Chapter 2: Chemical Bonding
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
Electronegative
Definition:
Electronegativity of an atom is a measure of its ability to attract the electrons in a covalent bond to itself.
Same electronegativity:
In H2, the bonding electrons are equally shared between the two hydrogen atoms as they have the same electronegativity.
Different electronegativity:
When the covalent bond is formed by two different atoms of different electronegativities, the bonding electrons are not shared equally.
For example in hydrogen chloride molecule (HCl), electron density is pulled closer to the more electronegative Cl atom.
Factors affecting Electronegativity
Across the Period:
Nuclear charge increases while shielding effect remains relatively constant.
Effective nuclear charge increases
Hence, electronegativity INCREASES across the period
Down the Group:
Elements in the same group experience roughly the same effective nuclear charge as both nuclear charge and shielding effect increase down the group.
However, as the number of quantum shells increases, atomic radius increases.
Hence, electronegativity DECREASES down the group
Most electronegative atom: F (fluorine)
Electronegativity and Types of Chemical Bonding
Covalent bonds
Formed between atoms of non-metals of similar electronegativity.
Ionic bonding
Occurs between a metal and non-metal with a large difference in electronegativity
Most chemical bonds are neither purely covalent nor purely ionic but have characteristics of both ionic and covalent bonding
ie: it is possible for a metal and non-metal to form covalent bonds (rather than ionic bonds)
Eg: Bonds in AlCl3 are considered covalent
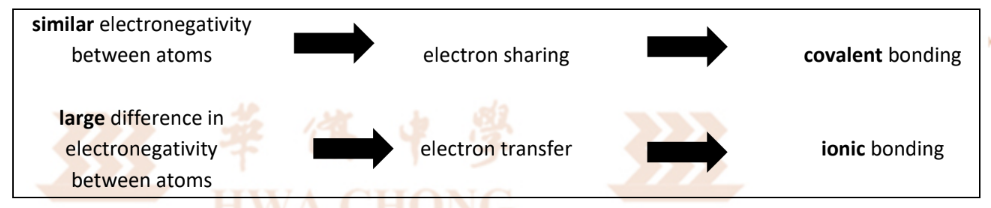
Types of Electrostatic Attractions
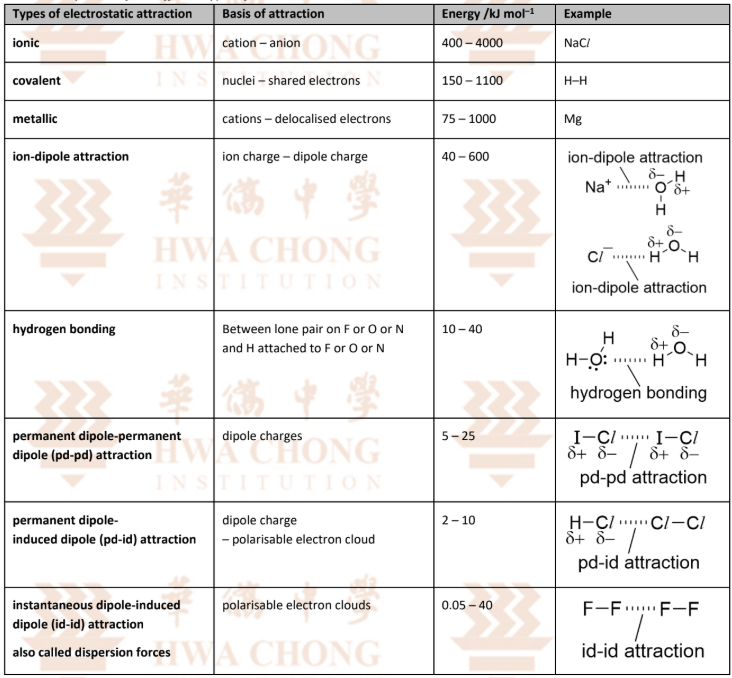
Metallic Bonding — Metallic Lattice
Metals in the solid state have a giant metallic lattice structure which is a lattice of metal cations and the sea of delocalised electrons held together by metallic bonding.
Definition of Metallic Bond:
Metallic bond is the electrostatic attraction between a lattice of positive ions and the sea of delocalised electrons.
Arrangement:
In solid (metal), (cation) cations are arranged in a regular manner.
