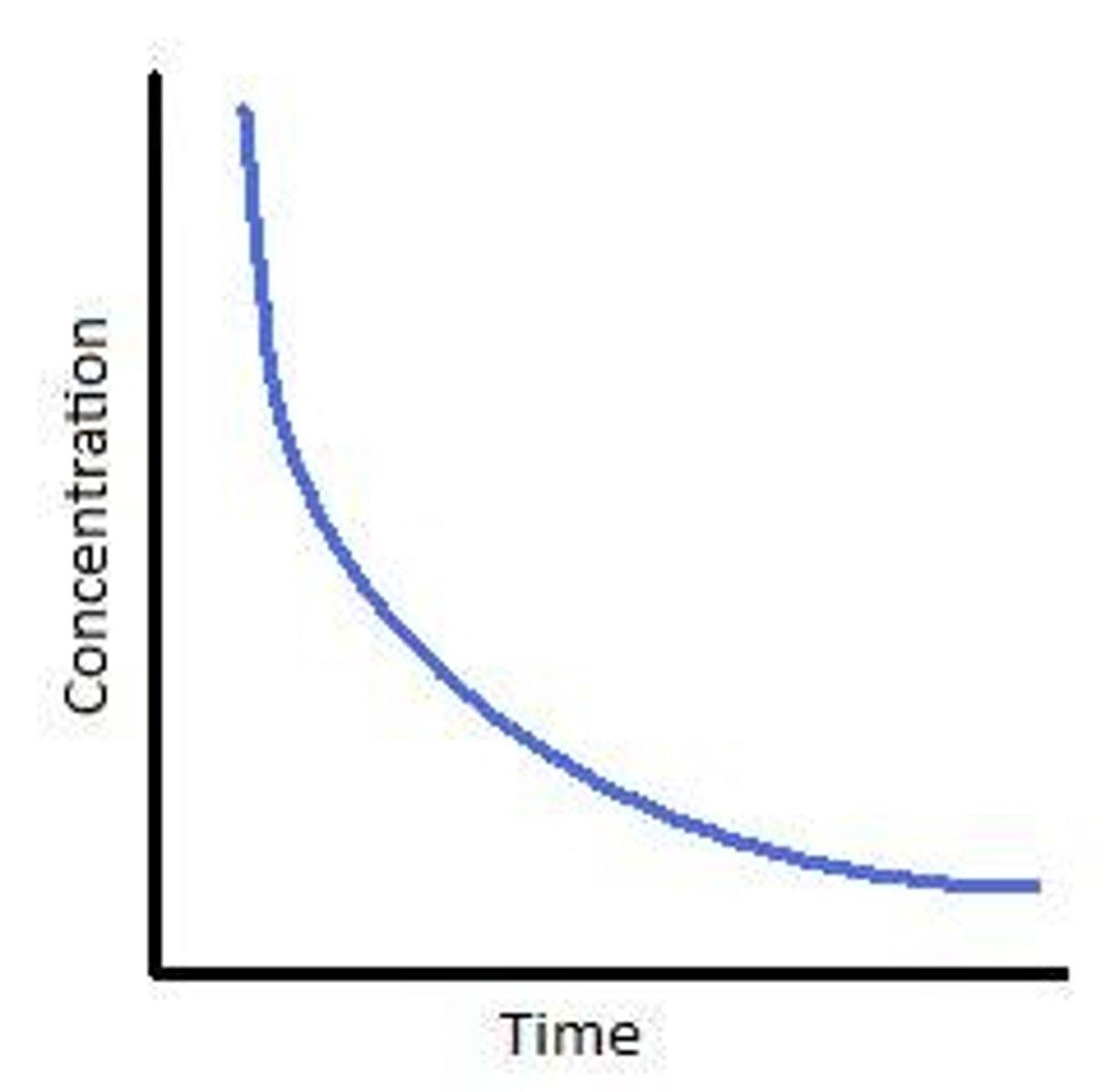
Rate of disappearance of reactant
-> exponential decrease graph -> negative gradient -> rate CANNOT be negative
-> Rate = - d[R]/dt
-> To include -ve sign in front of the rate equation
![<p>-> exponential decrease graph -> negative gradient -> rate CANNOT be negative</p><p>-> Rate = - d[R]/dt</p><p>-> To include -ve sign in front of the rate equation</p>](https://knowt-user-attachments.s3.amazonaws.com/c95b3266-4e20-4848-b107-1f78eaf8ab43.jpg)
Rate of formation of product
-> Exponential increase
-> Gradient = Positive : dy/dx = Rate
-> Rate = d(pX)/dt
Gradient of a concentration against time graph =
Rate
Units of rate = moldm-3s-1
Pressure (Nm-2) against time (s) ->
Rate = d(pX)/dt
Units: Nm-2s-1
Initial Rate
calculated by finding out the initial gradient of the graph
BrO3- (aq) + 5Br- (aq) + 5H+ (aq) ---> 3Br2 (aq) + 3H2O (l)
Rate at which reactants are used up:
-> exponential decrease graph -> negative gradient so - is added to make the rate positive
-> Rate = - d[BrO3-] / dt
-> Rate = -1/5 d[Br-]/dt
Rate at which products are formed:
-> Rate = + 1/3 d[Br2]/dt
-> +ve gradient -> exponential increase graph so positive gradient => positive rate
N2 (g) + 3H2 (g) ---> 2NH3 (g)....