Each (metal) atom contributes ____ valence electrons into the sea of delocalised electrons.
These delocalised electrons are no longer attracted to only one specific atom but can move freely throughout the lattice.
These delocalised electrons are responsible for the characteristic physical properties of metals such as good electrical and thermal conductivity.
Metallic Bonding — Metallic Lattice (Drawing)
Note: The metal is electrically neutral as the number of positive and negative charges balance out.
Metallic Bonding — Strength of Metallic Bonding
Number of valence electrons contributed per metal atom:
The larger the number of valence electrons contributed per atom, the greater the number of delocalised electrons, the stronger the metallic bonding.
Charge and radius of the metal cation:
The higher the charge and the smaller the radius of the metal cation, the higher its charge density*, and the stronger the metallic bonding.
*Charge density of an ion, sometimes called surface charge density, is the amount of charge per unit surface area of that ion. For simplicity, we can compare the charge densities of two ions by comparing their ionic charge / ionic radius ratio.
*Radius increases down the group, decreases across period
Metallic Bonding — Physical Properties
High Melting and Boiling Points
Metals have high melting and boiling points as a large amount of energy is needed to overcome the strong attraction between the metal cations and the delocalised electrons.
The stronger the metallic bonding, more energy is needed to overcome the metallic bonding, the higher the melting and boiling points.
When a solid metal melts, the closely packed cations become further apart but the metallic bonding is not completely broken, unlike during boiling. Hence, the boiling point is a better indicator of the strength of metallic bonding than the melting point.
Good electrical and thermal conductivity
In order for a material to conduct electricity, it must have mobile charge carriers which are typically electron or ions.
Metals are good electrical conductors because the delocalised electrons act as mobile charge carriers.
The delocalised electrons also explain why metals are good conductors of heat.
Thermal energy causes the electrons to move more quickly.
The energy is then transferred to the other parts of the metal by the mobile electrons.
Malleable and Ductile
Metals are usually malleable (easily beaten or rolled into sheets) and ductile (easily drawn into rods, wires and tubes).
When a large stress (e.g. hammering) is applied to a piece of solid metal, the layers of ions will slide over one another into new positions.
The overall shape of the metal changes (the metal deforms) but it does not break apart because the 'sea' of delocalised electrons prevents repulsion between the cations as they move past one another, and metallic bonding remains intact.
Hardness
The hardness of a solid material depends on how easily we can move the particles from their fixed positions in the solid lattice.
In metals, this depends on the strength of metallic bonding.
Metals are usually hard; EXCEPTION: Group 1 and 2 metals are soft.
Iron is hard and does nor deform easily while sodium can be easily cut with a pen knife.
Metallic Bonding — Alloy
What is an alloy:
Alloys are mixtures of metals involving incorporation of small quantities of other element(s) into the pure metal.
Alloying makes metals harder:
This is because atoms of the added element(s) have a different size.
This will disrupt the orderly arrangement of the main metal atoms such that they can no longer slide over each other easily when a force is applied.
This increases the hardness and strength of the alloy compared to the pure metal.
Ionic Bonding — Ionic Lattice
Structure:
Ionic compounds in the solid state have a giant ionic lattice structure which is a lattice of cations and anions held together by ionic bonding.
Definition of Ionic Bonding:
Ionic bond is the electrostatic attraction between oppositely charged ions.
Occurrence of Ionic Bonding:
Ionic bonding exists between any two adjacent ions of opposite charge.
Ionic VS Covalent:
Ionic bonding is non-directional and extends indefinitely in all directions
Covalent bond is directional as one covalent bond can only be formed between two specific atoms.
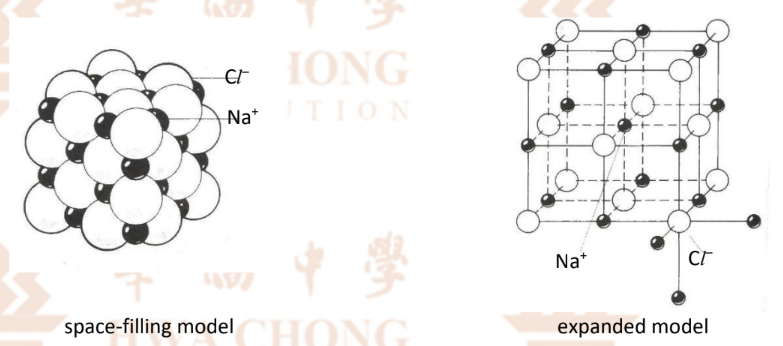
Ionic Bonding — Diagrams
Note: Cl- ion is larger than Na+ ion as Na+ loses an electron, which reduces its shielding effect, making the attraction between the valence electrons and the nucleus stronger so it has a smaller radius
Ionic Bonding — Coordination Number
Definition:
The number of ions that surround another ion (central ion) of the opposite charge in an ionic lattice is called the coordination number of that central ion.
Example:
In sodium chloride, one Na+ cation is surrounded by six Cl– anions and vice-versa so the coordination number is six
Coordination number depends on:
Relative sizes → a small ion typically has a lower coordination number as there will be less space for ions of the opposite charge to surround it
Relative charges → e.g. a cation with +2 charge needs twice as many anions with –1 charge, compared to a cation with +1 charge, to gain charge neutrality.
Ionic Bonding — Strength of Ionic Bonding
Dependent on lattice energy:
Larger the magnitude of lattice energy, the more heat is given out when the ionic bonds are formed, the stronger the ionic bonding.
Definition of Lattice Energy:
Lattice energy is defined as the heat evolved when 1 mole of a pure ionic solid is formed from its constituent
It is always -ve (exothermic)
Calculation of Lattice Energy:
Depends on the charge of the ions, and their radii.
Higher the charge and the smaller the radii of the ions, the larger the magnitude of lattice energy → Stronger ionic bonding
Remember to quote lattice energy formula when answering questions
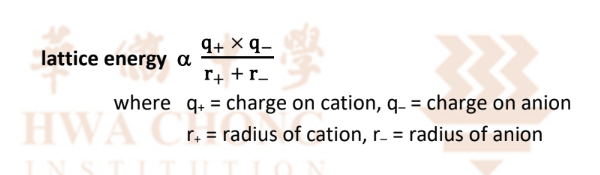
Ionic Bonding — Physical Properties of Ionic Compounds
High melting and boiling points:
Ionic solids have high melting and boiling points as a large amount of energy is needed to overcome the strong electrostatic attractions between oppositely charged ions.
The stronger the ionic bonding, more energy is needed to overcome the ionic bonding, the higher the melting and boiling points (look at lattice energy)
Good electrical conductivity:
In the solid state, ionic compounds do not conduct electricity as the ions are in fixed positions and are not free to move.
In the molten or aqueous state, ionic compounds are good electrical conductors because the ions can act as mobile charge carriers.
The electrical conductivity of an aqueous solution of an ionic compound increases as its concentration increases, because there are more ions to carry the charge.
Hard and rigid but brittle:
In an ionic lattice, oppositely charged ions are held in fixed positions throughout the crystal lattice by strong ionic bonding.
Moving the ions out of position requires large amounts of energy to overcome these bonds.
Ionic lattices are therefore quite hard (do not dent or deform easily) and rigid (do not bend easily).
If a strong enough force (e.g. by cutting or knocking) is applied, it will force ions of like charges to move next to each other. Repulsion between ions of like charges will cause the lattice to shatter. Ionic lattices are therefore brittle (crack without deforming).
Solubility:
Ionic compounds are usually soluble in water, e.g. NaCl, KBr and BaCl2.
However, there are examples of insoluble ionic compounds as well, e.g. AgCl.
Covalent Bonding — Bonding
Definition of Covalent Bond:
Covalent bond is the electrostatic attraction between the positively charged nuclei (of both bonded atoms) and their shared electrons.
Occurrence of Covalent Bond:
A covalent bond is formed when two atoms share a pair of electrons which are found in the valence orbitals of the atoms.
Covalent Bonding — Orbital Overlap
To form covalent bond:
In order for the bond to be formed, the orbitals of the two atoms must overlap.
The shared electrons or bond pairs occupy the region between the two nuclei formed by the overlap.