Rate at which R is used up:
-> Rate = -d(pN2)/dt
-> Rate = -1/3 d(pH2)/dt
Rate at which P formed:
-> Rate = + 1/2 d(pNH3)
Rate of Reactants being used UP (GRAPH)
Rate of products being FORMED - Graph
Rate equation is NOT determined by the overall reaction, but by the:
- slow rate determining step
How to calc. initial rate of RXN?
Via rate eq. using initial reacnt conc + catalyst
To alter the proportionality constant (K)
- Presence/absence of catalyst
- Temperature
Constant of proportionality (K)
K is equal to the RATE of RXN when all R substances concentrations are equal to 1
Rate = k[R]
[R] = 1, SO RATE = K
Order wrt substance
the power of the conc. term of the substance in the rate equation
overall order
the sum of the powers of conc. terms in rate eq
CH3COCH3 + I2 ----> CH3COCH2I + HI
Rate = k[CH3COCH3][H+]
I2 is not involved in the R.D.S -> has no effect on the initial rate
Units of k when rate = k(pNO2)^2
Nm+2s-1
To determine the rate equation:
1) mechanism -> note the R.D.S --> slow step --> rate equation (initial reactants)
2) experimental data
NO2 + CO ---> NO + CO2
(1) 2NO2 ---> NO + NO3 (SLOW)
(2) NO3 + CO ----> NO2 + CO2 (FAST)
Rate = k[NO2] ^2
(CH3)3Cl + OH- ----> (CH3)3OH + Cl- (via reflux)
-> Tertiary halogenoalkane w/ OH- (Nucleophilic Subs.)
- SN1 MECHANISM
-> Only (CH3)3Cl involved before the transition state => 1 SPECIES INVOLVED BEFORE T.STATE
- Compound breaks down heterolytically (C-Cl)
- C--Cl bond weakens
- Carbocation forms + OH- joins
- Alcohol forms, byproduct = Cl-
Rate = k[(CH3)3Cl]
-> Slow step between compound and T.S
CH3Cl + OH- ---> CH3OH + Cl- (Primary halogenoalkane --> SN2)
BImolecular Nucleophilic Subs. -> 2 species involved before transition state
1) OH- attacks the C partial positive atom
(SLOW)
2) Both Cl and OH are attached
3) Fast: Alcohol forms and Cl- released by product
Rate = k[CH3Cl][OH]
Between EXP 1 and 2: concentration of A doubles, concentration of B is constant and rate doubles:
2:2
1:1
Order with respect to [A] = 1
BETWEEN EXP 2 AND 3: Conc of A doubles, Conc of B doubles, and rate quadruples
-> Conc A is 1st order (Conc x2, Rate x2)
-> Conc B => x2 conc, x2 rate => 1st order
BETWEEN EXP 1 AND 2: Conc of [H] doubles, [C] stays constant, rate doubles,
2:2
1:1
-> [H] is first order
EXP 2 TO 3: Conc of [H] is constant, conc of [C] doubles, rate quadruples
2: 4
1: 2
2nd order
2X + Y ---> Z
Exp 1 to 2:
- Initial X doubles, Initial Y constant, Rate quadruples
2:4, 1:2, 2nd order wrt to X
Exp 2 to 3:
-> X doubles, Y doubles, Rate quadruples
X => second order
as conc doubles, rate quadruples, so Y has no effect
Y order = O
Thus, Rate = k[X]^2
First order
concentration doubles, rate doubles (whilst other reactants are constant)
Second order
Concentration doubles, rate quadruples (whilst other reactants are constant)
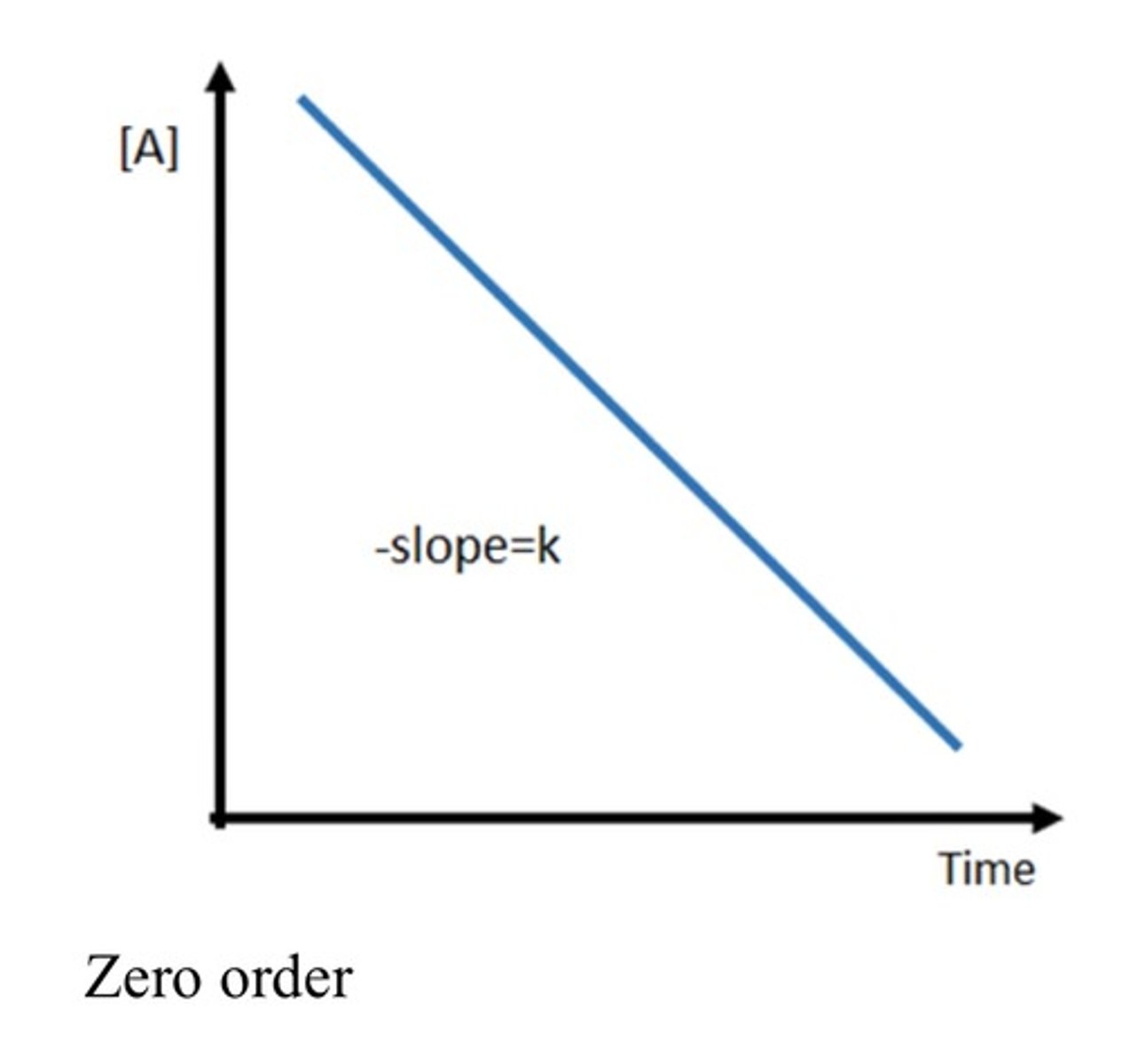
Zero order
no effect on the rate of rxn
1st/2nd order graph of concentration against time
-> Exponential curve decrease graph
To decipher between 1st order and 2nd order
1st order: constant half life
First T 1/2 = 200, second T 1/2 = 200
2nd order = no constant half life
- Changing T 1/2
Graph of concentration against t for zeroth order
Negative linear graph
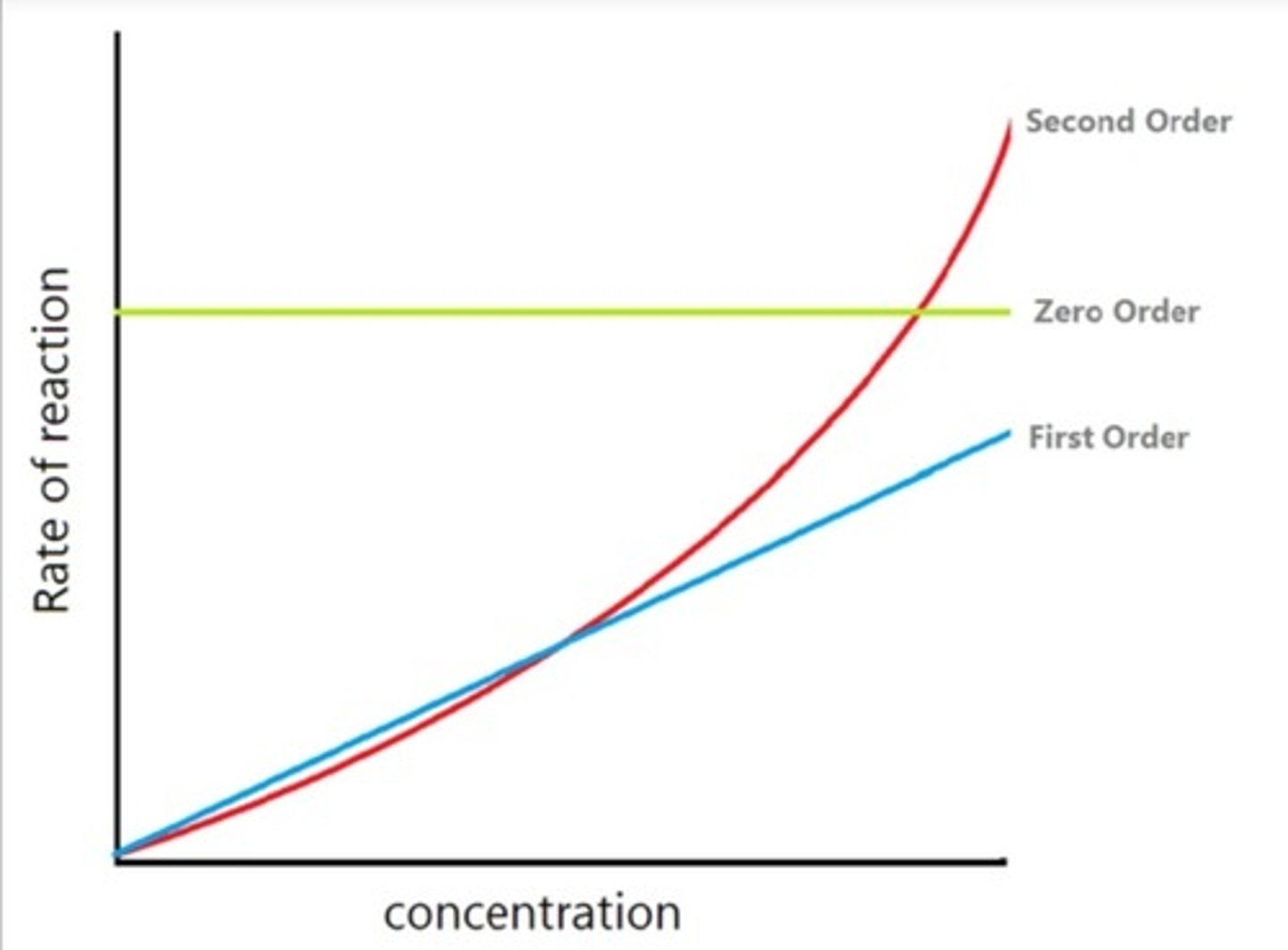
Rate against concentration graphs
-> second order: curve upwards
-> first order: linear upwards
-> zero order: horizontal
Rate of RXN =
depends on no. of collisions
For molecules to react
1) necessary Ea
2) favourable orientation
Ea
minimum amount of energy which must be possessed by the reactant molecules so that a collision w/ favorable orientation results in a chemical RXN
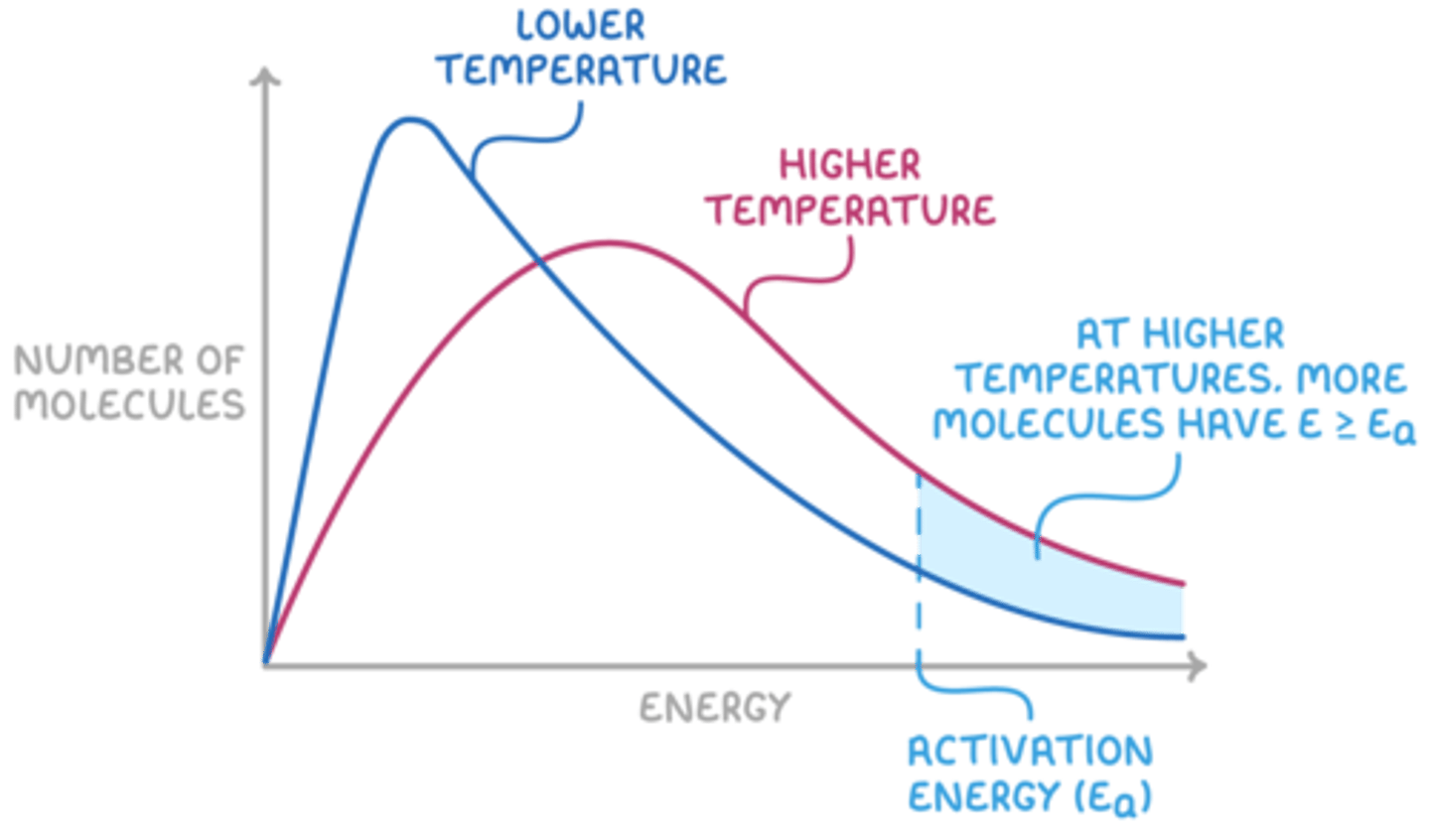
Maxwell Distribution Curve
-> inc in T -> Shifts graph to RHS -> more molecules w/ fav orientation + nec. Ea
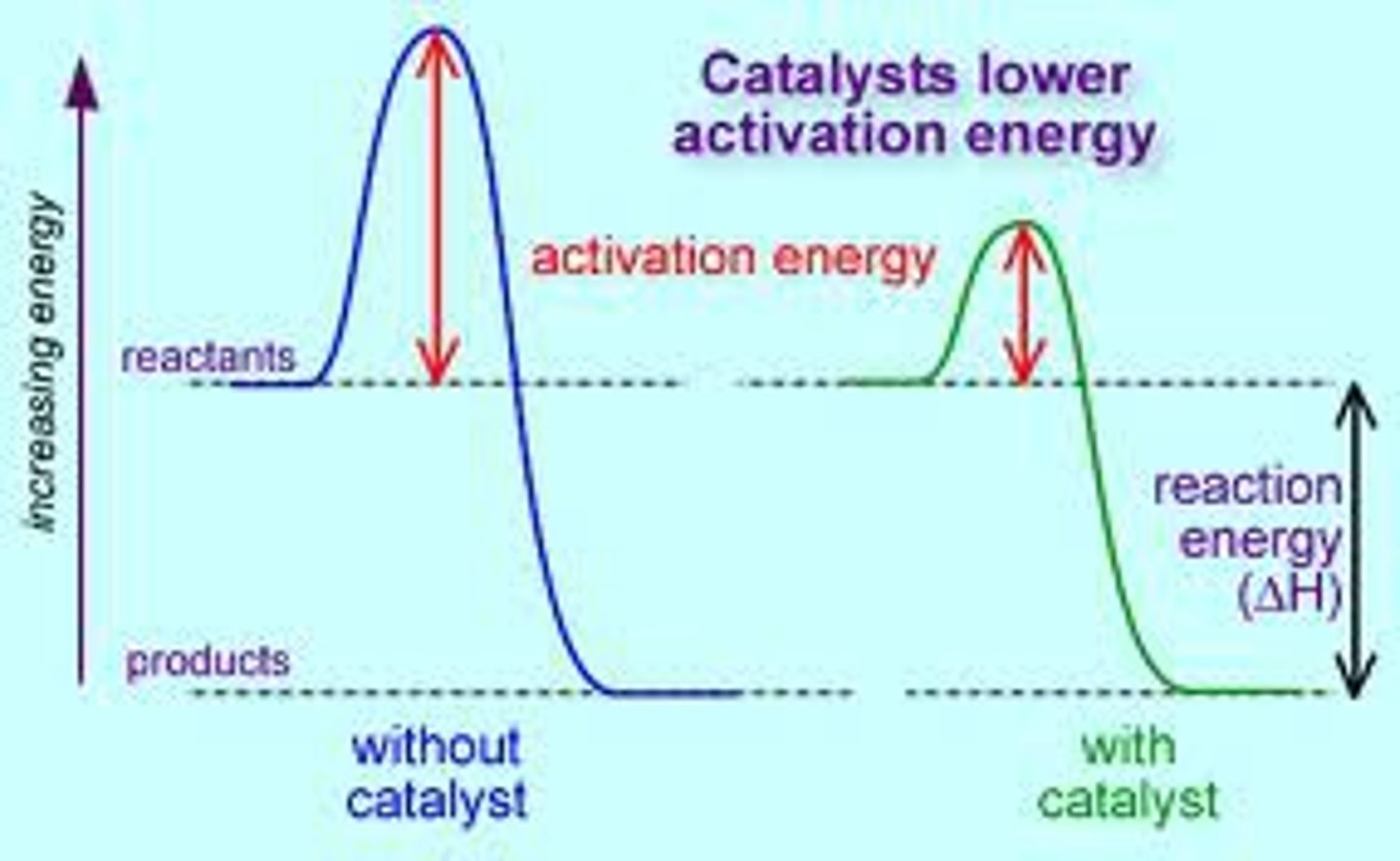
Catalysts
substances which LOWER the activation energy by providing an alternative mechanism to speed up the rate of reaction
-> Lower Ea --> more molecules w/ necessary Ea and fav. orientation
Arrhenius Equation
k = rate constant
A = frequency factor/ pre exponential factor
Ea = activation energy
e- = base of natural log
R = Constant
T = temp
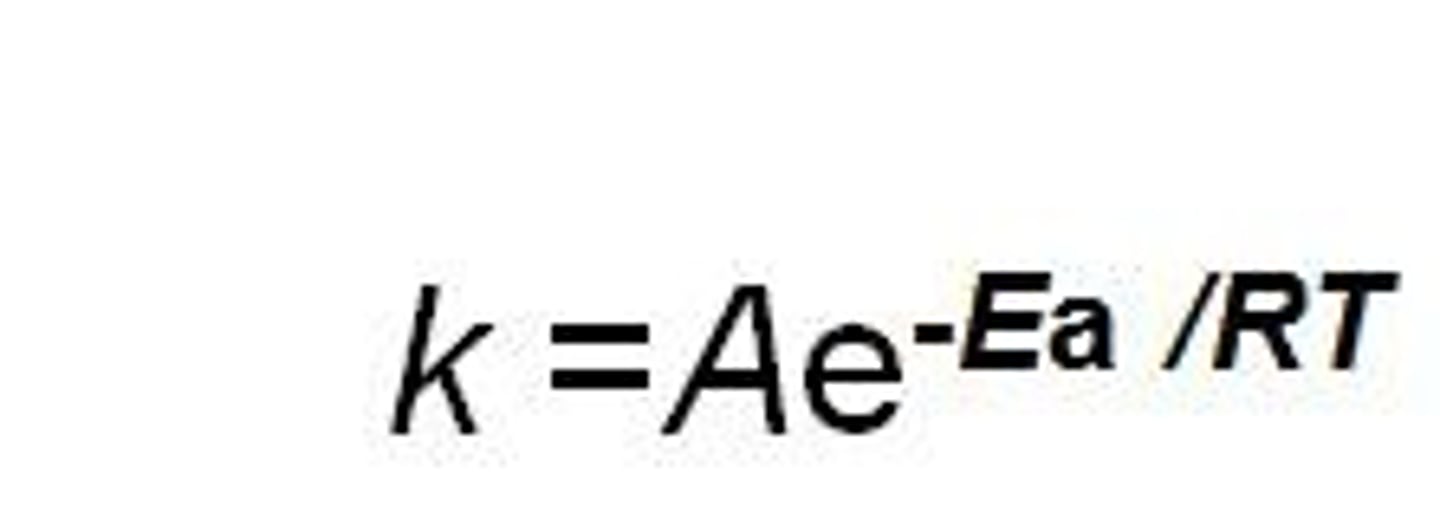
Inc in P
- no effect on Ea
- inc the no. of collisions -> inc in rate
AUTOCATALYSIS
catalysis in which one of the products of the reaction acts as a catalyst for the reaction
- Acid manganate (VII)
- Ethanedioate ions
Ethanedioic acid
primary standard -> preps for standard solns
[MnO4-] against time
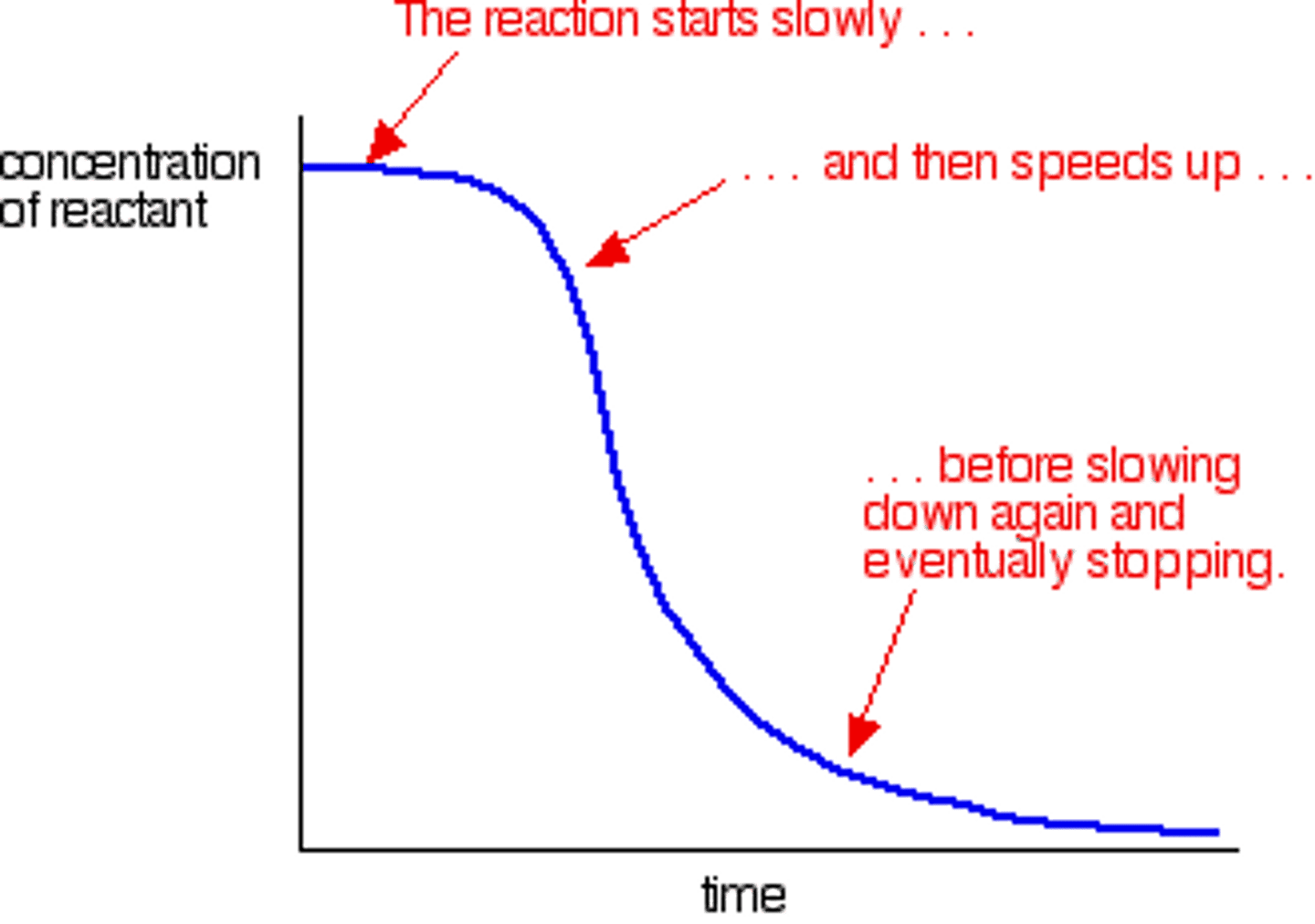
[Mn2+] against time
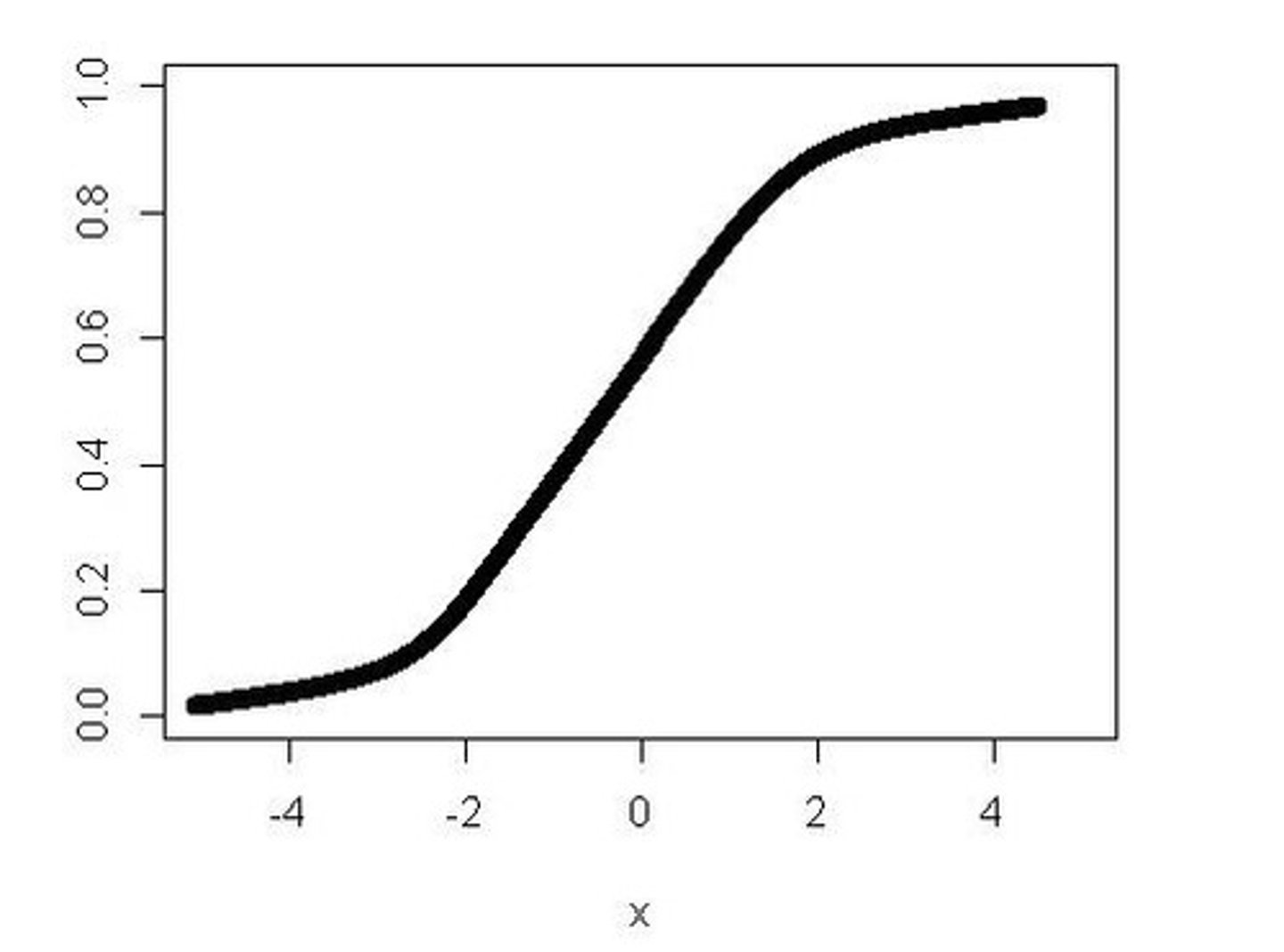
Catalytic convertor
- mix of metals
- harmful products from car exhausts -> harmless ones
Three way catalytic convertor structure
honey comb -> inc S.A for more efficiency
cat.convertor:
CO -> CO2
Unburnt hydrocarbons (octane ) --> CO2 + H2O
NO/NO2 ----> N2
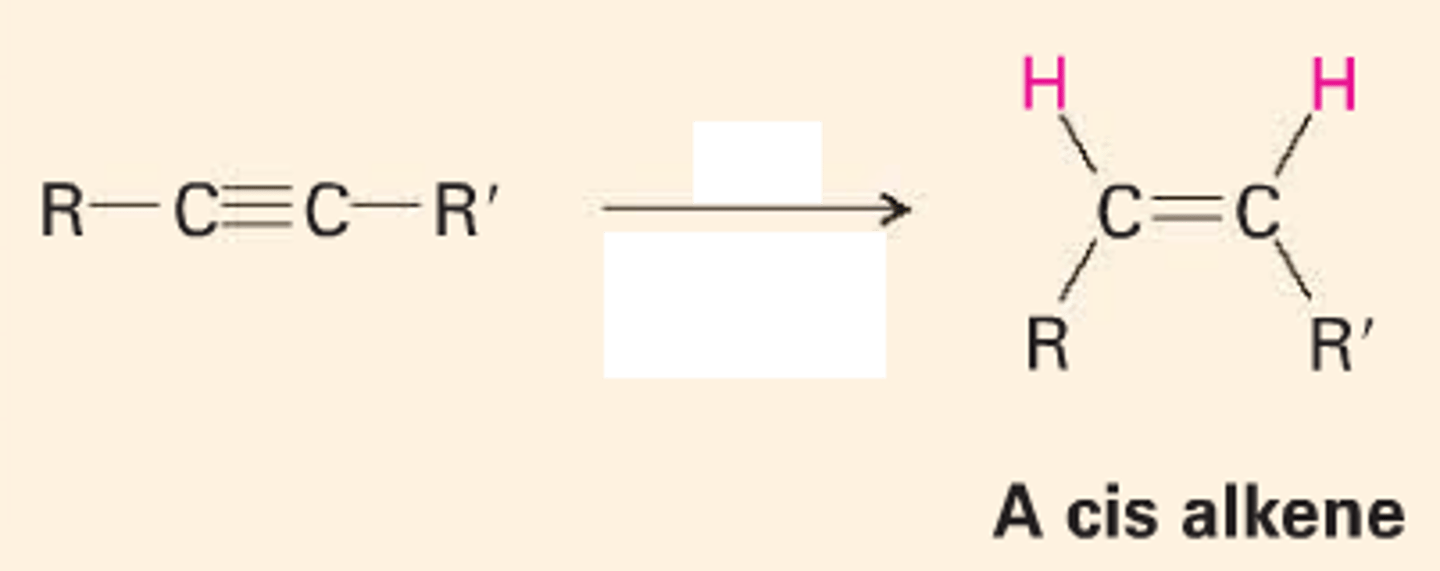
Lindlar Catalyst
Lindlar's catalyst is a poisoned palladium metal catalyst that performs partial hydrogenation of alkynes in the presence of hydrogen gas.
-> Palladium deposited on CaCO3 and poisoned with Pb and S
-> alkyne -> alkene