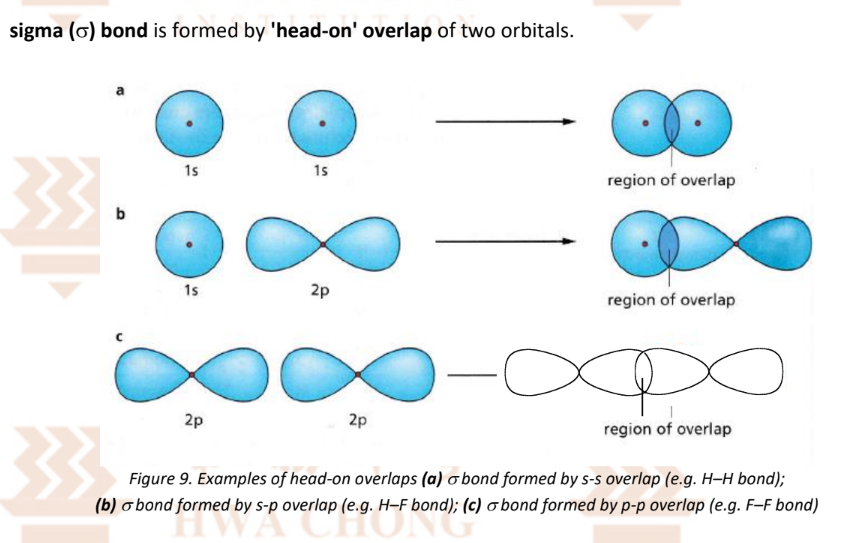
2 Types of Orbital Overlaps:
Sigma bond → 'head-on' overlap
Pi bond → 'side-on' overlap
Covalent Bonding — Sigma Bonds (‘head-on’ overlap)
s orbitals can only overlap head-on
Between 2 atoms, there can only be one sigma bond as it is not possible for a second pair of orbitals to overlap head-on as well.
Note: Shade the overlap and label it as head-on overlap
Note: Label the orbitals as: 1s / 2s../ 2p / 3p… orbital of (atom)
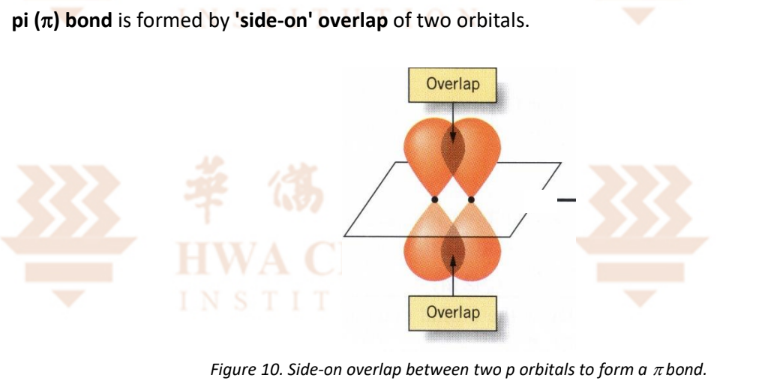
Covalent Bonding — Pi Bonds (‘side-on’ overlap)
p orbitals can overlap head-on or side-on
Side-on overlap for a pi bond is poorer than head-on overlap for a sigma bond, as the degree of overlap is smaller for the former.
Hence, a pi bond is weaker than a sigma bond.
Note: Shade the overlap and label it as head-on overlap
Note: Label the orbitals as: 2/3..p orbital of (atom) and 2/3…p orbital of (atom)
pi bond is formed only after a sigma bond is formed between 2 atoms
Single bond consists of a sigma bond (the two atoms share one pair of electrons)
Double bond consists of one sigma bond and one pi bond (the two atoms share four electrons)
Triple bond consists of one sigma bond and two pi bonds (the two atoms share six electrons)
Covalent Bonding — Strength of Covalent Bonding
Bond strength depends on:
Degree of orbital overlap between the two bonded atoms
How strongly the electrons in the overlap region are attracted to the nuclei of the atoms.
Bond energy:
It is the average amount of energy required to break 1 mole of a covalent bond in the gaseous state to form gaseous atoms.
Always endothermic
The larger the bond energy, the more heat is needed to break the bond, the stronger the covalent bonding.
Factors affecting Bond Energy:
Bond Length
Distance between the nuclei of the two bonded atoms
Shorter bond length = Stronger bond
Smaller atoms form shorter and stronger bonds, as the overlap between orbitals is more effective → Bond energy increases → Bond strength increases
Exception: